Abstract
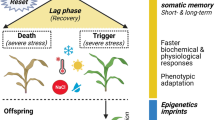
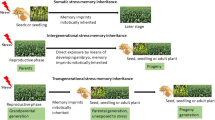
Plants leverage past stress experiences, whether occurring individually or in sequential combinations, to bolster their responses to future stressful situations, a phenomenon known as stress memory. Hydration cycles imitate a natural process within the environment when seeds are integrated into the soil seed bank, and experience numerous hydration and dehydration cycles due to inconsistent water supply. This study investigates the potential for stress exposure during the seed stage to enhance stress tolerance in subsequent seedlings, shedding light on the potential transfer of stress-related memories between various life stages following multiple stress-inducing experiences. We specifically employed hydration (soaking) and dehydration (simulating drought stress) cycles (0, 1, and 2 HD cycles) as stimuli to activate stress memory in seeds. Following germination, a randomized experiment was conducted, subjecting the seedlings to various water treatments, including control conditions, 50% and 75% reductions in soil humidity, and rewatering, with a focus on assessing recovery. Remarkably, all germinative parameters exhibited similar responses irrespective of the number of HD cycles. However, notable variations in plant growth and biomass accumulation were observed in seedlings subjected to HD cycles. These plants displayed reduced sensitivity to drought-induced damage, maintaining growth rates even under stressful conditions. Importantly, the relative water content remained unaltered in plants subjected to stress resulting from HD cycles. Consistently, the results highlight that intermittent hydration supports plant survival under stress by mitigating drought-induced damage. Stress memory acquired through stress exposure during the seed stage stress enhances drought resistance by delaying dehydration and reducing water loss, ultimately preserving growth.






Similar content being viewed by others
References
Abid, M., Shao, Y., Liu, S., Wang, F., Gao, J., Jiang, D., Tian, Z., & Dai, T. (2017). Pre-drought priming sustains grain development under post-anthesis drought stress by regulating the growth hormones in winter wheat (Triticum aestivum L.). Plant, 246, 509–524. https://doi.org/10.1007/s00425-017-2698-4
Bates, L. S., Waldren, R. P., & Teare, I. D. (1973). Rapid determination of free proline for water-stress studies. Plant and Soil, 39, 205–207. https://doi.org/10.1007/BF00018060
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72(1–2), 248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Brzezinka, K., Altmann, S., & Bäurle, I. (2019). BRUSHY1/TONSOKU/MGOUN3 is required for heat stress memory. Plant, Cell and Environment, 42(3), 771–781. https://doi.org/10.1111/pce.13365
DuBois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A., & Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 28(3), 350–356. https://doi.org/10.1021/ac60111a017
Dubrovsky, J. G. (1996). Seed hydration memory in sonoran desert cacti and its ecological implication. American Journal of Botany, 83(5), 624–632. https://doi.org/10.2307/2445922
Endres, L. (2007). Daily and seasonal variation of water relationship in sugar apple (Annona squamosal L.) under different irrigation regimes at semi-arid Brazil. Scientia Horticulturae, 113(2), 149–154. https://doi.org/10.1016/j.scienta.2007.03.007
Freitas, R. S., Meiado, M. V., & Silva, E. C. (2021). Seed discontinuous hydration does not benefit germination, but improves drought tolerance of Triplaris gardneriana seedlings. Acta Scientiarum Biological Science, 43(1), e55992. https://doi.org/10.4025/actascibiolsci.v43i1.55992
Hatzig, S. V., Nuppenau, J. N., Snowdon, R. J., & Schießl, S. V. (2018). Drought stress has transgenerational effects on seeds and seedlings in winter oilseed rape (Brassica napus L.). BMC Plant Biology, 18, 297. https://doi.org/10.1186/s12870-018-1531-y
Hayat, S., Hayat, Q., Alyemeni, M. N., Wani, A. S., Pichtel, J., & Ahmad, A. (2012). Role of proline under changing environments. Plant Signaling and Behavior, 7(11), 1456–1466. https://doi.org/10.4161/PSB.21949
Hilker, M., Schwachtje, J., Baier, M., Balazadeh, S., Bäurle, I., Geiselhardt, S., Hincha, D. K., Kunze, R., Mueller-Roeber, B., Rilling, M. C., Rolff, J., Romeis, T., Schmülling, T., Steppuhn, A., van Dongen, J., Whitcomb, S. J., Wurst, S., Zuther, E., & Kopka, J. (2015). Priming and memory of stress responses in organisms lacking a nervous system. Biological Reviews, 91(4), 1118–1133. https://doi.org/10.1111/brv.12215
Jacques, C., Salon, C., Barnard, R. L., Vernoud, V., & Prudent, M. (2021). Drought stress memory at the plant cycle level: A review. Plants, 10(9), 1873. https://doi.org/10.3390/plants10091873
Kambona, C. M., Koua, P. A., Léon, J., & Ballvora, A. (2023). Stress memory and its regulation in plants experiencing recurrent drought conditions. Theoretical and Applied Genetics, 136(2), 26. https://doi.org/10.1007/s00122-023-04313-1
Li, X., & Liu, F. (2016). Drought stress memory and drought stress tolerance in plants: Biochemical and molecular basis. In M. A. Hossain, S. H. Wani, S. Bhattacharjee, D. J. Burritt, & L. S. P. Tran (Eds.), drought stress tolerance in plants (pp. 17–44). Cham: Springer. https://doi.org/10.1007/978-3-319-28899-4_2
Lima, A. T., Cunha, P. H. J., Dantas, B. F., & Meiado, M. V. (2018). Does discontinuous hydration of Senna spectabilis (DC.) H.S. Irwin & Barneby var. excelsa (Schrad.) H.S. Irwin & Barneby (Fabaceae) seeds confer tolerance to water stress during seed germination? Journal of Seed Science, 40(1), 36–43. https://doi.org/10.1590/2317-1545v40n1182838
Lima, A. T., & Meiado, M. V. (2017). Discontinuous hydration alters seed germination under stress of two populations of cactus that occur in different ecosystems in Northeast Brazil. Seed Science Research, 27, 292–302. https://doi.org/10.1017/S0960258517000241
Liu, X., Quan, W., & Bartels, D. (2022). Stress memory responses and seed priming correlate with drought tolerance in plants: An overview. Planta, 255(2), 45. https://doi.org/10.1007/s00425-022-03828-z
Marques, F. R. F., Meiado, M. V., Castro, N. M. C. R., Campos, M. L. O., Mendes, K. R., Santos, O. O., & Pompelli, M. F. (2015). GerminaQuant: A new tool for germination measurements. Journal of Seed Science, 37(3), 248–255. https://doi.org/10.1590/2317-1545v37n3145605
Mauch-Mani, B., Baccelli, I., Luna, E., & Flors, V. (2017). Defense priming: An adaptive part of induced resistance. Annual Review of Plant Biology, 68(1), 485–512. https://doi.org/10.1146/annurev-arplant-042916-041132
Mladenov, V., Fotopoulos, V., Kaiserli, E., Karalija, E., Maury, S., Baranek, M., Segal, N., Testillano, P. S., Vassileva, V., Pinto, G., Nagel, M., Hoenicka, H., Miladinović, D., Gallusci, P., Vergata, C., Kapazoglou, A., Abraham, E., Tani, E., Gerakari, M., … Martinelli, F. (2021). Deciphering the epigenetic alphabet involved in transgenerational stress memory in crops. International Journal of Molecular Sciences, 22(13), 7118. https://doi.org/10.3390/ijms22137118
Nicolau, J. P. B., Silva, F. E., Felix, F. C., Torres, S. B., Pacheco, N. V., & Pereira, M. D. (2020). Discontinuous hydration on the germination of Mimosa caesalpiniifolia and Pityrocarpa moniliformis seeds under water stress. Revista Caatinga, 33(2), 555–561. https://doi.org/10.1590/1983-21252020v33n228rc
Ozturk, M., Unal, B. T., Gárcia-Caparrós, P., Khursheed, A., Gul, A., & Hasanuzzaman, M. (2021). Osmoregulation and its actions during the drought stress in plants. Physiologia Plantarum, 172(2), 1321–1335. https://doi.org/10.1111/PPL.13297
Pastor, V., Luna, E., Ton, J., & Flors, V. (2012). Primed plants do not forget. Environmental and Experimental Botany, 94, 46–56. https://doi.org/10.1016/j.envexpbot.2012.02.013
Ranal, M. A., & Santana, D. G. (2006). How and why to measure the germination process? Brazilian Journal of Botany, 29(1), 1–11. https://doi.org/10.1590/S0100-84042006000100002
Sadhukhan, A., Prasad, S. S., Mitra, J., Siddiqui, N., Sahoo, L., Kobayashi, Y., & Koyama, H. (2022). How do plants remember drought? Planta. https://doi.org/10.1007/s00425-022-03924-0
Sani, E., Herzyk, P., Perrella, G., Colot, V., & Amtmann, A. (2013). Hyperosmotic priming of Arabidopsis seedlings establishes a long-term somatic memory accompanied by specific changes of the epigenome. Genome Biology, 14, R59. https://doi.org/10.1186/gb-2013-14-6-r59
Santos, B. S., Lima, A. T., & Meiado, M. V. (2023). Interpopulational seed hydration memory does not affect seed germination of **quexique gounellei (F.A.C. Weber ex K. Schum) Lavor & Calvente subsp. gounellei (Cactaceae) under water deficit. Bradleya, 41, 105–116.
Schwachtje, J., Whitcomb, S. J., Firmino, A. A. P., Zuther, E., Hincha, D. K., & Kopka, J. (2019). Induced, imprinted, and primed responses to changing environments: Does metabolism store and process information? Frontiers in Plant Science, 10, 106. https://doi.org/10.3389/fpls.2019.00106
Seleiman, M. F., Al-Suhaibani, N., Ali, N., Akmal, M., Alotaibi, M., Refay, Y., Dindaroglu, T., Abdul-Wajid, H. H., & Battaglia, M. L. (2021). Drought stress impacts on plants and different approaches to alleviate its adverse effects. Plants, 10(2), 259. https://doi.org/10.3390/plants10020259
Sharma, M., Kumar, P., Verma, V., Sharma, R., Bhargava, B., & Irfan, M. (2022). Understanding plant stress memory response for abiotic stress resilience: Molecular insights and prospects. Plant Physiology and Biochemistry, 179, 10–24. https://doi.org/10.1016/j.plaphy.2022.03.004
Silva, E. C., Albuquerque, M. B., Azevedo Neto, A. D., & Silva Junior, C. D. (2013). Drought and its consequences to plants—From individual to ecosystem. In S. Akinic (Ed.), Responses of organisms to water stress (pp. 17–47). Rijeka: Intech Open. https://doi.org/10.5772/53833
Tardieu, F., Simonneau, T., & Muller, B. (2018). The physiological basis of drought tolerance in crop plants: A scenario-dependent probabilistic approach. Annual Review of Plant Biology, 29(69), 733–759. https://doi.org/10.1146/annurev-arplant-042817-040218
Varela, M. C., Reinoso, H., Luna, V., & Cenzano, A. M. (2018). Seasonal changes in morphophysiological traits of two native Patagonian shrubs from Argentina with different drought resistance strategies. Plant Physiology and Biochemistry, 127, 506–515. https://doi.org/10.1016/j.plaphy.2018.03.018
Walter, J., Jentsch, A., Beierkuhnlein, C., & Kreyling, J. (2013). Ecological stress memory and cross stress tolerance in plants in the face of climate extremes. Environmental and Experimental Botany, 94, 3–8. https://doi.org/10.1016/j.envexpbot.2012.02.009
Weatherley, P. (1950). Studies in the water relations of the cotton plant. I. The field measurement of water deficit in leaves. New Phytologist, 49(1), 81–97. https://doi.org/10.1111/j.1469-8137.1950.tb05146.x
Yang, M., He, J., Sun, Z., Li, Q., Cai, J., Zhou, Q., Wollenweber, B., Jiang, D., & Wang, X. (2023). Drought priming mechanisms in wheat elucidated by in-situ determination of dynamic stomatal behavior. Frontiers in Plant Science., 14, 1138494. https://doi.org/10.3389/fpls.2023.1138494
Acknowledgements
The authors thank COPES/UFS (Research Coordination of the Federal University of Sergipe) for the scholarship granted during the Scientific Initiation Program at the undergraduate level.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Freitas, R.S., da Silva, E.C. Unveiling the influence of stress memory: enhancing stress tolerance in seedlings through seed stage stress exposure. Plant Physiol. Rep. 29, 165–175 (2024). https://doi.org/10.1007/s40502-023-00751-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40502-023-00751-7