Abstract
Background
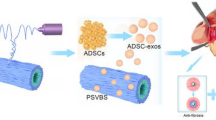
3D-printing is widely used in regenerative medicine and is expected to achieve vaginal morphological restoration and true functional reconstruction. Mesenchymal stem cells-derived exosomes (MSCs-Exos) were applyed in the regeneration of various tissues. The current study aimed to explore the effctive of MSCs-Exos in vaginal reconstruction.
Methods
In this work, hydrogel was designed using decellularized extracellular matrix (dECM) and gelatin methacrylate (GelMA) and silk fibroin (SF). The biological scaffolds were constructed using desktop-stereolithography. The physicochemical properties of the hydrogels were evaluated; Some experiments have been conducted to evaluate exosomes’ effect of promotion vaginal reconstruction and to explore the mechanism in this process.
Results
It was observed that the sustained release property of exosomes in the hydrogel both in vitro and in vitro.The results revealed that 3D scaffold encapsulating exosomes expressed significant effects on the vascularization and musule regeneration of the regenerative vagina tissue. Also, MSCs-Exos strongly promoted vascularization in the vaginal reconstruction of rats, which may through the PI3K/AKT signaling pathway.
Conclusion
The use of exosome-hydrogel composites improved the epithelial regeneration of vaginal tissue, increased angiogenesis, and promoted smooth muscle tissue regeneration. 3D-printed, lumenal scaffold encapsulating exosomes might be used as a cell-free alternative treatment strategy for vaginal reconstruction.
Graphical abstract









Similar content being viewed by others
References
Karapinar OS, Ozkan M, Okyay AG, Dolapçıoğlu K. Evaluation of vaginal agenesis treated with the modified McIndoe technique: a retrospective study. J Turk Ger Gynecol Assoc. 2016;17:101–5.
Motta GL, Tavares PM, Burttet LM, Burttet LM, Berger M, Neto BS, et al. Vaginoplasty with full-thickness mesh skin graft for vaginal agenesis. Urology. 2016;98:200–3.
Tian YP, Zhao SD, Zheng JH, Li ZK, Hou CX, Qi X, et al. A stereological study of 3D printed tissues engineered from rat vaginas. Ann Transl Med. 2020;8:1490.
Han C, Sun X, Liu L, Jiang HY, Shen Y, Xu XY, et al. Exosomes and their therapeutic potentials of stem cells. Stem Cells Int. 2016;2016:7653489.
Tolar J, Le Blanc K, Keating A, Blazar B. Concise review: hitting the right spot with mesenchymal stromal cells. Stem Cells. 2010;28:1446–55.
Lai RC, Yeo RWY, Lim SK. Mesenchymal stem cell exosomes. Semin Cell Dev Boil. 2015;40:82–8.
Gong M, Yu B, Wang JC, Wang YG, Liu M, Christian P, et al. Mesenchymal stem cells release exosomes that transfer miRNAs to endothelial cells and promote angiogenesis. Oncotarget. 2017;8:45200–12.
Fang S, Xu C, Zhang YT, Xue CY, Yang C, Bi HD, et al. Umbilical cord-derived mesenchymal stem cell-drived exosomal microRNAs suppress myofibroblast differentiation by inhibiting the transforming growth factor-/SMAD2 pathway during wound healing. Stem Cells Transl Med. 2016;5:1425–39.
Shao M, Xu Q, Wu Z, Chen YM, Shu YK, Cao X, et al. Exosomes derived from human umbilical cord mesenchymal stem cells ameliorated IL-6-induced acute liver injury through miR-455-3p. Stem Cell Res Ther. 2020;11:37.
Zhang B, Yeo RW, Tan KH, Lim S. Focus on extracellular vesicles: therapeutic potential of stem cell-derived extracellular vesicles. Int J Mol Sci. 2016;174:17.
Riau AK, Ong HS, Gary HFY, Mehta JS. Sustained delivery system for stem cell-derived exosomes. Front Pharmacol. 2019;10:1368.
Ding JX, Zhang XY, Chen LM, Huan KQ. Vaginoplasty using acellular porcine small intestinal submucosa graft in two patients with Meyer-von-Rokistansky-Kuster-Hauser syndrome: a prospective new technique for vaginal reconstruction. Gynecol Obstet Invest. 2013;75:93–6.
Zhang JK, Du RX, Zhang L, Li YN, Zhang ML, Zhao S, et al. A new material for tissue engineered vagina reconstruction: acellular porcine vagina matrix. J Biomed Mater Res A. 2017;105:1949–59.
Badylak SF, Freytes DO, Gilbert TW. Extracellular matrix as a biological scaffold material: structure and function. Acta Biomater. 2009;5:1–13.
Brown BN, Badylak SF. Extracellular matrix as an inductive scaffold for functional tissue reconstruction. Transl Res. 2014;163:268–85.
Saini G, Segaran N, Mayer JL, Saini A, Albadawi H, Oklu R. Applications of 3D Bioprinting in tissue engineering and regenerative medicine. J Clin Med. 2021;10:4966.
Noor N, Shapira A, Edri R, Gal I, Wertheim L, Dvir T. 3D printing of personalized thick and perfusable cardiac patches and Hearts. 2019;6:1900344.
Sher D. 3D biopringed blood vessels pump new life into bioficial. https://3dprintingindustry.com/news/fraunhofer-3d-bioprinted-blood-vessels-pumps-new-life-bioprinted-organ-research-57283. Accessed 16 November 2020
Samuelson K. 3D Printed ovaries produce healthy offspring. https://news.northwestern.edu/stories/2017/may/3-d-printed-ovaries-offspring. Accessed 16 May 2017
Wake Forest School of Medicine. Replacement organs and tissue. https://www.ncbiotech.org/news/wake-forest-institute-working-regenerative-medicine-therapies-40-organs-tissues. Accessed 19 June 2022
The European Space Agency. Upside-down 3D-printed skin and bone, for humans to mars. https://www.esa.int/Enabling_Support/Space_Engineering_Technology/Upside-down_3D-printed_skin_and_bone_for_humans_to_Mars. Accessed 16 November 2020.
Pandorum. Bio-engineered human cornea. http://www.pandorumtechnologies.com/cornea.php. Accessed on 16 November 2020.
Tsiapalis D, O’Driscoll L. Mesenchymal stem cell derived extracellular vesicles for tissue engineering and regenerative medicine applications. Cells. 2020;9:991.
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Fc M, Krause D, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The international society for cellular therapy position statement. Cytotherapy. 2006;8:315–7.
Lötvall J, Hill AF, Hochberg F, Buzás EI, Di Vizio D, Gardiner C, et al. Minimal experimental requirements for definition of extracellular vesicles and their functions: a position statement from the international society for extracellular vesicles. J Extracell Vesicles. 2014;3:26913.
**ao WQ, He JK, Nichol JW, Wang L, Hutson CB, Wang B, et al. Synthesis and characterization of photocrosslinkable gelatin and silk fibroin interpenetrating polymer network hydrogels. Acta Biomater. 2011;7:2384–93.
Potente M, Gerhardt H, Carmeliet P. Basic and therapeutic aspects of angiogenesis. Cell. 2011;146:873–87.
Antimisiaris SG, Mourtas S, Marazioti A. Exosomes and exosome-inspired vesicles for targeted drug delivery. Pharmaceutics. 2018;10:218.
Zhang S, Chu WC, Lai RC, Lim SK, Hui JH, Toh WS. Exosomes derived from humanembryonic mesenchymal stem cells promote osteochondral regeneration. Osteoarthr Cartil. 2016;24:2135–40.
Zhang YQ, Zhu ZY, Hua KQ, Yao LQ, Liu YJ, Ding JX. Umbilical cord-derived mesenchymal stem cell transplantation in vaginal replacement in vitro and in a rat model. Am J Transl Res. 2018;10:3762–72.
Xu YY, Qiu Y, Lin QN, Huang CS, Li J, Chen LQ, et al. miR-126-3p-loaded small extracellular vesicles secreted by urine-derived stem cells released from a phototriggered imine crosslink hydrogel could enhance vaginal epithelization after vaginoplasty. Stem Cell Res Ther. 2022;13:331.
Douroumis D. 3D printing of pharmaceutical and medical applications: a new era. Pharm Res. 2019;36:41–2.
Trubiani O, Marconi GD, Pierdomenico SD, Piattelli A, Diomede F, Pizzicannella J. Human oral stem cells, biomaterials and extracellular vesicles: a promising tool in bone tissue repair. Int J Mol Sci. 2019;20:4987.
Kim BS, Das S, Jang J, Cho D-W. Decellularized extracellular matrix-based bioinks for engineering tissue and organ-specific microenvironments. Chem Rev. 2020;120:10608–61.
Rowland CR, Glass KA, Ettyreddy AR, Catherine CG, Jared RLM, Nguyen PTH, et al. Regulation of decellularized tissue remodeling via scaffold-mediated lentiviral delivery in anatomically-shaped osteochondral constructs. Biomaterials. 2018;177:161–75.
Choudhury D, Tun HW, Wang T, Naing MW. Organ-derived decellularized extracellular matrix: a game changer for bioink manufacturing? Trends Biotechnol. 2018;36:787–805.
Abaci A, Guvendiren M. Designing decellularized extracellular matrix-based bioinks for 3D bioprinting. Adv Healthc Mater. 2020;9:e2000734.
Uygun BE, Soto-Gutierrez A, Yagi H, Izamis ML, Guzzardi MA, Shulman C, et al. Organ reengineering through development of a transplantable recellularized liver graft using decellularized liver matrix. Nat Med. 2010;16:814–20.
Petersen TH, Calle EA, Zhao LP, Lee EJ, Gui LQ, Raredon MB, et al. Tissue-engineered lungs for in vivo implantation. Science. 2010;329:538–41.
Hou CX, Zheng JH, Li ZK, Qi XJ, Tian YP, Zhang ML, et al. Printing 3D vagina tissue analogues with vagina decellularized extracellular matrix bioink. Int J Biol Macromol. 2021;180:177–86.
Chen PF, Zheng L, Wang YY, Tao M, **e Z, **a C, et al. Desktop-stereolithography 3D printing of a radially oriented extracellular matrix/mesenchymal stem cell exosome bioink for osteochondral defect regeneration. Theranostics. 2019;9:2439–59.
**ao YL, Zhang JK, Tian YP, Zhang ML, Du YF, Meng L, et al. Vaginal reconstruction with a double-sided biomembrane-a preclinical experimental study on large animals. Biomater Sci. 2023;11:7077–89.
**ao YL, Tian YP, Zhang JK, Li Q, Shi WX. Small intestinal submucosa promotes angiogenesis via the Hippo pathway to improve vaginal repair. Biomol Biomed. 2023;4:838–47.
Pennarossa G, Arcuri S, De Iorio T, Gandolfi F, Brevini TAL. Current advances in 3D tissue and organ reconstruction. Int J Mol Sci. 2021;22:830.
Khayambashi P, Iyer J, Pillai S, Upadhyay A, Zhang Y, Tran SD. Hydrogel encapsulation of mesenchymal stem cells and their derived exosomes for tissue engineering. Int J Mol Sci. 2021;22:684.
Yang J, Chen Z, Pan D, Li H, Shen J. Umbilical cord-derived mesenchymal stem cell-derived exosomes combined pluronic F127 hydrogel promote chronic diabetic wound healing and complete skin regeneration. Int J Nanomedicine. 2020;15:5911–26.
Acknowledgements
The authors would like to thank Dr. Yanbiao Song for his technical assistance. This work was supported by the Natural Precision Medicine Joint Fund Nurture Project of Hebei Province [H2021206463]; the Medical Science Research Project of Hebei Province [20240996]; the Medical Science Research Plan Project of Hebei Province [20210080]; and the Innovative Capacity Improvement Plan of Hebei Province [20577705D].
Author information
Authors and Affiliations
Contributions
Wenxin Shi, Jiahua Zheng and **anghua Huang designed the study; **kun Zhang; **aoli Dong and Zhongkang Li acquisited and analyzed the datas;Yanlai **ao Interpreted the datas. Wenxin Shi drafted the manuscript. **anghua Huang and Yanfang Du supervised the article. All authors edited and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no financial conflicts of interest.
Ethical statement
The animal studies were performed after receiving approval of the Institutional Animal Care and Use Committee (IACUC) in the Second Hospital of Hebei Medical University (Approval Letter No:2023-AE083).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shi, W., Zheng, J., Zhang, J. et al. Desktop-Stereolithography 3D Printing of a Decellularized Extracellular Matrix/Mesenchymal Stem Cell Exosome Bioink for Vaginal Reconstruction. Tissue Eng Regen Med (2024). https://doi.org/10.1007/s13770-024-00649-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13770-024-00649-x