Abstract
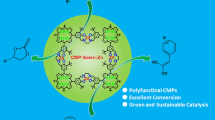
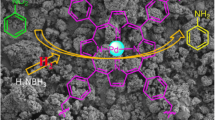
Salen-porphyrin-based conjugated microporous polymers(CMPs) have been demonstrated to be an attractive material platform for predesigned structures and promising applications. Herein, a new salen-porphyrin-based conjugated microporous polymer(SP-CMP-L) was solvothermally prepared by porphyrin-forming condensation reaction of pyrrole and salen-dialdehyde derivative. The SP-CMP-L was characterized by spectroscopy technologies, and also exhibited excellent thermal and chemical stability. The porosity of SP-CMP-L was examined by N2 adsorption/desorption isotherms. The BET specific surface area of the CMP material was calculated to be 290.4 m2/g with the pore volume of 0.19 cm3/g. The microstructure property of the resulting material was further evaculated by scanning electron microscopy(SEM) and transmission electron microscopy(TEM). The SP-CMP-L with salen and porphyrin multidentate coordination sites was proposed to serve as an initiator to promote the cross-coupling between aryl halides with unactivated arenes under base-mediated conditions. The transition-metal-free catalytic protocol provided high catalytic activity for direct C-H arylation reaction of unactivated arenes, and thus offered a convenient and efficient alternative for the construction biaryl scaffolds. In addition, the salen-porphyrin-based CMP material possessed remarkable adsorption capability for the removal of organic amines from water.
Similar content being viewed by others
References
Xu Y., ** S., Xu H., Nagai A., Jiang D., Chem. Soc. Rev., 2013, 42, 8012
Sprick R. S., Jiang J.-X., Bonillo B., Ren S., Ratvijitvech T., Guiglion P., Zwijnenburg M. A., Adams D. J., Cooper A. I., J. Am. Chem. Soc., 2015, 137, 3265
Zhou Y.-B., Wang Y.-Q., Ning L.-C., Ding Z.-C. Wang W.-L., Ding C.-K., Li R.-H., Chen J.-J., Lu X., Ding Y.-J., Zhan Z.-P., J. Am. Chem. Soc., 2017, 139, 3966
Xu M., Han X., Wang T., Li S., Hua D., J. Mater. Chem. A, 2018, 6, 13894
Ayed C., Silva L. C., Wang D., Zhang K., J. Mater. Chem. A, 2018, 6, 22145
Jiang J.-X., Su F. B., Trewin A., Wood C. D., Campbell N. L., Niu H. J., Dickinson C., Ganin A. Y., Rosseinsky M. J., Khimyak Y. Z., Cooper A. I., Angew. Chem. Int. Ed., 2007, 46, 8574
Li A., Lu R. F., Wang Y., Wang X., Han K. L., Deng W. Q., Angew. Chem. Int. Ed., 2010, 49, 3330
Wang Z., Yang X., Yang T., Zhao Y., Wang F., Chen Y., Zeng J. H., Yan C., Huang F., Jiang J.-X., ACS Catal., 2018, 8, 8590
Liu X., Xu Y., Jiang D., J. Am. Chem. Soc., 2012, 134, 8738
Sun L., Zou Y., Liang Z., Yu J., Xu R., Polym. Chem., 2014, 5, 471
Chen L., Honsho Y., Seki S., Jiang D., J. Am. Chem. Soc., 2010, 132, 6742
Xu F., Chen X., Tang Z., Wu D., Fu R., Jiang D., Chem. Commun., 2014, 50, 4788
Wei W., Chang G., Xu Y., Yang L., J. Mater. Chem. A, 2018, 6, 18794
Chaoui N., Trunk M., Dawson R., Schemidta J., Thomas A., Chem. Soc. Rev., 2017, 46, 3302
Zhang Y., Riduan S. N., Chem. Soc. Rev., 2012, 41, 2083
Liu X. A. S., Zhang Y., Luo X., **a H., Li H., Mu Y., RSC Adv., 2014, 4, 6447
Maity N., Barman S., Minenkov Y., Ould-Chikh S., Abou-Hamad E., Ma T., Qureshi Z. S., Cavallo L., D’Elia V., Gates B. C., ACS Catal., 2018, 8, 2715
Iwai T., Konishi S., Miyazaki T., Kawamorita S., Yokokawa N., Ohmiya H., Sawamura M., ACS Catal., 2015, 5, 7254
Chai G.-L., Boero M., Hou Z., Terakura K., Cheng W., ACS Catal., 2017, 7, 7908
Dams M., Drijkoningen L., Pauwels B., van Tendeloo G., de Vos D. E., Jacobs P. A., J. Catal., 2002, 209, 225
Zhi Y., Li Z., Feng X., **a H., Zhang Y., Shi Z., Mu Y., Liu X., J. Mater. Chem. A, 2017, 5, 22933
Wei P.-F., Qi M.-Z., Wang Z.-P., Ding S.-Y., Yu W., Liu Q., Wang L.-K., Wang H.-Z., An W.-K., Wang W., J. Am. Chem. Soc., 2018, 140, 4623
Zhang J., Han X., Wu X., Liu Y., Cui Y., J. Am. Chem. Soc., 2017, 139, 8277
Dhakshinamoorthy A., Li Z., Garcia H., Chem. Soc. Rev., 2018, 47, 8134
Zhang Y., Ying J. Y., ACS Catal., 2015, 5, 2681
Jiang J.-X., Wang C., Laybourn A., Hasell T., Clowes R., Khimyak Y. Z., **ao J., Higgins S. J., Adams D. J., Cooper A. I., Angew. Chem. Int. Ed., 2011, 50, 1072
Ishida T., Onuma Y., Kinjo K., Hamasaki A., Ohashi H., Honma T., Akita T., Yokoyama T., Tokunaga M., Haruta M., Tetrahedron, 2014, 70, 6150
Sadjadi S., Heravi M. M., RSC Adv., 2016, 6, 88588
Liu W., Su Q., Ju P., Guo B., Zhou H., Li G., Wu Q., ChemSusChem, 2017, 10, 664
Zhou H., Wu C., Wu Q., Guo B., Liu W., Li G., Su Q., Mu Y., Polym. Chem., 2017, 8, 1488
Guo B., Liu Q., Su Q., Liu W., Ju P., Li G., Wu Q., J. Mater. Sci., 2018, 53, 15746
Liu W., Wu S., Su Q., Guo B., Ju P., Li G., Wu Q., J. Mater. Sci., 2019, 54, 1205
Ju P., Wu S., Su Q., Li X., Liu Z., Li G., Wu Q., J. Mater. Chem. A, 2019, 7, 2660
**e Y., Wang T.-T., Liu X.-H., Zou K., Deng W.-Q., Nat. Commun., 2013, 4, 1960
Chen A., Zhang Y., Chen J., Chen L., Yu Y., J. Mater. Chem. A, 2015, 3, 9807
Shirakawa E., Itoh K.-I., Higashino T., Hayashi T., J. Am. Chem. Soc., 2010, 132, 15537
Liu W., Cao H., Zhang H., Zhang H., Chung K. H., He C., Wang H., Kwong F. Y., Lei A., J. Am. Chem. Soc., 2010, 132, 16737
**ao Y., Han T., **ao G., Ying Y., Huang H., Yang Q., Liu D., Zhong C., Langmuir, 2014, 30, 12229
Moda A., Pramanik M., Inagaki S., Bhaumik A., J. Mater. Chem. A, 2014, 2, 11642
Sharma A., Malani A., Medhekar N. V., Babarao R., CrystEngComm, 2017, 19, 6950
Li Z.-J., Ding S.-Y., Xue H.-D., Cao W., Wang W., Chem. Commun., 2016, 52, 7217
**ong S., Fu X., **ang L., Yu G., Guan J., Wang Z., Du Y., **ong X., Pan C., Polym. Chem., 2014, 5, 3424
Yang P., Wang R., Zhou M., Wang X., Angew. Chem. Int. Ed., 2018, 57, 8674
Sharma S., Kumar M., Kumar V., Kumar N., Tetrahedron Lett., 2013, 54, 4868
Zhao H., Shen J., Ren C., Zeng W., Zeng H., Org. Lett., 2017, 19, 2190
Zhang R., Miao C.-X., Wang S., **a C., Sun W., ChemCatChem, 2012, 4, 192
Ng Y. S., Chan C. S., Chan K. S., Tetrahedron Lett., 2012, 53, 3911
Chen Y., Wang B., Wang X., **e L. H., Li J., **e Y., Li J. R., ACS Appl. Mater. Interfaces, 2017, 9, 27027
Yang K., Wu W., **g Q., Zhu L., Environ. Sci. Technol., 2008, 42, 7931
Hlekelele L., Nomadolo N. E., Setshedi K. Z., Mofokeng L. E., Chetty A., Chauke V. P., RSC Adv., 2019, 9, 14531
Shahabuddin S., Sarih N. M., Mohamad S., Baharin S. N. A., RSC Adv., 2016, 6, 43388
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the National Natural Science Foundation of China(No.51703076).
Supporting Information
40242_2020_208_MOESM1_ESM.pdf
Polyfunctional Conjugated Microporous Polymers for Applications in Direct C-H Arylation of Unactivated Arenes and Aqueous Adsorption of Aromatic Amines
Rights and permissions
About this article
Cite this article
Luo, K., Su, Q., Ju, P. et al. Polyfunctional Conjugated Microporous Polymers for Applications in Direct C-H Arylation of Unactivated Arenes and Aqueous Adsorption of Aromatic Amines. Chem. Res. Chin. Univ. 36, 1302–1309 (2020). https://doi.org/10.1007/s40242-020-0208-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-020-0208-x