Abstract
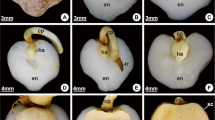
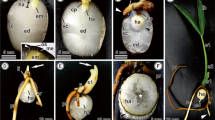
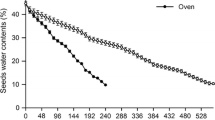
Seeds of several palms are reserve-rich, and have a complex and poorly understood mobilization. The massive endosperm and embryo of Acrocomia aculeata Lodd. ex Mart. possess large amounts of proteins, polysaccharides, and lipids. We evaluated cell features related to reserve mobilization during germination and initial development of seedlings of A. aculeata. Samples of the haustorium (the cotyledonary blade) at different developmental stages and of the entire digestion zone of the endosperm were processed by standard methods for ultrastructural evaluation. The haustorium reserve mobilization begins when germination starts, and proteins are the first to be metabolized, followed by polysaccharides and lipids. Haustorial cells present lipid bodies associated with glyoxysomes and protein vacuoles, which are involved in lipid mobilization. The digestion zone of the endosperm comprises cell layers adjacent to the haustorium, in which reserve mobilization begins after the protrusion of the cotyledonary petiole. The endosperm reserve mobilization is similar to observed in the haustorium, firstly proteins, polysaccharides, and lipids in the end. The abundant carbohydrates in the cell walls of endosperm are hydrolyzed, and the cells lose integrity, while digestion products accumulate around the haustorium. The sinuosities observed in the plasma membrane and the organelles predominating in the epidermal cells of the haustorium are consistent with absorption and transitory storage of reserves, and there is no evidence of the secretion of enzymes that can act in the mobilization of endosperm reserves. Therefore, the endosperm has a storage and self-degradation function, and the products of hydrolysis are transported towards the haustorium via the apoplastic route.







Similar content being viewed by others
References
Alang ZC, Moir GFJ, Jones LH (1988) Composition, degradation and utilization of endosperm during germination in the oil palm (Elaeis guineensis Jacq.). Ann Bot 61:261–268. https://doi.org/10.1093/oxfordjournals.aob.a087553
Baskin JM, Baskin CC (2014) What kind of seed dormancy might palms have? Seed Sci Res 24:17–22. https://doi.org/10.1017/S0960258513000342
Bewley JD, Black M (1994) Seeds: physiology of development and germination. Plenum Press, New York
Bewley JD, Bradford KJ, Hilhorst HWM, Nonogaki H (2013) Seeds: physiology of development, germination and dormancy. Springer, New York
Bicalho EM, Motoike SY, Borges EEL, Ataíde GM, Guimarães VM (2016) Enzyme activity and reserve mobilization during macaw palm (Acrocomia aculeata). Acta Bot Bras 30:437–444. https://doi.org/10.1590/0102-33062016abb0181
Buckeridge MS (2010) Seed cell wall storage polysaccharides: models to understand cell wall biosynthesis and degradation. Plant Physiol 154:1017–1023. https://doi.org/10.1104/pp.110.158642
DeMason DA (1984) Growth parameters in the cotyledon of date seedlings. Bot Gaz 145:176–183
DeMason DA (1985) Histochemical and ultrastructural changes in the haustorium of date (Phoenix dactylifera L.). Protoplasma 126:168–177
DeMason DA (1988) Seedling development in Washingtonia filifera (Arecaceae). Bot Gaz 149:45–56. https://doi.org/10.1086/337690
DeMason DA, Thomson WW (1981) Structure and ultrastructure of the cotyledon of date palm (Phoenix dactylifera L.). Bot Gaz 142:320–328. https://doi.org/10.1086/337230
DeMason DA, Sexton R, Grant Reid JS (1983) Structure, composition and physiological state of the endosperm of Phoenix dactylifera L. Ann Bot 52:71–80. https://doi.org/10.1093/oxfordjournals.aob.a086554
DeMason DA, Sexton R, Gorman M, Reid JSG (1985) Structure and biochemistry of endosperm breakdown in date palm (Phoenix dactylifera L.) seeds. Protoplasma 126:159–167
DeMason DA, Stillman JI, Ellmore GS (1989) Acid phosphatase localization in seedling tissues of the palms, Phoenix dactylifera and Washingtonia filifera, and its relevance to controls of germination. Can J Bot 67:1103–1110. https://doi.org/10.1139/b89-144
Dias DS, Ribeiro LM, Lopes PSN, Melo GA, Muller M, Munné-Bosch S (2018) Haustorium-endosperm relationships and the integration between developmental pathways during reserve mobilization in Butia capitata (Arecaceae). Ann Bot 122:267–277. https://doi.org/10.1093/aob/mcy065
Dransfield J, Uhl NW, Asmussen CBA, Baker WJ, Harley MM, Lewis CE (2008) Genera palmarum: the evolution and classification of palms. Kew Publishing, Kew
Graham IA (2008) Seed storage oil mobilization. Annu Rev Plant Biol 59:115–142. https://doi.org/10.1146/annurev.arplant.59.032607.092938
Hayashi Y, Hayashi M, Hayashi H, Hara-Nishimura I, Nishimura M (2001) Direct interaction between glyoxysomes and lipid bodies in cotyledons of the Arabidopsis thaliana ped1 mutant. Protoplasma 218:83–94. https://doi.org/10.1007/BF01288364
Henderson F (2006) Morphology and anatomy of palm seedlings. Bot Rev 72:273–329. https://doi.org/10.1663/0006-8101(2006)72%5b273:MAAOPS%5d2.0.CO;2
Herman EM, Larkins BA (1999) Protein storage bodies and vacuoles. Plant Cell 11:601–613. https://doi.org/10.1105/tpc.11.4.601
Huang AHC (1996) Oleosins and oil bodies in seeds and other organs. Plant Physiol 110:1055–1061. https://doi.org/10.1104/pp.110.4.1055
Jiang L, Phillips TE, Hamm CA, Drozdowicz YM, Rea PA, Maeshima M, Rogers SW, Rogers JC (2001) The protein storage vacuole: a unique compound organelle. J Cell Biol 155:991–1002. https://doi.org/10.1083/jcb.200107012
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. J Cell Biol 27:137–138
Lorenzi H, Noblick L, Kahn F, Ferreira E (2010) Flora Brasileira: Arecaceae (palmeiras). Instituto Plantarum, Nova Odessa
Mazzottini-dos-Santos HC, Ribeiro LM, Mercadante-Simões MO, Sant’Anna-Santos BF (2015) Ontogenesis of the pseudomonomerous fruit of Acrocomia aculeata (Arecaceae): a new approach to the development of pyrenarium fruits. Trees 29:199–214. https://doi.org/10.1007/s00468-014-1104-0
Mazzottini-dos-Santos HC, Ribeiro LM, Oliveira DMT (2017) Roles of the haustorium and endosperm during the development of seedlings of Acrocomia aculeata (Arecaceae): dynamics of reserve mobilization and accumulation. Protoplasma 254:1563–1578. https://doi.org/10.1007/s00709-016-1048-x
Mazzottini-dos-Santos HC, Ribeiro LM, Oliveira DMT (2018) Structural changes in the micropylar region and overcoming dormancy in Cerrado palms seeds. Trees 32:1415–1428. https://doi.org/10.1007/s00468-018-1723-y
Moura EF, Ventrella MC, Motoike SY (2010) Anatomy, histochemistry and ultrastructure of seed and somatic embryo of Acrocomia aculeata (Arecaceae). Sci Agr 67:375–495. https://doi.org/10.1590/S0103-90162010000400004
Neves SC, Ribeiro LM, Cunha IRG, Pimenta MAS, Mercadante-Simões MO, Lopes PSN (2013) Diaspore structure and germination ecophysiology of the babassu palm (Attalea vitrivir). Flora 208:68–78. https://doi.org/10.1016/j.flora.2012.12.007
Oliveira NCC, Lopes PSN, Ribeiro LM, Mercadante-Simões MO, Oliveira LAA, Silvério FO (2013) Seed structure, germination, and reserve mobilization in Butia capitata (Arecaceae). Trees 27:1633–1645. https://doi.org/10.1007/s00468-013-0910-0
Oo KC, Stumpf PK (1983) Some enzymic activities in the germinating oil palm (Elaeis guineensis) seedling. Plant Physiol 73:1028–1032. https://doi.org/10.1104/pp.73.4.1028
Orozco-Segovia A, Batis AI, Roja-Aréchiga M, Mendoza A (2003) Seed biology of palms: a review. Palms 47:79–94
Paiva EAS (2016) How do secretory products cross the plant cell wall to be released? A new hypothesis involving cyclic mechanical actions of the protoplast. Ann Bot 117:533–540. https://doi.org/10.1093/aob/mcw012
Panza V, Láinez V, Maldonado S (2004) Seed structure and histochemistry in the palm Euterpe edulis. Bot J Linn Soc 145:445–453. https://doi.org/10.1111/j.1095-8339.2004.00293.x
Pires TP, Souza ES, Kuki KN, Motoike SY (2013) Ecophysiological traits of the macaw palm: a contribution towards the domestication of a novel oil crop. Ind Crop Prod 44:200–210. https://doi.org/10.1016/j.indcrop.2012.09.029
Poxleitner M, Rogers SW, Samuels AL, Browse J, Rogers JC (2006) A role of caleosin in degradation of oil-body storage lipids during seed germination. Plant J 47:917–933. https://doi.org/10.1111/j.1365-313X.2006.02845.x
Ribeiro LM, Souza PP, Rodrigues-Junior AG, Oliveira TGS, Garcia QS (2011) Overcoming dormancy in macaw palm diaspores, a tropical species with potential for use as biofuel. Seed Sci Technol 39:303–317. https://doi.org/10.15258/sst.2011.39.2.04
Ribeiro LM, Oliveira DMT, Garcia QS (2012) Structural evaluations of zygotic embryos and seedlings of the macaw palm (Acrocomia aculeata, Arecaceae). Trees 26:851–863. https://doi.org/10.1007/s00468-011-0659-2
Ribeiro LM, Garcia QS, Müller M, Munné-Bosch S (2015) Tissue-specific hormonal profiling during dormancy release in macaw palm seeds. Physiol Plant 153:627–642. https://doi.org/10.1111/ppl.12269
Roland AM (1978) General preparations and staining of thin sections. In: Hall JL (ed) Electron Microscopy and cytochemistry of plant cells. Elsevier, New York, pp 1–62
Sekhar KNC, DeMason DA (1988) Quantitative ultrastructure and protein composition of date palm (Phoenix dactylifera) seeds: a comparative study of endosperm vs. embryo. Am J Bot 75:323–329. https://doi.org/10.1002/j.1537-2197.1988.tb13446.x
Sekhar KNC, DeMason DA (1990) Identification and immunocytochemical localization de α-galactosidase in resting and germinated date palm (Phoenix dactylifera L.) seeds. Planta 181:53–61. https://doi.org/10.1007/BF00202324
Sugimuma Y, Murakami T (1990) Structure and function of the haustorium in germinating coconut palm seed. Jpn Agr Res Q 24:1–14
Tan-Wilson AL, Wilson K (2012) Mobilization of seed protein reserves. Physiol Plantarum 145:140–153. https://doi.org/10.1111/j.1399-3054.2011.01535.x
Verdeil JL, Hocher V (2002) Digestion and absorption of food in plants: a plant stomach. Trends Plant Sci 7:280–281. https://doi.org/10.1016/S1360-1385(02)02269-0
Zienkiewicz A, Zienkiewicz K, Rejón JD, Alché JD, Castro AJ, Rodríguez-García MI (2014) Olive seed protein bodies store degrading enzymes involved in mobilization of oil bodies. J Exp Bot 65:103–115. https://doi.org/10.1093/jxb/ert355
Acknowledgements
The authors thank Centro de Microscopia of the Universidade Federal de Minas Gerais, Brazil (http://www.microscopia.ufmg.br), for processing samples, use of the equipment, and obtaining images; and Maria Olívia Mercadante Simões for providing equipment and support in several stages of this work.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brazil (CAPES, Finance Code 001), Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG, Brazil, process CRA-APQ-01335-13, and doctoral scholarship of H.C.M.S.), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil, processes 305686/2018-6, 305638/2018-1 and 302216/2018-9, respectively, for research productivity grants of D.M.T.O., E.A.S.P. and L.M.R.).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by HCMS. All authors performed data analysis and participated in writing the manuscript. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mazzottini-dos-Santos, H.C., Ribeiro, L.M., Oliveira, D.M.T. et al. Ultrastructural aspects of metabolic pathways and translocation routes during mobilization of seed reserves in Acrocomia aculeata (Arecaceae). Braz. J. Bot 43, 589–600 (2020). https://doi.org/10.1007/s40415-020-00622-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40415-020-00622-7