Abstract
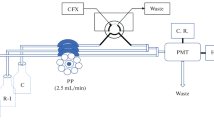
A novel chemiluminescence(CL) method is proposed for cefixime trihydride(CFX) determination based on its enhancement effect on diperiodatoargentate(III)(DPA)-rhodamine 6-G(Rh6-G) reaction in conjunction with flow injection analysis(FIA). A linear calibration curve was achieved over the range from 0.01 mg/L to 2.5 mg/L CFX(R2=0.999, n=8) with relative standard deviation(RSD) of 1.4%—3.8%(n=4)), limit of detection(LOD) of 3.0×10–3 mg/L(S/N=3), injection throughput of 180/h and regression equation of y=1113.2x–14.596[y=CL intensity (mV) and x=concentration of CFX(mg/L)]. The method was successfully applied to CFX determination in pharmaceutical formulations and the recoveries(%) for proposed FI-CL and a reported spectrophotometric method by applying the Student t-test [calculated t-test value: t=1.079215, and tabulated t-distributed(95%)=2.200985] were not significantly different. The CFX was efficiently extracted and no significant effect of commonly found excipients in the pharmaceutical formulations was observed. The mechanism of CL reaction is discussed briefly.
Similar content being viewed by others
References
Kalyani J. V., Rastogi R. B., Kumar D., RSC Adv., 2014, 4, 30500
Dubala A., Nagarajan J. S. K., Vimal C. S., George R., J. Chromatogr. Sci., 2014, 53, 694
Wani Y. B., Patil D. D., J. Saudi. Chem. Soc., 2017, 21, S101
Karimian N., Gholivand M. B., Malekzadeh G., J. Electroanal. Chem., 2016, 771, 64
Troy D. B., Beringer P., Remingtons, The Science and Practice of Pharmacy, 21st Ed., Vol. II, 2005, 1644
Katzung B., Basic and Clinical Pharmacology(10th Ed.), McGraw-Hill Medical, Charlottesville, 2006, 726
Nagaraju K., Chowdary Y. A., Int. J. Sci. Res. Sci. Tech., 2018, 4, 669
Chowdary A. H., Kiranjyothi R., Priyanka J., Int. J. Pharm. Biol. Sci. Arch., 2018, 6, 19
Hassouna M. E. M., Abdelrahman M. M., Mohamed M. A., J. Foren-sic. Sci., 2017, 2, 1
Memon N. A., Memon A. A., Memon F. N., Sherazi S. T. H., Memon S., Leghari M. K., Chromatographia, 2018, 81, 1373
Maheshwari M. L., Memon A. A., Memon S., Mughal U. U. R., Dayo A., Memon N., Ghoto M. A., Leghari M. K., Saudi Pharm. J., 2015, 23, 444
Kundu S., Majumder T., Barat P. K., Ray S. K., Int. J. Pharm. Sci. Res., 2015, 6, 884
Dehghani M., Nasirizadeh N., Yazdanshenas M. E., Mater. Sci. Eng., 2019, 96, 654
Balooei M., Raoof J. B., Chekin F., Ojani R., Anal. Bioanal. Elec-trochem., 2017, 9, 266
Yaman Y. T., Bolat G., Yardimci C., Abaci S., Turk. J. Chem., 2018, 42, 826
Norouzi B., Tajjedin S., Bulg. Chem. Commun., 2017, 49, 729
Fakhri A., Shahidi S., Agarwal S., Gupta V. K., Int. J. Electrochem. Sci., 2016, 11, 1530
Eskandari H., Amirzehni M., Asadollahzadeh H., Hassanzadeh J., Eslami P. A., Sensor. Actuat. B: Chem., 2018, 275, 145
Pan H., Wang S., Dao X., Ni Y., Inorg. Chem., 2018, 57, 1417
Akhgari F., Samadi N., Farhadi K., J. Fluoresc., 2017, 27, 921
Eskandari H., Amirzehni M., Asadollahzadeh H., Eslami P. A., New J. Chem., 2017, 41, 7186
Gandhi S. V., Sonawane P. S., Asian J. Res. Chem., 2018, 11, 705
Amraei A., Niazi A., Iran. J. Pharm. Res., 2018, 17, 1191
Masoudyfar Z., Elhami S., Spectrochim. Acta A, 2019, 211, 234
Khan M. N., Qayum A., Rehman U. U., Gulab H., Idrees M., J. Appl. Spectrosc., 2015, 82, 705
Keskar M. R., Jugade R. M., Anal. Chem. Insights, 2015, 10, ACI–S28463
Fletcher P., Andrew K. N., Calokerinos A. C., Forbes S., Worsfold, P. J., Luminescence, 2001, 16, 1
Li B., Zhang Z., Wu M., Talanta, 2000, 51, 515
Yue L., Chem. Res. Chinese Universities, 2018, 34(5), 758
Balikungeri A., Pelletier M., Inorg. Chim. Acta, 1978, 29, 141
Murthy C. P., Sethnram B., Rao T. N., Z. Phy. Chem., 1986, 267, 1212
Asghar M., Yaqoob M., Nabi A., Chem. Res. Chinese Universities, 2017, 33(2), 354
Su M., Chen P., Sun H., TrAC Trends Anal. Chem., 2018, 100, 36
Sun H., Chen P., Wang F., Wen H., Talanta, 2009, 79, 134
Sun H., Chen P., Wang F., Spectrochim. Acta Part A, 2009, 74, 819
Chen P., Sun H., Drug Test. Anal., 2010, 2, 24
Sun H., Chen P., Wang F., Anal. Lett., 2010, 43, 2234
Fu Z. F., Li G. K., Hu Y. F., Chinese J. Anal. Chem., 2015, 43(9), 1322
Sun H. W., Shi S. S., Wang J., Chen P. Y., Asian J. Chem., 2012, 24, 3865
Zhao F., Zhao W. H., **ong W., Luminescence, 2013, 28, 108
Fu Z., Li G., Hu Y., Anal. Methods, 2015, 7, 4590
Asghar M., Yaqoob M., Nabi A., Int. J. Environ. Anal. Chem., 2017, 97, 276
Ahmed M., Asghar M., Yaqoob M., Munawar N., Shahid F., Asad M., Nabi A., Anal. Sci., 2017, 33, 1259
Du J., Li, H., Appl. Spectrosc., 2010, 64(10), 1154
He S. H., He D. Y., J. Sichuan Normal University(Natural Science), 2008, 3, 350
Al Haddabi B., Al Lawati H. A., Suliman F. O., Talanta, 2016, 150, 399
Cui H., Zhang Q., Myint A., Ge X., Liu L., J. Photochem. Photobiol. A: Chem., 2006, 181, 238
Liu H., Hao Y., Ren J., He P., Fang Y., Luminescence, 2007, 22, 302
He D., He S., Zhang Z., Anal. Lett., 2007, 40, 2935
Kubin R. F., Fletcher A. N., J. Luminescence, 1982, 27, 455
Inoue Y., Hamashima H., J. Biomater. Nanobiotechnol., 2012, 3, 136
Acknowledgments
The authors acknowledge Nexus Pharma(Pvt.) Limited, Karachi, Pakistan for providing pure cefixime trihydrate as a gift.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Muhammad, Y., Mohammed, Y., Muhammad, A. et al. Chemiluminescence Determination of Cefixime Trihydride Based on Acidic Diperiodatoargentate(III)-Rhodamine 6-G System. Chem. Res. Chin. Univ. 35, 775–781 (2019). https://doi.org/10.1007/s40242-019-9073-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-019-9073-x