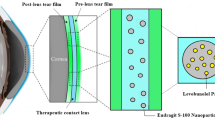
Abstract
Background
Increased intraocular pressure is a common symptom of glaucoma. In severe circumstances, it may result in loss of eyesight. Glaucoma treatment is difficult due to ocular physiological barriers that prevent medications from reaching the afflicted area. Traditional formulations (eye drops) have a short residence period and are rapidly drained away via the nasolacrimal duct, resulting in increased adverse drug responses and lower efficacy. The usage of nanoparticles such as niosomes could be one potential answer to these problems. While niosomes improve drug penetration, they have little effect on ocular retention of the medication. Contact lenses containing niosomes can assist to overcome this disadvantage.
Objective
This study aims to prepare and evaluate Brimonidine niosomes laden contact lenses for the treatment of Glaucoma.
Methods
Brimonidine niosomes were prepared using thin film hydration method and evaluated. The contact lenses were soaked in the niosomal formulation at varying intervals (3–10 days). Thereafter, the contact lenses were evaluated for %transmittance, %swelling index, drug quantification and in vitro drug release. The pharmacodynamic studies were conducted to assess the reduction in intraocular pressure (IOP) in albino rabbits. The research compared the results of the reduction in intraocular pressure caused by Brimonidine niosomes laden contact lenses with a marketed preparation of niosomes.
Results
Higher concentration of the drug was loaded in contact lenses loaded with Brimonidine niosomes compared to the marketed formulation, by soaking method. The contact lenses exhibited an optimal %transmittance of 98.02 ± 0.36 and %swelling index of 50.35 ± 0.57. Increase in the soaking time up to 7 days led to an increase in the drug concentration in the contact lenses. However, no further increase was observed after the 7th day due to saturation of the contact lenses. Brimonidine niosomes laden contact lenses provided a reduction in intraocular pressure that was similar to the marketed preparation. Further, the contact lenses provided extended release up to 20 h.
Conclusion
Brimonidine niosomes laden contact lenses exhibited superior drug loading through the soaking method, displaying optimal %transmittance and %swelling index. Soaking for 7 days increased drug concentration in contact lenses with no further increase due to saturation. These lenses reduced intraocular pressure like the marketed formulation, offering extended release for 20 h.
Graphical abstract







Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper. Any other data required can be made available upon request to the authors.
References
Yadav KS, Rajpurohit R, Sharma S. Glaucoma: current treatment and impact of advanced drug delivery systems. Life Sci. 2019;221:362–76. https://doi.org/10.1016/j.lfs.2019.02.029.
Yadav KS, Sharma S, Londhe VY. Bio-tactics for neuroprotection of retinal ganglion cells in the treatment of glaucoma. Life Sci. 2020;243:117303. https://doi.org/10.1016/j.lfs.2020.117303.
Peral A. Contact lenses as drug delivery system for glaucoma: a review. Appl Sci. 2020;10(15):5151. https://doi.org/10.3390/app10155151.
Sartini F. In vivo efficacy of contact lens drug-delivery systems in glaucoma management. A systematic review. Appl Sci. 2021;11(2):724. https://doi.org/10.3390/app11020724.
Maulvi FA, Shetty KH, Desai DT, Shah DO, Willcox MDP. Recent advances in ophthalmic preparations: Ocular barriers, dosage forms and routes of administration. Int J Pharm. 2021;608:121105. https://doi.org/10.1016/j.ijpharm.2021.121105.
Oh DJ, Chen JL, Vajaranant TS, Dikopf MS. Brimonidine for the treatment of glaucoma. Expert Opin Pharmacother. 2019;20:115–22. https://doi.org/10.1080/14656566.2018.1544241.
Cantor LB. Brimnidine in the treatment of glaucoma and ocular hypertension. Ther Clin Risk Manag. 2006;2(4):337–46. https://doi.org/10.2147/2Ftcrm.2006.2.4.337.
Alyami H. Nonionic surfactant vesicles (niosomes) for ocular drug delivery: Development, evaluation and toxicological profiling. J Drug Deliv Sci Technol. 2020;60:102069. https://doi.org/10.1016/j.jddst.2020.102069.
Kumar N, Aggarwal R, Chauhan KM. Extended levobunolol release from Eudragit nanoparticle-laden contact lenses for glaucoma therapy. Future J Pharm Sci. 2020;6:109. https://doi.org/10.1186/s43094-020-00128-9.
Fathala D, Fouad EA, Soliman GM. Latanoprost niosomes as a sustained release ocular delivery system for the management of glaucoma. Drug Dev Ind Pharm. 2020;46:806–13. https://doi.org/10.1080/03639045.2020.1755305.
Xu J, Ge Y, Bu R. Co-delivery of latanoprost and timolol from micelles-laden contact lenses for the treatment of glaucoma. J Control Release. 2019;305:18–28. https://doi.org/10.1016/j.jconrel.2019.05.025.
Xu W, Wanzhen J. Bimatoprost loaded microemulsion laden contact lens to treat glaucoma. J Drug Deliv Sci Technol. 2019;54:101330. https://doi.org/10.1016/j.jddst.2019.101330.
Maulvi FA, Patil RJ, Desai AR, Shukla MR, Vaidya RJ, Ranch KM, Vyas BA, Shah SA, Shah DO. Effect of gold nanoparticles on timolol uptake and its release kinetics from contact lenses: In vitro and in vivo evaluation. Acta Biomater. 2019;86:350–62. https://doi.org/10.1016/j.actbio.2019.01.004.
Kumar GP, Rajeshwarrao P. Nonionic surfactant vesicular systems for effective drug delivery—an overview. Acta Pharm Sin B. 2011;1(4):208–19. https://doi.org/10.1016/j.apsb.2011.09.002.
Verma A. Emerging potential of niosomes in ocular delivery. Expert Opin Drug Deliv. 2021;18:55–71. https://doi.org/10.1080/17425247.2020.1822322.
Gugleva V, Titeva S. Design and in vitro evaluation of doxycycline hyclate niosomes as a potential ocular delivery system. Int J Pharm. 2019;567:118431. https://doi.org/10.1016/j.ijpharm.2019.06.022.
Gupta P, Yadav KS. Formulation and evaluation of brinzolamide encapsulated niosomal in-situ gel for sustained reduction of IOP in rabbits. J Drug Deliv Sci Technol. 2022;67:103004. https://doi.org/10.1016/j.jddst.2021.103004.
Moiseev RV, Morrison PW. Penetration enhancers in ocular drug delivery. Pharmaceutics. 2019;11(7):321. https://doi.org/10.3390/2Fpharmaceutics11070321.
Waqas MK, Sadia H. Development and characterization of niosomal gel of fusidic acid: in-vitro and ex-vivo approaches. Des Monomers Polym. 2022;2022:165–74. https://doi.org/10.1080/2F15685551.2022.2086411.
Khan DH, Bashir S. Process optimization of ecological probe sonication technique for production of rifampicin loaded niosomes. J Drug Deliv Sci Technol. 2019;50:27–33. https://doi.org/10.1016/j.jddst.2019.01.012.
Verma P, Yadav KS. Quality by design (QbD) enabled and Box-Behnken design assisted approach for formulation of tranexamic acid loaded stratum corneum lipid liposomes. J Drug Deliv Sci Technol. 2023;86:104571. https://doi.org/10.1016/j.jddst.2023.104571.
Silva LD, Htar TT. Characterization, optimization, and in vitro evaluation of Technetium-99m-labeled niosomes. Int J Nanomed. 2019;14:1101–17. https://doi.org/10.2147/2FIJN.S184912.
Singh KH, Shinde UA. Chitosan nanoparticles for controlled delivery of Brimonidine to the ocular membrane. Pharmazie. 2011;66:594–9. https://doi.org/10.1691/ph.2011.0349.
Bayindir ZS, Yuksel N. Investigation of formulation variables and excipient interaction on the production of niosomes. AAPS PharmSciTech. 2012;13:826–35. https://doi.org/10.1208/2Fs12249-012-9805-4.
Mohamad EA, Fahmy HM. Niosomes and liposomes as promising carriers for dermal delivery of Annona squamosa extract. Braz J Pharm Sci. 2020;56:e18096. https://doi.org/10.1590/s2175-97902019000318096.
Eldeeb AE, Salah S, Ghorab M. Proniosomal gel-derived niosomes: an approach to sustain and improve the ocular delivery of Brimonidine; formulation, in-vitro characterization, and in-vivo pharmacodynamic study. Drug Deliv. 2019;26:509–21. https://doi.org/10.1080/10717544.2019.1609622.
Zheng J, Clogston DJ, Patri AK. Dobrovolskaia AM, McNeil SE. Sterilization of silver nanoparticles using standard gamma irradiation procedure affects particle integrity and biocompatibility. J Nanomed Nanotechnol 2011:95–101. https://doi.org/10.4172/2F2157-7439.S5-001.
Padala S, Sripada R, Gundabattula SB, Tadi KJ, Nallamothula PR, Raveshi F, Magharla DD. A comparative study on the efficacy of brinzolamide/timolol versus brinzolamide/brimonidine fixed drug combinations in primary open-angle glaucoma. Future J Pharm Sci. 2020;6(1):1–7.
Dubey A, Prabhu P. Development and investigation of niosomes of Brimonidine and timolol maleate for the treatment of glaucoma. Int J PharmTech Res. 2014;6(3):942–50.
Maulvi FA, Soni TG, Shah DO. A review on therapeutic contact lenses for ocular drug delivery. Drug Deliv. 2016;23:3017–26. https://doi.org/10.3109/10717544.2016.1138342.
Maulvi FA, et al. Controlled bimatoprost release from graphene oxide laden contact lenses: in vitro and in vivo studies. Colloids Surf B. 2020;208:112096. https://doi.org/10.1016/j.colsurfb.2021.112096.
Jung HJ, Jaoude MA. Glaucoma therapy by extended release of timolol from nanoparticle loaded. J Control Rel. 2012;2012:82–9. https://doi.org/10.1016/j.jconrel.2012.10.010.
Nguyen DC. Pharmaceutical-loaded contact lenses as an ocular drug delivery system: A review of critical lens characterization methodologies with reference to ISO standards. Cont Lens Anterior Eye. 2021;44(6):101487. https://doi.org/10.1016/j.clae.2021.101487.
Pereira-da-Mota AF, Lopez MV. Atorvastatin-eluting contact lenses: effects of molecular imprinting and sterilization on drug loading and release. Pharmaceutics. 2021;13(5):606. https://doi.org/10.3390/pharmaceutics13050606.
Gulsen D, Li CC, Chauhan A. Dispersion of DMPC liposomes in contact lenses for opthalmic drug delivery. Curr Eye Res. 2005;30:1071–80. https://doi.org/10.1080/02713680500346633.
Jain N, Verma A, Jain N. Formulation and investigation of pilocarpine hydrochloride niosomal gels for the treatment of glaucoma: intraocular pressure measurement in white albino rabbits. Drug Deliv. 2020;27:888–99. https://doi.org/10.1080/10717544.2020.1775726.
Elshaer A, et al. Nanoparticle-laden contact lens for controlled ocular delivery of prednisolone: Formulation optimization using statistical experimental design. Pharmaceutics. 2016;8(2):14. https://doi.org/10.3390/pharmaceutics8020014.
Basiri L, Rajabzadeh G, Bostan A. α-Tocopherol-loaded niosome prepared by heating method and its release behavior. Food Chem. 2017;221:620–8. https://doi.org/10.1016/j.foodchem.2016.11.129.
Abdelkader H, Ismail S, Kamal A, Alany RG. Preparation of niosomes as an ocular delivery system for naltrexone hydrochloride: Physicochemical characterization. Pharmazie. 2010;65:811–7.
Souza JF, Maia KN. Ocular inserts based on chitosan and Brimonidine: development, characterization and biocompatibility. J Drug Deliv Sci Technol. 2016:21–30. https://doi.org/10.1016/j.jddst.2016.01.008.
Pramod K, Ali J. Unveiling the compatibility of eugenol with formulation excipients by systematic drug-excipient compatibility studies. Jo Anal Sci Technol. 2015. https://doi.org/10.1186/s40543-015-0073-2.
Mohamed HB, El-Shanawany SM, Hamad MA, Elsabahy M. Niosomes: a strategy toward prevention of clinically significant drug incompatibilities. Sci Rep. 2017;7(1):6340. https://doi.org/10.1038/s41598-017-06955-w.
Sharma PK, Chauhan MK. Optimization and characterization of Brimonidine Nanoparticles-loaded in situ gel for the treatment of glaucoma. Curr Eye Res. 2021:1703–1716. https://doi.org/10.1080/02713683.2021.1916037.
Gupta U. Spectroscopic studies of cholesterol: Fourier Transform Infra-red and vibrational Frequency Analysis. Materials Focus. 2014:211–217. https://doi.org/10.1166/mat.2014.1161.
Soltys-Robitaille CE. The relationship between contact lens surface charge and in-vitro protein deposition levels. Biomaterials. 2001:3257–3260. https://doi.org/10.1016/S0142-9612(01)00163-6.
Farmoudeh A, Akbari J, Saeedi M. Methylene blue-loaded niosome: preparation, physicochemical characterization, and in vivo wound healing assessment. Drug Deliv Transl Res. 2020. https://doi.org/10.1007/s13346-020-00715-6.
Hao YM, Li K. Entrapment and release difference resulting from hydrogen bonding interactions in niosome. Int J Pharm. 2011:245–253. https://doi.org/10.1016/j.ijpharm.2010.10.027.
Mavaddati MA, Moztarzadeh F, Baghbani F. Effect of formulation and processing variables on dexamethasone entrapment and resease of niosomes. J Clust Sci. 2015:2065–2078. https://doi.org/10.1007/s10876-015-0908-4.
Barakat HS. Vancomycin-eluting niosomes: a new approach to the inhibition of staphylococcal biofilm on abiotic surfaces. AAPS PharmSciTech. 2014:1263–1274. https://doi.org/10.1208/2Fs12249-014-0141-8.
Gharbhavi M, Amani J. Niosome: a promising nanocarrier for natural drug delivery through Blood brain barrier. Adv Pharmacol Sci. 2018:6847971. https://doi.org/10.1155/2018/6847971.
Franco P, Marco ID. Contact lenses as ophthalmic drug delivery systems: a review. Polymers. 2021;13(7):1102. https://doi.org/10.3390/2Fpolym13071102.
Moreddu R, Vigolo D, Yetisen AK. Contact lens technology: from fundamentals to applications. Adv Healthc Mater. 2019;8(15):1900368. https://doi.org/10.1002/adhm.201900368.
Riley C, Chalmers RL, Pence N. The impact of lens choice in the relief of contcat lens related symptoms and ocular surface findings. Cont Lens Anterior Eye. 2005;23:13–9. https://doi.org/10.1016/j.clae.2004.09.002.
Mun J, Jeong S, Cho S. Drug-eluting contact lens containing cyclosporineloaded cholesterol-hyaluronate micelles for dry eye syndrome. RSC Adv. 2019;9(29):16578–85. https://doi.org/10.1039/C9RA02858G.
Kesavan K, Kant S, Singh PN, Pandit JK. Mucoadhesive chitosan-coated cationic microemulsion of dexamethasone for ocular delivery: in vitro and in vivo evaluation. Curr Eye Res. 2013;38(3):342–52. https://doi.org/10.3109/02713683.2012.745879.
Aj R, Hn Y, Sb S. Natural gums as sustained release carriers: development of gastroretentive drug delivery system of ziprasidone HCl. DARU J Pharm Sci. 2012;20:58. https://doi.org/10.1186/2008-2231-20-58.
Rad MS, Mohajeri SA. Simultaneously load and extended release of betamethasone and ciprofloxacin from Vitamin E-loaded silicone-based soft contact lenses. Curr Eye Res. 2016:1–7. https://doi.org/10.3109/02713683.2015.1107591.
Ammar HO. Nanoemulsion as a potential ophthalmic delivery system for dorzolamide hydrochloride. AAPS PharmSciTEch. 2009:808. https://doi.org/10.1208/s12249-009-9268-4.
Funding
The work did not receive any fundings.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
The work did not involve any human study so no need of any ethical approval.
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tripathi, S., Yadav, K.S. Development of brimonidine niosomes laden contact lenses for extended release and promising delivery system in glaucoma treatment. DARU J Pharm Sci 32, 161–175 (2024). https://doi.org/10.1007/s40199-023-00500-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40199-023-00500-z