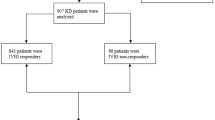Abstract
Background and Objective
Intravenous immunoglobulin (IVIG) is a prominent therapeutic agent for Kawasaki disease (KD) that significantly reduces the incidence of coronary artery anomalies. Various methodologies, including machine learning, have been employed to develop IVIG non-responder prediction models; however, their validation and reproducibility remain unverified. This study aimed to develop a predictive scoring system for identifying IVIG nonresponders and rigorously test the accuracy and reliability of this system.
Methods
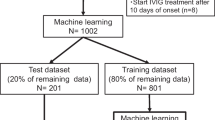
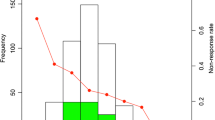
The study included an exposure group of 228 IVIG non-responders and a control group of 997 IVIG responders. Subsequently, a predictive machine learning model was constructed. The Shizuoka score, including variables such as the “initial treatment date” (cutoff: < 4 days), sodium level (cutoff: < 133 mEq/L), total bilirubin level (cutoff: ≥ 0.5 mg/dL), and neutrophil-to-lymphocyte ratio (cutoff: ≥ 2.6), was established. Patients meeting two or more of these criteria were grouped as high-risk IVIG non-responders. Using the Shizuoka score to stratify IVIG responders, propensity score matching was used to analyze 85 patients each for IVIG and IVIG-added prednisolone treatment in the high-risk group. In the IVIG plus prednisolone group, the IVIG non-responder count significantly decreased (p < 0.001), with an odds ratio of 0.192 (95% confidence interval 0.078–0.441).
Conclusions
Intravenous immunoglobulin non-responders were predicted using machine learning models and validated using propensity score matching. The initiation of initial IVIG-added prednisolone treatment in the high-risk group identified by the Shizuoka score, crafted using machine learning models, appears useful for predicting IVIG non-responders.



Similar content being viewed by others
References
Dahdah N. A tale of a trail on how it takes 5 days of Kawasaki disease to initiate coronary artery injury and change the lives of children. Turk Arch Pediatr. 2024;59:131–4. https://doi.org/10.5152/TurkArchPediatr.2024.23254.
Furusho K, Kamiya T, Nakano H, Kiyosawa N, Shinomiya K, Hayashidera T, et al. High-dose intravenous gammaglobulin for Kawasaki disease. Lancet. 1984;2:1055–8. https://doi.org/10.1016/s0140-6736(84)91504-6.
Newburger JW, Takahashi M, Burns JC, Beiser AS, Chung KJ, Duffy CE, et al. The treatment of Kawasaki syndrome with intravenous gamma globulin. N Engl J Med. 1986;315:341–7. https://doi.org/10.1056/NEJM198608073150601.
Durongpisitkul K, Gururaj VJ, Park JM, Martin CF. The prevention of coronary artery aneurysm in Kawasaki disease: a meta-analysis on the efficacy of aspirin and immunoglobulin treatment. Pediatrics. 1995;96:1057–61.
Terai M, Shulman ST. Prevalence of coronary artery abnormalities in Kawasaki disease is highly dependent on gamma globulin dose but independent of salicylate dose. J Pediatr. 1997;131:888–93. https://doi.org/10.1016/s0022-3476(97)70038-6.
Burns JC, Capparelli EV, Brown JA, Newburger JW, Glode MP. Intravenous gamma-globulin treatment and retreatment in Kawasaki disease. US/Canadian Kawasaki Syndrome Study Group. Pediatr Infect Dis J. 1998;17:1144–8. https://doi.org/10.1097/00006454-199812000-00009.
Han RK, Silverman ED, Newman A, McCrindle BW. Management and outcome of persistent or recurrent fever after initial intravenous gamma globulin therapy in acute Kawasaki disease. Arch Pediatr Adolesc Med. 2000;154:694–9. https://doi.org/10.1001/archpedi.154.7.694.
Kobayashi T, Inoue Y, Takeuchi K, Okada Y, Tamura K, Tomomasa T, et al. Prediction of intravenous immunoglobulin unresponsiveness in patients with Kawasaki disease. Circulation. 2006;113:2606–12. https://doi.org/10.1161/CIRCULATIONAHA.105.592865.
Sano T, Kurotobi S, Matsuzaki K, Yamamoto T, Maki I, Miki K, et al. Prediction of non-responsiveness to standard high-dose gamma-globulin therapy in patients with acute Kawasaki disease before starting initial treatment. Eur J Pediatr. 2007;166:131–7. https://doi.org/10.1007/s00431-006-0223-z.
Egami K, Muta H, Ishii M, Suda K, Sugahara Y, Iemura M, et al. Prediction of resistance to intravenous immunoglobulin treatment in patients with Kawasaki disease. J Pediatr. 2006;149:237–40. https://doi.org/10.1016/j.jpeds.2006.03.050.
Kobayashi T, Saji T, Otani T, Takeuchi K, Nakamura T, Arakawa H, et al. Efficacy of immunoglobulin plus prednisolone for prevention of coronary artery abnormalities in severe Kawasaki disease (RAISE study): a randomised, open-label, blinded-endpoints trial. Lancet. 2012;379:1613–20. https://doi.org/10.1016/S0140-6736(11)61930-2.
Miyata K, Kaneko T, Morikawa Y, Sakakibara H, Matsushima T, Misawa M, et al. Efficacy and safety of intravenous immunoglobulin plus prednisolone therapy in patients with Kawasaki disease (Post RAISE): a multicentre, prospective cohort study. Lancet Child Adolesc Health. 2018;2:855–62. https://doi.org/10.1016/S2352-4642(18)30293-1.
Ogata S, Ogihara Y, Honda T, Kon S, Akiyama K, Ishii M. Corticosteroid pulse combination therapy for refractory Kawasaki disease: a randomized trial. Pediatrics. 2012;129:e17-23. https://doi.org/10.1542/peds.2011-0148.
Hamada H, Suzuki H, Onouchi Y, Ebata R, Terai M, Fuse S, et al. Efficacy of primary treatment with immunoglobulin plus ciclosporin for prevention of coronary artery abnormalities in patients with Kawasaki disease predicted to be at increased risk of non-response to intravenous immunoglobulin (KAICA): a randomised controlled, open-label, blinded-endpoints, phase 3 trial. Lancet. 2019;393:1128–37. https://doi.org/10.1016/S0140-6736(18)32003-8.
Mori M, Matsubara T. Overview of guidelines for the medical treatment of acute Kawasaki disease in Japan (2020 revised version) and positioning of plasma exchange therapy in the acute phase. Pediatr Infect Dis J. 2023;42:e328–32. https://doi.org/10.1097/INF.0000000000003974.
Bi Q, Goodman KE, Kaminsky J, Lessler J. What is machine learning? A primer for the epidemiologist. Am J Epidemiol. 2019;188: 222239. https://doi.org/10.1093/aje/kwz189.
Sidey-Gibbons JAM, Sidey-Gibbons CJ. Machine learning in medicine: a practical introduction. BMC Med Res Methodol. 2019;19:64. https://doi.org/10.1186/s12874-019-0681-4.
Obermeyer Z, Emanuel EJ. Predicting the future: big data, machine learning, and clinical medicine. N Engl J Med. 2016;375:1216–9. https://doi.org/10.1056/NEJMp1606181.
Goecks J, Jalili V, Heiser LM, Gray JW. How machine learning will transform biomedicine. Cell. 2020;181:92–101. https://doi.org/10.1016/j.cell.2020.03.022.
Wang T, Liu G, Lin H. A machine learning approach to predict intravenous immunoglobulin resistance in Kawasaki disease patients: a study based on a Southeast China population. PLoS ONE. 2021;16: e0253675. https://doi.org/10.1371/journal.pone.0253675.
Kuniyoshi Y, Tokutake H, Takahashi N, Kamura A, Yasuda S, Tashiro M. Comparison of machine learning models for prediction of initial intravenous immunoglobulin resistance in children with Kawasaki disease. Front Pediatr. 2020;8: 570834. https://doi.org/10.3389/fped.2020.570834.
Liu J, Zhang J, Huang H, Wang Y, Zhang Z, Ma Y, et al. A machine learning model to predict intravenous immunoglobulin-resistant Kawasaki disease patients: retrospective study based on the Chongqing population. Front Pediatr. 2021;9: 756095. https://doi.org/10.3389/fped.2021.756095.
Takeuchi M, Inuzuka R, Hayashi T, Shindo T, Hirata Y, Shimizu N, et al. Novel risk assessment tool for immunoglobulin resistance in Kawasaki disease: application using a random forest classifier. Pediatr Infect Dis J. 2017;36:821–6. https://doi.org/10.1097/INF.0000000000001621.
Lam JY, Song MS, Kim GB, Shimizu C, Bainto E, Tremoulet AH, et al. Intravenous immunoglobulin resistance in Kawasaki disease patients: prediction using clinical data. Pediatr Res. 2023;16:1–6. https://doi.org/10.1038/s41390-023-02519-z.
Sakai H, Iwashima S, Sano S, Akiyama N, Nagata E, Harazaki M, et al. Targeted use of prednisolone with intravenous immunoglobulin for Kawasaki disease. Clin Drug Investig. 2021;41:77–88. https://doi.org/10.1007/s40261-020-00984-6.
Kimura M, Harazaki M, Fukuoka T, Asakura I, Sakai H, Kamimaki T, et al. Targeted use of prednisolone with the second IVIG dose for refractory Kawasaki disease. Pediatr Int. 2017;59:397–403. https://doi.org/10.1111/ped.13190.
Ayusawa M, Sonobe T, Uemura S, Ogawa S, Nakamura Y, Kiyosawa N, et al. Revision of diagnostic guidelines for Kawasaki disease (the 5th revised edition). Pediatr Int. 2005;47:232–4. https://doi.org/10.1111/j.1442-200x.2005.02033.x.
Research Committee on Kawasaki Disease. Report of subcommittee on standardization of diagnostic criteria and reporting of coronary artery lesions in Kawasaki disease. Tokyo: Ministry of Health and Welfare; 1984. https://www.scirp.org/(S(351jmbntvnsjt1aadkposzje))/reference/ReferencesPapers.aspx?ReferenceID=410827 Accessed 10 Oct 2023.
Kobayashi T, Fuse S, Sakamoto N, Mikami M, Ogawa S, Hamaoka K, et al. A new Z score curve of the coronary arterial internal diameter using the lambda-mu-sigma method in a pediatric population. J Am Soc Echocardiogr. 2016;29:794-801.e29. https://doi.org/10.1016/j.echo.2016.03.017.
Fuse S, Kobayashi T, Arakaki Y, Ogawa S, Katoh H, Sakamoto N, et al. Standard method for ultrasound imaging of coronary artery in children. Pediatr Int. 2010;52:876–82. https://doi.org/10.1111/j.1442-200X.2010.03252.x.
Tremoulet AH, Best BM, Song S, Wang S, Corinaldesi E, Eichenfield JR, et al. Resistance to intravenous immunoglobulin in children with Kawasaki disease. J Pediatr. 2008;153:117–21. https://doi.org/10.1016/j.jpeds.2007.12.021.
Noble WS. What is a support vector machine? Nat Biotechnol. 2006;24:15657. https://doi.org/10.1038/nbt1206-1565.
Association for Computing Machinery Special Interest Group on Management of Data & ACM Special Interest Group on Knowledge Discovery in Data. Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining. New York: ACM; 2016.
Rahman T, Khandakar A, Abir FF, Faisal MAA, Hossain MS, Podder KK, et al. QCovSML: a reliable COVID-19 detection system using CBC biomarkers by a stacking machine learning model. Comput Biol Med. 2022;143: 105284. https://doi.org/10.1016/j.compbiomed.2022.105284.
Rodríguez-Pérez R, Bajorath J. Interpretation of machine learning models using shapley values: application to compound potency and multi-target activity predictions. J Comput Aided Mol Des. 2020;34:1013–26. https://doi.org/10.1007/s10822-020-00314-0.
Lundberg SM, Lee SI. A unified approach to interpreting model predictions. In: von Luxburg U, Guyon I, Bengio S, Wallach H, Fergus R (eds) Proceedings of the 31st international conference on neural information processing systems. Red Hook: Curran Associates; 2017; pp. 4768–77.
Lundberg SM, Erion G, Chen H, DeGrave A, Prutkin JM, Nair B, et al. From local explanations to global understanding with explainable AI for trees. Nat Mach Intell. 2020;2:56–67. https://doi.org/10.1038/s42256-019-0138-9.
Kanda Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant. 2013;48:452–8. https://doi.org/10.1038/bmt.2012.244.
Kanai T, Takeshita S, Kawamura Y, Kinoshita K, Nakatani K, Iwashima S, et al. The combination of the neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios as a novel predictor of intravenous immunoglobulin resistance in patients with Kawasaki disease: a multicenter study. Heart Vessels. 2020;35:1463–72. https://doi.org/10.1007/s00380-020-01622-z.
Kuniyoshi Y, Tsujimoto Y, Banno M, Taito S, Ariie T, Takahashi N, et al. Prediction models for intravenous immunoglobulin resistance in Kawasaki disease: a meta-analysis. Pediatrics. 2023;151: e2022059175. https://doi.org/10.1542/peds.2022-059175.
Debray TPA, Damen JAAG, Snell KIE, Ensor J, Hooft L, Reitsma JB, et al. A guide to systematic review and meta-analysis of prediction model performance. BMJ. 2017;356: i6460. https://doi.org/10.1136/bmj.i6460.
Andaur Navarro CL, Damen JAA, Takada T, Nijman SWJ, Dhiman P, Ma J, et al. Risk of bias in studies on prediction models developed using supervised machine learning techniques: systematic review. BMJ. 2021;375: n2281. https://doi.org/10.1136/bmj.n2281.
Tsoukas P, Yeung RSM. Kawasaki disease and MIS-C share a host immune response. Nat Rev Rheumatol. 2022;18:555–6. https://doi.org/10.1038/s41584-022-00820-5.
Ghosh P, Katkar GD, Shimizu C, Kim J, Khandelwal S, Tremoulet AH, et al. An Artificial Intelligence-guided signature reveals the shared host immune response in MIS-C and Kawasaki disease. Nat Commun. 2022;13:2687. https://doi.org/10.1038/s41467-022-30357-w.
Yoshikane Y, Okuma Y, Miyamoto T, Hashimoto J, Fukazawa R, Kato T, et al. Serum tenascin-C predicts resistance to steroid combination therapy in high-risk Kawasaki disease: a multicenter prospective cohort study. Pediatr Rheumatol Online J. 2021;19:82. https://doi.org/10.1186/s12969-021-00562-w.
Kitoh T, Ohara T, Muto T, Okumura A, Baba R, Koizumi Y, et al. Increased pentraxin 3 levels correlate with IVIG responsiveness and coronary artery aneurysm formation in Kawasaki disease. Front Immunol. 2021;12: 624802. https://doi.org/10.3389/fimmu.2021.624802.
Chen Z, Sai S, Nagumo K, Wu Y, Chiba H, Hui SP. Distinctive serum lipidomic profile of IVIG-resistant Kawasaki disease children before and after treatment. PLoS ONE. 2023;18: e0283710. https://doi.org/10.1371/journal.pone.0283710.
Kobayashi T, Ayusawa M, Suzuki H, Abe J, Ito S, Kato T, et al. Revision of diagnostic guidelines for Kawasaki disease (6th revised edition). Pediatr Int. 2020;62:1135–8. https://doi.org/10.1111/ped.14326.
Acknowledgements
We thank the current and former members of the Shizuoka Kawasaki Disease Study Group for their cooperation in conducting this study. We also thank Hidemasa Sakai, Department of Pediatrics, Shizuoka City Shizuoka Hospital; Shinichiro Sano, Department of Diabetes and Metabolism, Shizuoka Children’s Hospital; Naoe Akiyama, Department of Pediatrics, Fuji City General Hospital; Masashi Harazaki, Department of Pediatrics, Medical Genetics, Shizuoka General Hospital; Tetuya Fukuoka, Department of Pediatrics, Shizuoka Saiseikai General Hospital; and Isao Miyairi, Department of Pediatrics, Hamamatsu University School of Medicine. ChatGPT was used for English proofreading.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of Interest
Yoshifumi Miyagi and Satoru Iwashima declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical Approval
The study protocol conformed to the principles of the Declaration of Helsinki and was approved by the Ethics Committee of Chutoen Medical Center (approval date: 5 August, 2019; approval number: 100).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Availability of Data and Material
Not applicable.
Code Availability
Not applicable.
Author Contributions
All authors made substantial contributions to the conception of the study. All authors read and approved the final manuscript. YM and SI interpreted the data and wrote the paper.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Miyagi, Y., Iwashima, S. Prediction Models for Intravenous Immunoglobulin Non-Responders of Kawasaki Disease Using Machine Learning. Clin Drug Investig 44, 425–437 (2024). https://doi.org/10.1007/s40261-024-01373-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-024-01373-z