Abstract
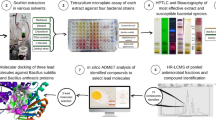
Plants are being used in treating diseases since the beginning of human civilization. Cinnamomum tamala is a traditionally used medicinal plant used for various medicinal purposes. Methanolic leaf extract of the plant was subjected to bioassay-guided fractionation. Significant antimicrobial activity was recorded in the chloroform and methanolic fraction. Methanolic fraction was further fractionated by column chromatography and six fractions were collected of which F4 showed high antimicrobial activity. Bioautography of chloroform fraction and F4 reconfirm the presence of bioactive compounds. GC–MS analysis showed the presence of 7 compounds in F4 (dominating compound 2-pentanone,4-hydroxy-4-methyl) and 10 compounds in chloroform fraction (dominating compound 1,5-heptadien-4-one,3,6,3-trimethyl). Both the dominating compounds satisfied Lipinski rule and were within permissible limits to act like drug. Both interacted with drug targets, however 1,5-heptadien-4-one,3,6,3-trimethyl showed a better binding affinity and thus can be a potential inhibitor against the selected drug targets. Thus from the study, it can be concluded that the compound 1,5-heptadien-4-one,3,6,3-trimethyl has the potential to be a lead molecule that can be used in formulation of antimicrobial agents. Further studies in animal model system are needed to establish its efficacy.





Similar content being viewed by others
References
Das S, Khan ML, Rabha A et al (2009) Ethnomedicinal plants of Manash National park, Assam Northeast India. Indian J Tradit Knowl 8(4):514–517
Saxena M, Saxena J, Nema R, Singh D, Gupta A (2013) Phytochemistry of medicinal plants. J Pharmacogn Phytochem 1(6):168–172
Buragohain J (2011) Ethnomedicinal plants used by the ethnic communities of Tinsukia district of Assam. India Recent Res Sci Technol 3(9):31–42
Kambu K, Di Phenzu N, Coune C, Wauter JN, Angenot L (1982) Contributions to the study of insecticidal and chemical properties of Eucalyptus saligna of Zaire. Plants Med Phytother (Plantes médicinales et phytothérapie) 16(1):34–38
Yen GC, Duh PD, Tsai CL (1993) Relationship between antioxidant activity and maturity of peanut hulls. J Agric Food Chem 41:67–70
Lemos TLG, Matos FJA, Alencar JW, Crareiro AA, Clark AM, Chesnary JD (1990) Antimicrobial activity of essential oils of Brazilian plants. Phytother Res 4:82–84. https://doi.org/10.1002/ptr.2650040210
Benoitvical F, Valentin A, Mallic M, Bassierc JM (2001) Antiplasmodial activity of Colchlospermum planchonii and C. tinctorium tubercle essential oils. J Essent Oil Res 13:65–67. https://doi.org/10.1080/10412905.2001.9699609
Chew YL, Chan EWL, Tan PL, Lim YY, Stanslas J, Goh JK (2011) Assessment of phytochemical content, polyphenolic composition, antioxidant and antibacterial activities of Leguminosae medicinal plants in Peninsular Malaysia. BMC Complement Altern Med 11:1–10. https://doi.org/10.1186/1472-6882-11-12
Veeresham C (2012) Natural products derived from plants as a source of drugs. J Adv Pharm Technol Res 3:200–201. https://doi.org/10.4103/2231-4040.104709
Qazi MA, Molvi K (2016) Herbal medicine: a comprehensive review. Int J Pharm Res 8(2):1–5
Farnsworth NR (1966) Biological and phytochemical screening of plants. J Pharm Sci 55:225–276. https://doi.org/10.1002/jps.2600550302
Eswaran MB, Surendran S, Vijayakumar M, Ojha SK, Rawat AKS, Rao CV (2010) Gastroprotective activities of Cinnamomum tamala leaves on experimental gastric ulcers in rats. J Ethnopharmacol 128:534–540. https://doi.org/10.1016/j.jep.2010.01.036
Shah M, Panchal M (2010) Ethnopharmacological properties of Cinnamomum tamala—a review. Int J Pharm Sci 5(3):141–144
Barukial J, Sharma JN (2011) Ethnomedicinal plants used by the people of Gholaghat Dist, Assam India. Int J Med Aromat Plants 1(3):203–211
Sharma UK, Pegu S (2011) Ethnobotany of religious and supernatural beliefs of the mising tribes of Assam with special reference to the ‘Dobur Uie.’ J Ethnobiol Ethnomed 7:1–13. https://doi.org/10.1186/1746-4269-7-16
Rahman M, Khatun A, Islam MM, Akter MN, Chowdhury SA, Ali Khan MA, Shahid MIZ, Rahman AA (2013) Evaluation of antimicrobial, cytotoxic, thrombolytic, diuretic properties and total phenolic content of Cinnamomum tamala. Int J Green Pharm 7(3):236–243. https://doi.org/10.22377/ijgp.v7i3.329
Adegoke AA, Adebayo-tayo BC (2009) Antibacterial activity and phytochemical analysis of leaf extracts of Lasienthera africanum. Afr J Biotechnol 8(1):77–80
Mahlo SM (2009) Characterization and biological activity of antifunga lcomponents present in Breonadia salicina (Rubiaceae) leaves. In: PhD Thesis. University of Pretoria, South Africa
Thirach S, Tragoolpua K, Punjaise S, Khamwan C, Jatisatienr C, Kunyanone N (2003) Antifungal activity of some medicinal plant extracts against Candida albicans and Cryptococcus neoformans. Acta Hortic 597(31):217–221
Balouiri M, Sadik M, Ibnsouda SK (2016) Methods for in vitro evaluating antimicrobial activity: a review. J Pharm Anal 6(2):71–79. https://doi.org/10.1016/j.jpha.2015.11.005
Valgas C, De Souza SM, Smania EFA, Smania A Jr (2007) Screening methods to determine antibacterial activity of natural products. Braz J Microbiol 38(2):369–380. https://doi.org/10.1590/S1517-83822007000200034
Gherraf N, Zellagui A, Kabouche A, Lahouel M, Salhi R, Rhouati S (2013) Chemical constituents and antimicrobial activity of Ammodaucus leucotricus. Arab J Chem 10(2):2476–2478. https://doi.org/10.1016/j.arabjc.2013.09.013
Daina A, Michielin O, Zoete V (2017) Swiss ADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci Rep 71:1–13. https://doi.org/10.1038/srep42717
Trott O, Olson AJ (2010) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreading. J Compu Chem 31(2):455–461. https://doi.org/10.1002/jcc.21334
Suleiman MM, Mc Gaw LJ, Naidoo V, Eloff JN (2009) Detection of antimicrobial compounds by bioautography of different extracts of the leavesof selected South African tree species. Afr J Tradit Complement Altern Med 7(1):64–78. https://doi.org/10.4314/ajtcam.v7i1.57269
Xu GL, Geng D, **e M, Teng KY, Tian YX, Liu ZZ, Yan C, Wang Y, Zhang X, Sing Y, Yang Y, She GM (2015) Chemical composition, antioxidative and anticancer activities of the essential oil: Curcumae rhizoma and Sparganii rhizoma, a traditional herbal pair. Molecules 20(9):15781–15796
Huang X, Ge SY, Lui JH, Wang Y, Liang XY, Yuan HB (2018) Chemical composition and bioactivity of essential oil from Artemisia lavandulaefolia (Asteraceae) on Plutella xylostella (lepidoptera: Plutellidae). Fla Entomol 101(1):44–48. https://doi.org/10.1653/024.101.0109
Durojaye OA, Ajuluchukwu PS, Cosmas S, Igomu R, Okagu IA, Sani SB (2018) Binding energy prediction and molecular docking studies of falcarindiol and its monosubstituted analogues against Aspergillus fumigatus chitinase, the in-silico pharmacokinetics. Int J Eng Res 9(6):271–283
Sahare P, Moon A (2016) In-silico docking studies of phytoligands against E coli PBP3, Approach towards novel antibacterial therapeutic agents. Int J Pharm Sci 7(9):3703–3711
Sharanya M, Satishkumar R (2015) In silico screening of phytocompounds of Vitex negundo linn leaves for fungal chitinase. J Sci Technol 3(1):1–10
Acknowledgements
The authors are thankful to the Department of Life Sciences, Dibrugarh University for help and support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Significance statement: The emergence of MDR microbes is increasing day by day. This has led to an urgent need to search for new antimicrobial compounds with novel mode of action. In the present study, Cinnamomum tamala has been selected based on its ethnomedicinal uses.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shah, R.K., Yadav, R.N.S. Bioassay-Guided Fractionation of Methanolic Leaf Extract of Cinnamomum tamala, GC–MS Analysis and In Silico Studies. Proc. Natl. Acad. Sci., India, Sect. B Biol. Sci. (2024). https://doi.org/10.1007/s40011-024-01578-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40011-024-01578-4