Abstract
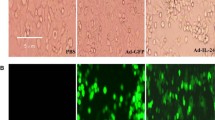
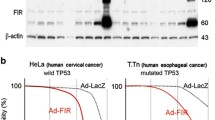
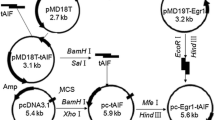
Radio genetic therapy which combines gene therapy with radiotherapy has shown promising results in cancer treatment. In this study, an oncolytic adenovirus-based gene therapy system regulated by radiation was constructed to improve the cancer curative effect. This gene therapy system incorporated the radiation-inducible early growth response gene (Egr-1) promoter and the anticancer gene tumor necrosis factor-related apoptosis-inducing ligand (TRAIL). To confirm the antitumor effect of Ad-ET combined with \(^{12}\hbox {C}^{6+}\) ion irradiation, the survival and apoptosis fraction of tumor cells HT1080 and normal cells MRC-5 in combination treatment were detected by CCK-8 assay and FACS analysis. Then the expression levels of TRAIL gene and protein were tested by real-time PCR and western blotting. The results show that \(^{12}\hbox {C}^{6+}\) ion irradiation could induce cell growth inhibition and apoptosis by activating the TRAIL gene expression in tumor cells, while exhibiting no obvious toxicity to the normal lung cell line MRC-5. The results also demonstrate that use of an oncolytic adenovirus-based radiation-inducible gene therapy system together with \(^{12}\hbox {C}^{6+}\) ion irradiation could cause synergistic antitumor effect specifically in tumor cells but not in normal cells. The results indicate that the novel radio genetic therapy could potentiate radiation treatment by improving the safety and efficiency of monotherapy, and provide theoretical support for clinical application of combination treatment.




Similar content being viewed by others
References
I.M. Verma, N. Somia, Gene therapy-promises, problems and prospects. Nature 389, 239–242 (1997). doi:10.1038/38410
M.L. Edelstein, M.R. Abedi, J. Wixon et al., Gene therapy clinical trials worldwide 1989–2004-an overview. J. Gene. Med. 6, 597–602 (2004). doi:10.1002/jgm.619
R.D. Alvarez, J. Gomez-Navarro, M. Wang et al., Adenoviral-mediated suicide gene therapy for ovarian cancer. Mol. Ther. 2, 524–530 (2000). doi:10.1006/mthe.2000.0194
K. Garber, China approves world’s first oncolytic virus therapy for cancer treatment. J. Natl. Cancer. Inst. 98, 298–300 (2006). doi:10.1093/jnci/djj111
H. LeBlanc, A. Ashkenazi, Apo2L/TRAIL and its death and decoy receptors. Cell Death Differ, 10, 66–75 (2003). doi:10.1038/sj.cdd.4401187
A. Ashkenazi, R. Pal, S. Fong et al., Safety and antitumor activity of recombinant soluble Apo2 ligand. J. Clin. Invest. 104, 155–162 (1999). doi:10.1172/JCI6926
G. Pan, G. Ni, Y.F. Wei et al., An antagonist decoy recep-tor and a death domain-containing receptor for TRAIL. Science 277, 815–818 (1997). doi:10.1126/science.277.5327.815
T. Hori, T. Kondo, M. Kanamori et al., Ionizing radiation enhances tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced apoptosis through up-regulations of death receptor 4 (DR4) and death receptor 5 (DR5) in human osteosarcoma cells. J. Orthop. Res. 28, 739–745 (2009). doi:10.1002/jor.21056
A.M. Chinnaiyan, U. Prasad, S. Shankar et al., Combined effect of tumor necrosis factor-related apoptosis-inducing ligand and ionizing radiation in breast cancer therapy. Proc. Natl. Acad. Sci. 97, 1754–1759 (2000). doi:10.1073/pnas.030545097
W. Liu, E. Bodle, J. Chen et al., Tumor necrosis factor-related apoptosis-inducing ligand and chemotherapy cooperate to induce apoptosis in mesothelioma cell lines. Am. J. Respir. Cell Mol. 25, 111–118 (2001). doi:10.1165/ajrnmeterb.25.1.4472
P. Todd, Heavy-ion irradiation of cultured human cells. Radiat. Res. Suppl. 7, 196–207 (1967). doi:10.2307/3583713
T. Kanai, M. Endo, S. Minohara et al., Biophysical characteristics of HIMAC clinical irradiation system for heavy-ion radiation therapy. Int. J. Radiat. Oncol. 44, 201–210 (1999). doi:10.1016/S0360-3016(98)00544-6
H. Tsujii, J. Mizoe, T. Kamada et al., Clinical results of carbon ion radiotherapy at NIRS. J. Radiat. Res. 48, A1–13 (2007). doi:10.1269/jrr.48.A1
H. Tsujii, J. Mizoe, T. Kamada et al., Overview of clinical experiences on carbon ion radiotherapy at NIRS. Radiother. Oncol. 73, S41–S49 (2004). doi:10.1016/S0167-8140(04)80012-4
J.R. Castro, D.H. Char, P.L. Petti et al., 15 years experience with helium ion radiotherapy for uveal melanoma. Int. J. Radiat. Oncol. 39, 989–996 (1997). doi:10.1016/S0360-3016(97)00494-X
A. Gashler, V.P. Sukhatme, Early growth response protein 1 (Egr-1): prototype of a zinc-finger family of transcription factors. Prog. Nucleic Acid Res. Mol. Biol. 50, 191–224 (1995). doi:10.1016/S0079-6603(08)60815-6
R. Datta, E. Rubin, V. Sukhatme et al., Ionizing radiation activates transcription of the EGR1 gene via CArG elements. Proc. Natl. Acad. Sci. 89, 10149–10153 (1992)
R.G. Meyer, J.H. Küpper, R. Kandolf et al., Early growth response-1 gene (Egr-1) promoter induction by ionizing radiation in U87 malignant glioma cells in vitro. Eur. J. Biochem. 269, 337–346 (2002). doi:10.1046/j.0014-2956.2001.02658.x
T. Joki, M. Nakamura, T. Ohno, Activation of the radiosensitive EGR-1 promoter induces expression of the herpes simplex virus thymidine kinase gene and sensitivity of human glioma cells to ganciclovir. Human Gene Ther. 6, 1507–1513. doi:10.1089/hum.1995.6.12-1507
H.B. Wang, X. Song, H. Zhang, Potentiation of tumor radiotherapy by a radiation-inducible oncolytic and oncoapoptotic adenovirus in cervical cancer xenografts. Int. J. Cancer 130, 443–453 (2012). doi:10.1002/ijc.26013
C.A. Heid, J. Stevens, K.J. Livak et al., Real time quantitative PCR. Genome Res. 6, 986–994 (1996). doi:10.1101/gr.6.10.986
Y.C. Wu, G. Song, R.F. Cao et al., Development of accurate/advanced radiotherapy treatment planning and quality assurance system (ARTS). Chin. Phys. C 32, 177–182 (2008)
S. Tao, Y. Wu, Y. Chen et al., Repeated positioning in accurate radiotherapy based on virtual net technique and contrary reconstruction scheme. Comput. Med. Imaging Graph. 30, 273–278 (2006). doi:10.1016/j.compmedimag.2006.05.006
S. Tao, A. Wu, Y. Wu et al., Patient set-up in radiotherapy with video-based positioning system. Clin. Oncol. 18, 363–366 (2006). doi:10.1016/j.clon.2005.11.018
R.F. Cao, Y.C. Wu, X. Pei et al., Multi-objective optimization of inverse planning for accurate radiotherapy. Chin. Phys. C 35, 313–317 (2011). doi:10.1088/1674-1137/35/3/019
Y.C. Wu, Development of reliability and probabilistic safety assessment program RiskA. Ann. Nucl. Energy 83, 316–321 (2015). doi:10.1016/j.anucene.2015.03.020
J. Song, G. Sun, Z. Chen et al., Benchmarking of CAD-based SuperMC with ITER Benchmark Model. Fusion Eng. Des. 89, 2499–2503 (2014). doi:10.1016/j.fusengdes.2014.05.003
L. Hu, Y. Wu, Probabilistic safety assessment of the dual-cooled waste transmutation blanket for the FDS-I. Fusion Eng. Des. 81, 1403–1407 (2006). doi:10.1016/j.fusengdes.2005.08.091
H. Zheng, G. Sun, G. Li, et al., Photon dose calculation method based on Monte Carlo finite-size pencil beam model in accurate radiotherapy. Commun. Comput. Phys. 14, 1415–1422 (2013)
Y. Wu, G. Li, S. Tao et al., Research and development of an accurate/advanced radiation therapy system (ARTS). Chin. J. Med. Phys. 22, 683–690 (2006). doi:10.3969/j.issn.1005-202X.2005.06.001
W.Y. Li, R.F. Cao, X. Pei et al., Radiotherapy reliability analysis based on PSA method. Nucl. Sci. Tech. 25, 060501 (2014). doi:10.13538/j.1001-8042/nst.25.060501
K. Zhao, M.Y. Cheng, P.C. Long et al., A hybrid voxel sampling method for constructing Rad-HUMAN phantom. Nucl. Sci. Tech. 25, 020503 (2014). doi:10.13538/j.1001-8042/nst.25.020503
J. Wang, X. Pei, R.F. Cao et al., A multiphase direct aperture optimization for inverse planning in radiotherapy. Nucl. Sci. Tech. 26, 010502 (2015). doi:10.13538/j.1001-8042/nst.26.010502
R.R. Weichselbaum, D.E. Hallahan, M.A. Beckett et al., Gene Therapy targeted by radiation preferentially radiosensitized tumor cells. Cancer Res. 54, 4266–4269 (1994)
V.K. Gupta, J.O. Park, N.T. Jackowiak et al., Combined gene therapy and ionizing radiation is a novel approach to treat human esophageal adenocarcinoma. Ann. Surg. Oncol. 9, 500–504 (2002). doi:10.1007/BF02557275
R.R. Weichselbaum, D.E. Hallahan, V.P. Sukhatme et al., Gene therapy targeted by ionizing radiation. Int. J. Radiat. Oncol. 24, 565–567 (1992). doi:10.1016/0360-3016(92)91075-X
E. Wissink, I. Verbrugge, S. Vink et al., TRAIL enhances efficacy of radiotherapy in a p53 mutant, Bcl-2 overexpressing lymphoid malignancy. Radiother. Oncol. 80, 214–222 (2006). doi:10.1016/j.radonc.2006.07.030
D. Lawrence, Z. Shahrokh, S. Marsters et al., Differential hepatocyte toxicity of recombinant Apo2L/TRAIL versions. Nat. Med. 7, 383–385 (2001). doi:10.1038/86397
C. Belka, B. Schmid, P. Marini et al., Sensitization of resistant lymphoma cells to irradiation-induced apoptosis by the death ligand TRAIL. Oncogene 20, 2190–2196 (2001)
Acknowledgments
We thank other members of the FDS team for their assistance in this work. We thank Professors ZHOU Guang-Ming and HU Bu-Rong who kindly provide us the human fibrosarcoma cell line HT1080 and normal human lung embryonic cell line MRC-5.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported by National Magnetic Confinement Fusion Science Program of China (No. 2014GB112006), National Natural Science Foundation of China (No. 11305204) and Natural Science Foundation of Anhui Province of China (No. 1508085SME220).
Rights and permissions
About this article
Cite this article
Liu, H., **, CF., Ge, SF. et al. Antitumor and radiosensitization effect of 12C6+ heavy-ion irradiation mediated by radiation-inducible gene therapy. NUCL SCI TECH 27, 18 (2016). https://doi.org/10.1007/s41365-016-0021-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41365-016-0021-x