Abstract
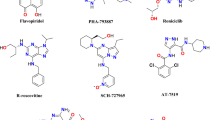
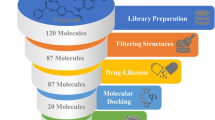
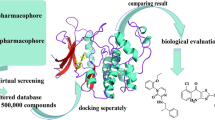
In women who die from cancer, breast cancer is the most common cause of death. The development of small molecular scaffolds as specific Cyclin-dependent kinase (CDK) inhibitors is a promising strategy in the discovery of anti-breast cancer drugs. Isoindolin-1-ones are heterocyclic compounds with useful therapeutic properties. In this study, a library of 48 isoindolinones has been virtually screened by molecular docking that showed high binding affinity up to − 10.1 kcal/mol and conventional hydrogen bonding interactions with active amino acid residues of CDK7. The molecular dynamics simulation (MDS), fragment molecular orbital (FMO), density functional theory (DFT), and pharmacokinetics studies of the best two docked scored ligands 7 and 14 have been studied. Examining the ligand root mean square deviation and hydrogen bonding occupancy of the 100 ns MDS trajectory, both ligands 7 and 14 showed docked pose stability. FMO calculations displayed that LYS139 and LYS41 are majorly contributing to the binding interactions with ligands 7 and 14 in the docked poses. DFT studies of ligands 7 and 14 showed high values of global softness and low values of global hardness and chemical potential thus displaying chemically reactive soft molecules and this influences their anti-cancer activity. Our hits exhibited superior qualities to known CDK7 inhibitors, according to the comprehensive pharmacokinetic parameters that were predicted. The results indicate that isoindolin-1-one moieties are good candidates for anti-cancer action and could serve as effective CDK7 inhibitors.











Similar content being viewed by others
Data availability
A supplementary data file of complete library of isoindolin-1-ones with CDK7 protein is provided.
References
Abraham M, Alekseenko A, Bergh C, Blau C, Briand E, Doijade M, Fleischmann S, Gapsys V, Garg G, Gorelov S, Gouaillardet G, Gray A, Irrgang ME, Jalalypour F, Jordan J, Junghans C, Kanduri P, Keller S, Kutzner C, Lemkul JA, Lundborg M, Merz P, Miletic V, Morozov D, Pall S, Schulz R, Shirts M, Shvetsov A, Soproni B, van der Spoel D, Turner P, Uphoff C, Villa A, Wingbermuhle S, Zhmurov A, Bauer P, Hess B, Lindahl E (2023) GROMACS 2023 manual (Version 2023). Zenodo. https://doi.org/10.5281/zenodo7588711
Anwer KE, Sayed GH, Kozakiewicz-Piekarz A, Ramadan RM (2023) Novel annulated thiophene derivatives: Synthesis, spectroscopic, X-ray, Hirshfeld surface analysis, DFT, biological, cytotoxic and molecular docking studies. J Mol Struct 1276:134798. https://doi.org/10.1016/j.molstruc.2022.134798
Bansode P, Jadhav J, Kurane R, Choudhari P, Bhatia M, Khanapure S, Salunkhe R, Rashinkar G (2016) Potentially antibreast cancer enamidines: via azide-alkyne-amine coupling and their molecular docking studies. RSC Adv 6:90597–90606. https://doi.org/10.1039/c6ra20583f
Bansode P, Anantacharya R, Dhanavade M, Kamble S, Barale S, Sonawane K, Satyanarayan ND, Rashinkar G (2019) Evaluation of drug candidature: In silico ADMET, binding interactions with CDK7 and normal cell line studies of potentially anti-breast cancer enamidines. Comput Biol Chem 83:107124. https://doi.org/10.1016/j.compbiolchem.2019.107124
Beegum S, Panicker CY, Armaković S, Armaković SJ, Arisoy M, Temiz-Arpaci O, Alsenoy CV (2019) Spectroscopic, antimicrobial and computational study of novel benzoxazole derivative. J Mol Struct 1176:881–894. https://doi.org/10.1016/jmolstruc201809019
BIOVIA Dassault Systèmes [Discovery Studio 2021 client] [Discovery Studio Visualizer v211020298] San Diego: Dassault Systèmes 2021
Brahmchari D, Verma AK, Mehta S (2018) Regio- and stereoselective synthesis of isoindolin-1-ones through BuLi-Mediated Iodoaminocyclization of 2-(1-Alkynyl)benzamides. J Org Chem 83:3339–3347. https://doi.org/10.1021/acs.joc.7b02903
Chashoo G, Saxena AK (2014) Targetting CDKs in cancer: an overview and new insights. J Cancer Sci Ther 6:488–496. https://doi.org/10.4172/1948-5956.1000313
Chatelut E, Delord JP, Canal P (2003) Toxicity patterns of cytotoxic drugs. Invest New Drugs 21:141–148. https://doi.org/10.1023/a:1023565227808
Chohan TA, Qian HY, Pan YL, Chen JZ (2016) Molecular simulation studies on the binding selectivity of 2-anilino-4-(thiazol-5-yl)-pyrimidines in complexes with CDK2 and CDK7. Mol Biosyst 12:145–161. https://doi.org/10.1039/c5mb00630a
Cui W, Aouidate A, Wang S, Yu Q, Li Y, Yuan S (2020) Discovering anti-cancer drugs via computational methods. Front Pharmacol 11:733. https://doi.org/10.3389/fphar.2020.00733
Curtin ML, Frey RR, Heyman HR, Sarris KA, Steinman DH, Holmes JH, Bousquet PF, Cunha GA, Moskey MD, Ahmed AA, Pease LJ, Glaser KB, Stewart KD, Davidsen SK, Michaelides MR (2004) Isoindolinone ureas: a novel class of KDR kinase inhibitors. Bioorg Med Chem Lett 14:4505–4509. https://doi.org/10.1016/j.bmcl.2004.06.041
Dallakyan S, Olson AJ (2015) Small-molecule library screening by docking with PyRx Methods. Mol Biol 1263:243–250. https://doi.org/10.1007/978-1-4939-2269-7_19
Dennington R, Keith TA, Millam JM (2016) Gauss view 6.0.16. Semichem Inc Shawnee Mission, KS, USA
Deshpande A, Sicinski P, Hinds PW (2005) Cyclins and cdks in development and cancer: a perspective. Oncogene 24:2909–2915. https://doi.org/10.1038/sj.onc.1208618
Diab S, Yu M, Wang S (2020) CDK7 inhibitors in cancer therapy: the sweet smell of success? J Med Chem 63:7458–7474. https://doi.org/10.1021/acs.jmedchem.9b01985
El Saghir NS, Khalil MK, Eid T, El Kinge AR, Charafeddine M, Geara F, Seoud M, Shamseddine AI (2007) Trends in epidemiology and management of breast cancer in develo** arab countries: a literature and registry analysis. Int J Surg 5:225–233. https://doi.org/10.1016/j.ijsu.2006.06.015
Er-rajy M, El Fadili M, Hadni H, Mrabti NN, Zarougui S, Elhallaoui M (2022) 2D-QSAR modeling, drug-likeness studies, ADMET prediction, and molecular docking for anti-lung cancer activity of 3-substituted-5-(phenylamino) indolone derivatives. Struct Chem 33:973–986. https://doi.org/10.1007/s11224-022-01913-3
Er-rajy M, Mujwar S, Imtara H, Alshawwa SZ, Nasr FA, Zarougui S, Elhallaoui M (2023) Design of novel anti-cancer agents targeting COX-2 inhibitors based on computational studies. Arab J Chem 16:105193. https://doi.org/10.1016/j.arabjc.2023.105193
Er-Rajy M, El Fadili M, Mujwar S, Zarougui S, Elhallaoui M (2023) Design of novel anti-cancer drugs targeting TRKs inhibitors based 3D QSAR, molecular docking and molecular dynamics simulation. J Biomol Struct Dyn 41:11657–11670. https://doi.org/10.1080/07391102.2023.2170471
Er-Rajy M, Faris A, Zarougui S, Elhallaoui M (2024) Design of potential anti-cancer agents as COX-2 inhibitors, using 3D-QSAR modeling, molecular docking, oral bioavailability proprieties, and molecular dynamics simulation. Anticancer Drugs 35:117–128. https://doi.org/10.1097/CAD.0000000000001492
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL (2009) Gaussian. Inc, CT Wallingford
Galbraith MD, Bender H, Espinosa JM (2019) Therapeutic targeting of transcriptional cyclin dependent kinases. Transcription 10:118–136. https://doi.org/10.1080/21541264.2018.1539615
Gupta VP (2016) Electro Density analysis and electrostatic potential. Princ Appl Quantum Chem. https://doi.org/10.1016/B978-0-12-803478-100006-6
Halgren TA (1999) MMFF VI MMFF94s option for energy minimization studies. J Comput Chem 20:720–729. https://doi.org/10.1002/(SICI)1096-987X(199905)20:7%3c720::AID-JCC7%3e30CO,2-X
Hazel P, Kroll SH, Bondke A, Barbazanges M, Patel H, Fuchter MJ, Coombes RC, Ali S, Barrett AG, Freemont PS (2017) Inhibitor selectivity for cyclin-dependent kinase 7: a structural, thermodynamic, and modelling study. ChemMedChem 12:372–380. https://doi.org/10.1002/cmdc.201600535
Ismail MF, Aboelnaga A, Naguib MM, Ismaeel HM (2023) Synthesis, anti-proliferative activity, DFT and docking studies of some novel chloroquinoline-based heterocycles. Polycycl Aromat Compd. https://doi.org/10.1080/10406638.2023.2271112
Kaliszczak M, Patel H, Kroll SH, Carroll L, Smith G, Delaney S, Heathcote DA, Bondke A, Fuchter MJ, Coombes RC, Barrett AG, Ali S, Aboagye EO (2013) Development of a cyclin-dependent kinase inhibitor devoid of ABC transporter-dependent drug resistance. Br J Cancer 109:2356–2367. https://doi.org/10.1038/bjc.2013.584
Khanapure S, Jagadale M, Bansode P, Choudhari P, Rashinkar G (2018) Anticancer activity of ruthenocenyl chalcones and their molecular docking studies. J Mol Struct 1173:142–147. https://doi.org/10.1016/jmolstruc201806091
Kim J, Cho YJ, Ryu JY, Hwang I, Han HD, Ahn HJ, Kim WY, Cho H, Chung JY, Hewitt SM, Kim JH, Kim BG, Bae DS, Choi CH, Lee JW (2020) CDK7 is a reliable prognostic factor and novel therapeutic target in epithelial ovarian cancer. J Oncol 156:211–221. https://doi.org/10.1016/j.ygyno.2019.11.004
Kitaura K, Ikeo E, Asada T, Nakano T, Uebayasi M (1999) Fragment molecular orbital method: an approximate computational method for large molecules. Chem Phys Lett 313:701–706. https://doi.org/10.1016/S0009-2614(99)00874-X
Kumar V, Parate S, Thakur G, Lee G, Ro HS, Kim Y, Kim HJ, Kim MO, Lee KW (2021) Identification of CDK7 inhibitors from natural sources using pharmacoinformatics and molecular dynamics simulations. Biomedicines 9:1197. https://doi.org/10.3390/biomedicines9091197
Lagorce D, Douguet Miteva MA, Villoutreix BO (2017) Computational analysis of calculated physicochemical and ADMET properties of protein-protein interaction inhibitors. Sci Rep 7:46277. https://doi.org/10.1038/srep46277
Lapenna S, Giordano A (2009) Cell cycle kinases as therapeutic targets for cancer. Nat Rev Drug Discov 8:547–566. https://doi.org/10.1038/nrd2907
Liu K, Zheng M, Lu R, Du J, Zhao Q, Li Z, Li Y, Zhang S (2020) The role of CDC25C in cell cycle regulation and clinical cancer therapy: a systematic review. Cancer Cell Int 20:213. https://doi.org/10.1186/s12935-020-01304-w
Lolli G, Lowe ED, Brown NR, Johnson LN (2004) The crystal structure of human CDK7 and its protein recognition properties. Struct 12:2067–2079. https://doi.org/10.1016/j.str.2004.08.013
Mary YS, Yalcin G, Mary YS, Resmi KS, Thomas R, Önkol T, Kasap EN, Yildiz I (2020) Spectroscopic, quantum mechanical studies, ligand protein interactions and photovoltaic efficiency modeling of some bioactive benzothiazolinone acetamide analogs. Chem Pap 74:1957–1964. https://doi.org/10.1007/s11696-019-01047-7
Mehta S, Mangyan M, Brahmchari D (2023) Design, synthesis and anti-cancer activity evaluation of a 3-methyleneisoindolin-1-one library. Comb Chem High Throughput Screen 26:1775–1792. https://doi.org/10.2174/1386207325666221003093623
Mellaoui MD, Zaki K, Abbiche K, Imjjad A, Boutiddar R, Sbai A, Jmiai A, El Issami S, Lamsabhi AM, Zejli H (2024) In silico anticancer activity of isoxazolidine and isoxazolines derivatives: DFT study, ADMET prediction, and molecular docking. J Mol Struct. https://doi.org/10.1016/j.molstruc.2024.138330
Meng XY, Zhang HX, Mezei M, Cui M (2011) Molecular docking: a powerful approach for structure-based drug discovery. Curr Comput-Aided Drug Des 7:146–157. https://doi.org/10.2174/157340911795677602
National Center for Biotechnology Information. PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/thz. Accessed 18 December 2023
National Center for Biotechnology Information. PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/bs-181. Accessed 18 December 2023
National Center for Biotechnology Information. PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/Milciclib. Accessed 18 December 2023
Norman RA, Toader D, Ferguson AD (2012) Structural approaches to obtain kinase selectivity. Trends Pharmacol Sci 33:273–278. https://doi.org/10.1016/j.tips.2012.03.005
O’Boyle NM, Banck M, James CA, Morley C, Vandermeersch T, Hutchison GR (2011) Open Babel: An open chemical toolbox. J Cheminform 3:33. https://doi.org/10.1186/1758-2946-3-33
Parrinello M, Rahman A (1981) Polymorphic transitions in single crystals: a new molecular dynamics method. J Appl Phys 52:7182–7190. https://doi.org/10.1063/1328693
Pearson G (1986) Absolute electronegativity and hardness correlated with molecular orbital theory. Proc Natl Acad Sci 83:8440–8441. https://doi.org/10.1073/pnas83228440
Peyressatre M, Prével C, Pellerano M, Morris MC (2015) Targeting cyclin-dependent kinases in human cancers: from small molecules to peptide inhibitors. Cancers 7:179–237. https://doi.org/10.3390/cancers7010179
Pires DEV, Blundell TL, Ascher DB (2015) pkCSM: Predicting small-molecule pharmacokinetic and toxicity properties using graph-based signatures. J Med Chem 58:4066–4072. https://doi.org/10.1021/acsjmedchem5b00104
Pronk S, Páll S, Schulz R, Larsson P, Bjelkmar P, Apostolov R, Shirts MR, Smith JC, Kasson PM, van der Spoel D, Hess B, Lindahl E (2013) GROMACS 4.5: a high-throughput and highly parallel open source molecular simulation toolkit. Bioinformatics 29:845–854. https://doi.org/10.1093/bioinformatics/btt055
Renuga S, Muthu S (2014) Molecular structure, normal coordinate analysis, harmonic vibrational frequencies, NBO, HOMO–LUMO analysis and detonation properties of (S)-2-(2-oxopyrrolidin-1-yl) butanamide by density functional methods. Spectrochim Acta A Mol Biomol Spectrosc 118:702–715. https://doi.org/10.1016/jsaa201309055
Ricci-Lopez J, Aguila SA, Gilson MK, Brizuela CA (2021) Improving structure-based virtual screening with ensemble docking and machine learning. J Chem Inf Model 61:5362–5376. https://doi.org/10.1021/acsjcim1c00511
Roe DR, Cheatham TE III (2013) PTRAJ and CPPTRAJ: software for processing and analysis of molecular dynamics trajectory data. J Chem Theory Comput 9:3084–3095. https://doi.org/10.1021/ct400341p
Roe DR, Cheatham TE (2018) Parallelization of CPPTRAJ enables large scale analysis of molecular dynamics trajectory data. J Chem Theory Comput 39:2110–2117. https://doi.org/10.1002/jcc25382
Sánchez-Martínez C, Lallena MJ, Sanfeliciano SG, de Dios A (2019) Cyclin dependent kinase (CDK) inhibitors as anticancer drugs: recent advances (2015–2019). Bioorganic Med Chem Lett 29:126637. https://doi.org/10.1016/j.bmcl.2019.126637
Sanchez-Vega F, Mina M, Armenia J, Chatila WK, Luna A, La KC, Dimitriadoy S, Liu DL, Kantheti HS, Saghafinia S, Chakravarty D, Daian F, Gao Q, Bailey M, Liang WW, Foltz SM, Shmulevich I, Ding L, Heins Z, Ochoa A, Gross B, Gao J, Zhang H, Kundra R, Kandoth C, Bahceci I, Dervishi L, Dogrusoz U, Zhou W, Shen H, Laird PW, Way GP, Greene CS, Liang H, **ao Y, Wang C, Lavarone A, Berger AH, Bivona TG, Lazar AJ, Hammer GD, Giordano T, Kwong LN (2018) Oncogenic signaling pathways in the cancer genome atlas. Cell 173:321–337. https://doi.org/10.1016/j.cell.2018.03.035
Sapay N, Tieleman DP (2011) Combination of the CHARMM27 force field with united-atom lipid force fields. J Comput Chem 32:1400–1410. https://doi.org/10.1002/jcc21726
Savela R, Méndez-Gálvez C (2021) Isoindolinone synthesis via one-pot type transition metal catalyzed C-C bond forming reactions. Chem Eur J 27:5344–5378. https://doi.org/10.1002/chem.20200437
Schmidt MW, Baldridge KK, Boatz JA, Elbert ST, Gordon MS, Jensen JH, Koseki S, Matsunaga N, Nguyen KA, Su S, Windus TL, Dupuis M, Montgomery JA Jr (1993) General atomic and molecular electronic structure system. J Comput Chem 14:1347–1363. https://doi.org/10.1002/jcc540141112
Shalaby MA, Fahim AM, Rizk SA (2023) Microwave-assisted synthesis, antioxidant activity, docking simulation, and DFT analysis of different heterocyclic compounds. Sci Rep 13:4999. https://doi.org/10.1038/s41598-023-31995-w
Shukla AK, Chaudhary AP, Pandey J (2020) Synthesis, spectral analysis, molecular docking and DFT studies of 3-(2, 6-dichlorophenyl)-acrylamide and its dimer through QTAIM approach. Heliyon. https://doi.org/10.1016/j.heliyon.2020.e05016
Singla P, Luxami V, Paul K (2013) Benzimidazole-biologically attractive scaffold for protein kinase inhibitors. RSC Adv 4:12422–12440. https://doi.org/10.1039/c3ra46304d
Sonawane K, Barale SS, Dhanavade MJ, Waghmare SR, Nadaf NH, Kamble SA, Naik NM (2020) Homology modeling and docking studies of TMPRSS2 with experimentally known inhibitors camostat mesylate, nafamostat and bromhexine hydrochloride to control SARS-coronavirus-2. ChemRxiv. https://doi.org/10.26434/chemrxiv.12162360.v1
Suenaga M (2005) Facio: new computational chemistry environment for PC GAMESS. J Comput Chem 4:25–32. https://doi.org/10.2477/jccj425
US National Library of Medicine National Centre for Biotechnology Information. PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/Trilaciclib. Accessed 7 March 2023
US National Library of Medicine National Centre for Biotechnology Information. PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/thz1. Accessed 7 March 2023
US National Library of Medicine National Centre for Biotechnology Information. PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/bs-181. Accessed 7 March 2023
US National Library of Medicine National Centre for Biotechnology Information. PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/Milciclib. Accessed 7 March 2023
Vijayakumar V, Prabakran A, Radhakrishnan N, Muthu S, Chidambaram RK, Paulraj EI (2019) Synthesis, characterization, spectroscopic studies, DFT and molecular docking analysis of N4, N4’-dibutyl-3 3’-diaminobenzidine. J Mol Struc 1179:325–335. https://doi.org/10.1016/jmolstruc201811018
Wang S, Wood G, Meades C, Griffiths G, Midgley C, McNae I, Fischer PM (2004) Synthesis and biological activity of 2-anilino-4-(1H-pyrrol-3-yl) pyrimidine CDK inhibitors. Bioorg Med Chem Lett 14:4237–4240. https://doi.org/10.1016/j.bmcl.2004.06.012
Wang M, Wang T, Zhang X, Wu X, Jiang S (2020) Cyclin-dependent kinase 7 inhibitors in cancer therapy. Future Med Chem 12:813–833. https://doi.org/10.4155/fmc-2019-0334
Zhang Z, Peng H, Wang X, Yin X, Ma P, **g Y, Cai MC, Liu J, Zhang M, Zhang S, Shi K, Gao WQ, Di W, Zhuang G (2017) Preclinical efficacy and molecular mechanism of targeting CDK7-dependent transcriptional addiction in ovarian cancer. Mol Cancer Ther 16:1739–1750. https://doi.org/10.1158/1535-7163.MCT-17-0078
Zhang X, Li T, Zhao L, Xu H, Yan C, ** Y, Wang Z (2023) DFT-aided infrared and electronic circular dichroism spectroscopic study of cyclopeptide S-PK6 and the exploration of its antitumor potential by molecular docking. J Mol Struct 1278:134903. https://doi.org/10.1016/j.molstruc.2023.134903
Zoete V, Cuendet MA, Grosdidier A, Michielin O (2011) SwissParam: a fast force field generation tool for small organic molecules. J Comput Chem 32:2359–2368. https://doi.org/10.1002/jcc21816
Acknowledgements
The authors are thankful to Delhi Technological University for infrastructure and support.
Author information
Authors and Affiliations
Contributions
C.A. and K.M. wrote the main manuscript text, did the docking studies, S.M. gave the initial concept, and R.S. reviewed the manuscript, and supervised.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Arora, C., Madaan, K., Mehta, S. et al. Exploring isoindolin-1-ones as potential CDK7 inhibitors using cheminformatic tools. In Silico Pharmacol. 12, 51 (2024). https://doi.org/10.1007/s40203-024-00225-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40203-024-00225-0