Abstract
Background
Empagliflozin is a selective sodium–glucose co-transporter (SGLT2) inhibitor that is approved for the treatment of type 2 diabetes. The beneficial effects of empagliflozin on other organ systems including the heart and kidneys have been proven. The aim of this study is to evaluate the role of empagliflozin on acute lung injury induced by intestinal ischemia–reperfusion (I/R).
Materials and methods
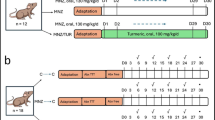
A total of 27 male Wistar albino rats were divided into three groups: sham, I/R, and I/R + empagliflozin; each group containing nine animals. Sham group rats underwent laparotomy without I/R injury. Rats in the I/R group underwent laparotomy, 1 h of after ischemia–reperfusion injury (superior mesenteric artery ligation was followed by 2 h of reperfusion). Rats in I/R were given empagliflozin (30 mg/kg) by gastric gavage for 7 days before the ischemia–reperfusion injury. All animals were killed at the end of reperfusion and lung tissue samples were obtained for immunohistochemical staining and histopathological investigation in all groups.
Results
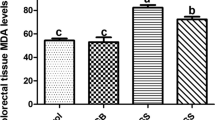
Serum glucose, AST, ALT, creatinine, native thiol, total thiol, and disulfide levels and disulfide–native thiol, disulfide–total thiol, and native thiol–total thiol ratios as well as the IMA levels were analyzed and compared among the groups. While intestinal I/R significantly increases serum aspartate aminotransferase (AST), alanine aminotransferase (ALT), and creatinine levels; did not cause any change in homeostasis parameters and IMA level. Empagliflozin treatment had no significant effect on biochemical parameters. Empagliflozin treatment induced a significant decrease in positive immunostaining for IL-1, IL-6, TNF-alpha, caspase 3, caspase 8, and caspase 9 compared to the I/R group in lung tissue samples. Intestinal I/R caused severe histopathological injury including edema, hemorrhage, increased thickness of the alveolar wall, and infiltration of inflammatory cells into alveolar spaces. Empagliflozin treatment significantly attenuated the severity of intestinal I/R injury.
Conclusions
It was concluded that empagliflozin treatment may have beneficial effects in acute lung injury, and, therefore, has the potential for clinical use.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Deitch EA (1992) Multiple organ failure pathophysiology and potential future therapy. Ann Surg 216(2):117–134. https://doi.org/10.1097/00000658-199208000-00002. (PMID: 1503516; PMCID: PMC1242583)
Guzel A, Kanter M, Guzel A (2013) Protective effect of curcumin on acute lung injury induced by intestinal ischaemia/reperfusion. Toxicol Ind Health 29(7):633–642. https://doi.org/10.1177/0748233711430984. (Epub 2012 Jan 17. Erratum in: Toxicol Ind Health. 2013 Nov;29(10):969. PMID: 22252860)
Laubach VE, Sharma AK (2016) Mechanisms of lung ischemia–reperfusion injury. Curr Opin Organ Transpl 21(3):246–252. https://doi.org/10.1097/MOT.0000000000000304. (PMID:26945320;PMCID:PMC4861054)
Gulpamuk B, Tekin K, Sonmez K (2018) The significance of thiol/disulfide homeostasis and ischemia-modified albumin levels to assess the oxidative stress in patients with different stages of diabetes mellitus. Scand J Clin Lab Invest 78(1–2):136–142. https://doi.org/10.1080/00365513.2017.1422540. (Epub 2018 Jan 3. PMID: 29298534)
Ferrari RS, Andrade CF (2015) Oxidative stress and lung ıschemia–reperfusion ınjury. Oxid Med Cell Longev 2015:590987. https://doi.org/10.1155/2015/590987. (Epub 2015 Jun 16. PMID: 26161240; PMCID: PMC4487720)
Quadri SM, Segall L, de Perrot M (2005) Caspase inhibition improves ischemia–reperfusion injury after lung transplantation. Am J Transpl 5(2):292–299. https://doi.org/10.1111/j.1600-6143.2004.00701.x. (PMID: 15643988)
Packer M, Anker SD, Butler J, EMPEROR-Reduced Trial Investigators (2020) Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med 383(15):1413–1424. https://doi.org/10.1056/NEJMoa2022190. (Epub 2020 Aug 28. PMID: 32865377)
Kabel AM, Estfanous RS, Alrobaian MM (2020) Targeting oxidative stress, proinflammatory cytokines, apoptosis and toll like receptor 4 by empagliflozin to ameliorate bleomycin induced lung fibrosis. Respir Physiol Neurobiol 273:103316. https://doi.org/10.1016/j.resp.2019.103316. (Epub 2019 Oct 7 PMID: 31600583)
Li C, Zhang J, Xue M (2019) SGLT2 inhibition with empagliflozin attenuates myocardial oxidative stress and fibrosis in diabetic mice heart. Cardiovasc Diabetol 18(1):15. https://doi.org/10.1186/s12933-019-0816-2.PMID:30710997;PMCID:PMC6359811
Abbas NAT, El Salem A, Awad MM (2018) Empagliflozin, SGLT2 inhibitor, attenuates renal fibrosis in rats exposed to unilateral ureteric obstruction: potential role of klotho expression. Naunyn Schmiedebergs Arch Pharmacol 391(12):1347–1360. https://doi.org/10.1007/s00210-018-1544-y. (Epub 2018 Aug 8 PMID: 30090949)
Nasiri-Ansari N, Nikolopoulou C, Papoutsi K (2021) Empagliflozin attenuates non-alcoholic fatty liver disease (NAFLD) in high fat diet fed ApoE(-/-) Mice by activating autophagy and reducing ER stress and apoptosis. Int J Mol Sci 22(2):818. https://doi.org/10.3390/ijms22020818. (PMID:33467546;PMCID:PMC7829901)
Takil A, Umuroğlu T, Göğüş YF, Eti Z (2003) Histopathologic effects of lipid content of enteral solutions after pulmonary aspiration in rats. Nutrition 19(7–8):666–669. https://doi.org/10.1016/s0899-9007(03)00057-1. (PMID: 12831956)
Erel O, Neselioglu S (2014) A novel and automated assay for thiol/disulphide homeostasis. Clin Biochem 47(18):326–332. https://doi.org/10.1016/j.clinbiochem.2014.09.026. (Epub 2014 Oct 7 PMID: 25304913)
Bar-Or D, Lau E, Winkler JV (2000) A novel assay for cobalt-albumin binding and its potential as a marker for myocardial ischemia-a preliminary report. J Emerg Med 19(4):311–315. https://doi.org/10.1016/s0736-4679(00)00255-9. (PMID: 11074321)
de Perrot M, Liu M, Waddell TK, Keshavjee S (2003) Ischemia–reperfusion-induced lung injury. Am J Respir Crit Care Med 167(4):490–511. https://doi.org/10.1164/rccm.200207-670SO. (PMID: 12588712)
Forgiarini LA Jr, Grün G, Kretzmann NA (2013) When is injury potentially reversible in a lung ischemia–reperfusion model? J Surg Res 179(1):168–174. https://doi.org/10.1016/j.jss.2012.08.026. (Epub 2012 Sep 10 PMID: 22989553)
Lin F, Song C, Zeng Y (2020) Canagliflozin alleviates LPS-induced acute lung injury by modulating alveolar macrophage polarization. Int Immunopharmacol 88:106969. https://doi.org/10.1016/j.intimp.2020.106969. (Epub 2020 Sep 11 PMID: 33182027)
Kingir ZB, Özdemir Kural ZN, Cam ME (2019) Effects of dapagliflozin in experimental sepsis model in rats. Ulus Travma Acil Cerrahi Derg 25(3):213–221. https://doi.org/10.5505/tjtes.2018.82826. (PMID: 31135951)
Hess DA, Terenzi DC, Trac JZ, Quan A, Mason T, Al-Omran M, Bhatt DL, Dhingra N, Rotstein OD, Leiter LA, Zinman B, Sabongui S, Yan AT, Teoh H, Mazer CD, Connelly KA, Verma S (2019) SGLT2 inhibition with empagliflozin increases circulating provascular progenitor cells in people with type 2 diabetes mellitus. Cell Metab 30(4):609–613. https://doi.org/10.1016/j.cmet.2019.08.015. (Epub 2019 Aug 30 PMID: 31477497)
Fernandez-Fernandez B, D’Marco L, Górriz JL (2020) Exploring sodium glucose co-transporter-2 (SGLT2) inhibitors for organ protection in COVID-19. J Clin Med 9(7):2030. https://doi.org/10.3390/jcm9072030. (PMID:32605278;PMCID:PMC7409231)
Abd Elmaaboud MA, Kabel AM, Elrashidy M (2019) Pre-treatment with empagliflozin ameliorates cisplatin induced acute kidney injury by suppressing apoptosis. J Appl Biomed 17(1):90. https://doi.org/10.32725/jab.2019.003. (Epub 2019 Feb 6. PMID: 34907751)
Kinaneh S, Knany Y, Khoury EE, Ismael-Badarneh R, Hamoud S, Berger G, Abassi Z, Azzam ZS (2021) Identification, localization and expression of NHE isoforms in the alveolar epithelial cells. PLoS One 16(4):e0239240. https://doi.org/10.1371/journal.pone.0239240. (PMID:33882062;PMCID:PMC8059851)
Huetsch J, Shimoda LA (2015) Na(+)/H(+) exchange and hypoxic pulmonary hypertension. Pulm Circ 5(2):228–243. https://doi.org/10.1086/680213.PMID:26064449;PMCID:PMC4449235
Adams D, Choi CS, Sayner SL (2022) Pulmonary endothelial cells from different vascular segments exhibit unique recovery from acidification and Na+/H+ exchanger isoform expression. PLoS One 17(5):e0266890. https://doi.org/10.1371/journal.pone.0266890. (PMID:35503765;PMCID:PMC9064095)
Huang D, Ju F, Du L, Liu T, Zuo Y, Abbott GW, Hu Z (2022) Empagliflozin protects against pulmonary ischemia/reperfusion injury via an extracellular signal-regulated kinases 1 and 2-dependent Mechanism. J Pharmacol Exp Ther 380(3):230–241. https://doi.org/10.1124/jpet.121.000956. (Epub 2021 Dec 10 PMID: 34893552)
Funding
None.
Author information
Authors and Affiliations
Contributions
PG: substantial contributions to the conception, acquisition, analysis, or interpretation of data for the work drafting of the work, or revising it critically for important intellectual content, final approval of the version to be published, agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. SMK: substantial contributions to the conception, drafting the work, final approval of the version to be published, agreement to be accountable for all aspects of the work. GK: design of the work, revising for important intellectual content, final approval of the version to be published, agreement to be accountable for all aspects of the work. CEO: analysis of data for the work,, drafting the work, final approval of the version to be published, agreement to be accountable for all aspects of the work. NY: revising for important intellectual content, final approval of the version to be published, agreement to be accountable for all aspects of the work. OE: substantial contributions to the conception, final approval of the version to be published, agreement to be accountable for all aspects of the work. ASN: substantial contributions to the conception, final approval of the version to be published, agreement to be accountable for all aspects of the work. CC: interpretation of data for the work,, revising for important intellectual content, final approval of the version to be published, agreement to be accountable for all aspects of the work. All authors read and approved the manuscript and all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare there are no conflicts of interest.
Ethical approval
The study was conducted according to the ethical standards specified in the 1964 Declaration of Helsinki. Research and publication ethical rules were followed in our study. Our study was approved by the ethics committee of Ankara Training and Research Hospital Animal Experiments Local Ethics Committee, dated 26.07.2021, with a 0066 meeting and 668 decision number.
Informed constent
In our animal study titled ‘Evaluation of the effects of empagliflozin on acute lung injury in rat intestinal ischemia– reperfusion model’, no informed consent is required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gokbulut, P., Kuskonmaz, S.M., Koc, G. et al. Evaluation of the effects of empagliflozin on acute lung injury in rat intestinal ischemia–reperfusion model. J Endocrinol Invest 46, 1017–1026 (2023). https://doi.org/10.1007/s40618-022-01978-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-022-01978-1