Abstract
Biochar-based chromium adsorption is a relatively new and affordable method for remediation of hazardous metal species from contaminated environments. Coir fiber is an agricultural waste produced from coconut processing industry. This study focused on the synthesis of coconut coir-based biochars and evaluated their Cr(VI) removal efficiency. Biochar samples were prepared under different pyrolysis temperatures (300–700 °C) and characterized using Thermogravimetric, Carbon–Hydrogen–Nitrogen–Sulfur–Oxygen and Brunauer–Emmett–Teller surface area analyzer. Following a screening process to determine the efficacy of removing Cr(VI) of all biochar samples, coconut coir biochar prepared at 400 °C (CBC400) showed relatively better (~ 90%) Cr(VI) removal. The effects of various physicochemical factors such as pH, initial metal concentration, treatment time, and adsorbent dosage on Cr(VI) removal process are investigated and optimized. It was also found that at 50 mg/L initial Cr(VI) concentration, the CBC400 can remove 40.3 mg (qmax) Cr(VI) per 1 g adsorbent dosage within 24 h under optimum condition. It was demonstrated that Cr(VI) adsorption using CBC400 has a good fit with Langmuir isotherm and pseudo-second-order kinetics model. The interaction between Cr(VI) ions and biosorbent were well established by characterising prior and post-Cr(VI)-treatment of biochar samples using scanning electron microscopy with energy-dispersive X-ray spectroscopy, X-ray diffraction and Fourier transform infrared spectroscopy. From the overall findings of the study, it was concluded that CBC400 can be suitably applied for Cr(VI) removal with possible safe disposal in cost-effective and environmentally safe way.
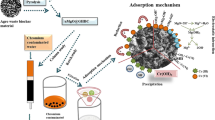
Graphical abstract









Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Ahmad A, Al-Swaidan HM, Alghamdi AH, Alotaibi KM, Alswieleh AM, Albalwi AN, Bajuayfir E (2021) Efficient sequester of hexavalent chromium by chemically active carbon from waste valorization (Phoenix Dactylifera). J Anal Appl Pyrolysis 155:105075. https://doi.org/10.1016/j.jaap.2021.105075
Ali A, Alharthi S, Al-Shaalan NH, Naz A, Fan HJS (2023) Efficient removal of hexavalent chromium (Cr (VI)) from wastewater using amide-modified biochar. Molecules 28:5146. https://doi.org/10.3390/molecules28135146
Altundogan HS (2005) Cr (VI) removal from aqueous solution by iron (III) hydroxide-loaded sugar beet pulp. Process Biochem 40:1443–1452. https://doi.org/10.1016/j.procbio.2004.06.027
Angalaeeswari K, Kamaludeen SPB (2017) Production and characterization of coconut shell and mesquite wood biochar. Int J Chem Stud 5:442–446
APHA, AWWA (1995). Standard methods for the examination of water and wastewater. American public health association/American water works association/water environment federation, Washington DC, USA.
Bakshi S, Banik C, Rathke SJ, Laird DA (2018) Arsenic sorption on zero-valent iron-biochar complexes. Water Res 137:153–163. https://doi.org/10.1016/j.watres.2018.03.021
Behera SS, Das S, Murmu BM, Mohapatra RK, Sahoo TR, Parhi PK (2016) Kinetics, thermodynamics and isotherm studies on adsorption of methyl violet from aqueous solution using activated bio-adsorbent: phragmites karka. Curr Environ Eng 3:229–241. https://doi.org/10.2174/22127178036661610271443
Behera SS, Sourav D, Parhi PK, Tripathy SK, Mohapatra RK, Mayadhar D (2017) Kinetics, thermodynamics and isotherm studies on adsorption of methyl orange from aqueous solution using ion exchange resin Amberlite IRA-400. Desalin Water Treat 60:249–260
Brewer CE (2012) Biochar characterization and engineering. Iowa State University. https://lib.dr.iastate.edu/etd/12284
Bulgariu L, Lupea M, Bulgariu D, Rusu C, Macoveanu M (2013) Equilibrium study of Pb (II) and Cd (II) biosorption from aqueous solution on marine green algae biomass. Environ Eng Manag J 12:183–190
Cantrell KB, Hunt PG, Uchimiya M, Novak JM, Ro KS (2012) Impact of pyrolysis temperature and manure source on physicochemical characteristics of biochar. Bioresour Technol 107:419–428. https://doi.org/10.1016/j.biortech.2011.11.084
Chaudhary A, gul JavidBughioFaisal KEN (2023) Study on histo-biochemical biomarkers of chromium induced toxicity in Labeo rohita. Emerg Contam 9:100204. https://doi.org/10.1016/j.emcon.2023.100204
Chen B, Chen Z, Lv S (2011) A novel magnetic biochar efficiently sorbs organic pollutants and phosphate. Bioresour Technol 102:716–723. https://doi.org/10.1016/j.biortech.2010.08.067
Chen T, Zhang Y, Wang H, Lu W, Zhou Z, Zhang Y, Ren L (2014) Influence of pyrolysis temperature on characteristics and heavy metal adsorptive performance of biochar derived from municipal sewage sludge. Biores Technol 164:47–54. https://doi.org/10.1016/j.biortech.2014.04.048
Chen YY, Wang BY, **n J, Sun P, Wu D (2018) Adsorption behaviour and mechanism of Cr(VI) by modified biochar derived from Enteromorpha prolifera. Ecotoxicol Environ Saf 164:440–447. https://doi.org/10.1016/j.ecoenv.2018.08.024
Chen X, Zhang R, Zhao B, Fan G, Li H, Xu X, Zhang M (2020) Preparation of porous biochars by the co-pyrolysis of municipal sewage sludge and hazelnut shells and the mechanism of the nano-zinc oxide composite and Cu (II) adsorption kinetics. Sustainability 12:8668. https://doi.org/10.3390/su12208668
Choudhary B, Paul D, Singh A, Gupta T (2017) Removal of hexavalent chromium upon interaction with biochar under acidic conditions: mechanistic insights and application. Environ Sci Pollut Res 24:16786–16797. https://doi.org/10.1007/s11356-017-9322-9
Dong X, Ma LQ, Li Y (2011) Characteristics and mechanisms of hexavalent chromium removal by biochar from sugar beet tailing. J Hazard Mater 190:909–915. https://doi.org/10.1016/j.jhazmat.2011.04.008
Duan S, Ma W, Pan Y, Meng F, Wu L (2017) Synthesis of magnetic biochar from iron sludge for the enhancement of Cr (VI) removal from solution. J Taiwan Inst Chem Eng 80:835–841. https://doi.org/10.1016/j.jtice.2017.07.002
Fang S, Huang X, **e S, Du J, Zhu J, Wang K, Zhuang Q, Huang X (2022) Removal of chromium (VI) by a magnetic nanoscale zerovalent iron-assisted chicken manure-derived biochar: adsorption behavior and synergetic mechanism. Front Bioeng Biotechnol 10:935525–935616. https://doi.org/10.3389/fbioe.2022.935525
Foong SY, Chan YH, Chin BLF, Lock SSM, Yee CY, Yiin CL, Peng W, Lam SS (2022) Production of biochar from rice straw and its application for wastewater remediation−an overview. Bioresour Technol 360:127588. https://doi.org/10.1016/j.biortech.2022.127588
Gabhane JW, Bhange VP, Patil PD, Bankar ST, Kumar S (2020) Recent trends in biochar production methods and its application as a soil health conditioner: a review. SN Appl Sci 2:1–21. https://doi.org/10.1007/s42452-020-3121-5
Gonzalez MH, Araújo GC, Pelizaro CB, Menezes EA, Lemos SG, De Sousa GB, Nogueira ARA (2008) Coconut coir as biosorbent for Cr(VI) removal from laboratory wastewater. J Hazard Mater 159:252–256. https://doi.org/10.1016/j.jhazmat.2008.02.014
Guo X, Liu A, Lu J, Niu X, Jiang M, Ma Y, Li M (2020) Adsorption mechanism of hexavalent chromium on biochar: kinetic, thermodynamic, and characterization studies. ACS Omega 5:27323–27331. https://doi.org/10.1021/acsomega.0c03652
Gupta GK, Mondal MK (2023) Mechanism of Cr(VI) uptake onto sagwan sawdust derived biochar and statistical optimization via response surface methodology. Biomass Conv Bioref 13:709–725. https://doi.org/10.1007/s13399-020-01082-5
Gupta VK, Rastogi A (2009) Biosorption of hexavalent chromium by raw and acid-treated green alga Oedogonium hatei from aqueous solutions. J Hazard Mater 163:396–402. https://doi.org/10.1016/j.jhazmat.2008.06.104
Gupta VK, Rastogi A, Nayak A (2010) Adsorption studies on the removal of hexavalent chromium from aqueous solution using a low cost fertilizer industry waste material. J Colloid Interface Sci 342:135–141. https://doi.org/10.1016/j.jcis.2009.09.065
Gupta S, Yadav A, Verma N (2017) Simultaneous Cr (VI) reduction and bioelectricity generation using microbial fuel cell based on alumina-nickel nanoparticles-dispersed carbon nanofiber electrode. J Chem Eng 307:729–738. https://doi.org/10.1016/j.cej.2016.08.130
Gupta GK, Ram M, Bala R, Kapur M, Mondal MK (2018) Pyrolysis of chemically treated corncob for biochar production and its application in Cr (VI) removal. Environ Prog Sustain Energy 37:1606–1617. https://doi.org/10.1002/ep.12838
Gupta AD, Kavitha E, Singh S, Karthikeyan S (2020) Toxicity mechanism of Cu2+ ion individually and in combination with Zn2+ ion in characterizing the molecular changes of Staphylococcus aureus studied using FTIR coupled with chemometric analysis. J Biol Phys 46:395–414. https://doi.org/10.1007/s10867-020-09560-7
Gupta A, Sharma V, Sharma K, Kumar V, Choudhary S, Mankotia P, Kumar B, Mishra H, Moulick A, Ekielski A, Mishra PK (2021) A review of adsorbents for heavy metal decontamination: Growing approach to wastewater treatment. Materials 14:4702. https://doi.org/10.3390/ma14164702
Hashem MA, Payel S, Mim S, Hasan MA, Nur-A-Tomal MS, Rahman MA, Sarker MI (2022) Chromium adsorption on surface activated biochar made from tannery liming sludge: a waste-to-wealth approach. Wat Sci Eng 15:328–336. https://doi.org/10.1016/j.wse.2022.09.001
Hu Y, Ling M, Li X (2021) Preparation of lignin-based mesoporous biochar nano and microparticles, and their adsorption properties for hexavalent chromium. BioResources 16:6363–6377
Intani K, Latif S, Kabir AR, Müller J (2016) Effect of self-purging pyrolysis on yield of biochar from maize cobs, husks and leaves. Bioresour Technol 218:541–551. https://doi.org/10.1016/j.biortech.2016.06.114
Issac IR, Rajamehala M, Prabha ML, Renitta RE (2012) Comparitive study on biosorption of hexavalent chromium using Aspergillus oryzae NCIM 637 and Aspergillus sojae NCIM 1198 from electroplating effluent. Int J Chemtech Res 4:1708–1719
Jiang Y, Dai M, Yang F, Ali I, Naz I, Peng C (2022) Remediation of chromium (VI) from groundwater by metal-based biochar under anaerobic conditions. Water 14:894. https://doi.org/10.3390/w14060894
Kabir G, Hameed BH (2017) Recent progress on catalytic pyrolysis of lignocellulosic biomass to high-grade bio-oil and bio-chemicals. Renew Sust Energ 70:945–967. https://doi.org/10.1016/j.rser.2016.12.001
Katenta J, Nakiguli C, Mukasa P, Ntambi E (2020) Removal of chromium (VI) from tannery effluent using bio-char of phoenix reclinata seeds. Green Sustain Chem 10:91–107. https://doi.org/10.4236/gsc.2020.103007
Kedia R, Sharma A (2015) Bioremediation of industrial effluents using arbuscular mycorrhizal fungi. Biosci Biotechol Res Asia 12:197–200. https://doi.org/10.13005/bbra/1651
Keiluweit M, Nico PS, Johnson MG, Kleber M (2010) Dynamic molecular structure of plant biomass-derived black carbon (biochar). Environ Sci Technol 44:1247–1253. https://doi.org/10.1021/es9031419
Kumar A, Bhattacharya T (2021) Biochar: a sustainable solution. Environ Dev Sustain 23:6642–6680. https://doi.org/10.1007/s10668-020-00970-0
Le PT, Bui HT, Le DN, Nguyen TH, Pham LA, Nguyen HN, Le TPQ (2021) Preparation and characterization of biochar derived from agricultural by-products for dye removal. Adsorp Sci Technol. https://doi.org/10.1155/2021/9161904
Li M, Liu Q, Guo L, Zhang Y, Lou Z, Wang Y, Qian G (2013a) Cu (II) removal from aqueous solution by Spartina alterniflora derived biochar. Bioresour Technol 141:83–88. https://doi.org/10.1016/j.biortech.2012.12.096
Li M, Liu Q, Guo L, Zhang Y, Lou Z, Wang Y, Qian G (2013b) Cu(II) removal from aqueous solution by Spartina alterniflora derived biochar. Bioresour Techonol 141:83–88. https://doi.org/10.1016/j.biortech.2012.12.096
Li Q, Zhao S, Wang Y (2021) Mechanism of oxytetracycline removal by coconut shell biochar loaded with nano-zero-valent iron. Int J Environ Res Public Health 18:13107. https://doi.org/10.3390/ijerph182413107
Li L, Jia Z, Ma H, Bao W, Li X, Tan H, Li Y (2019) The effect of two different biochars on remediation of Cd-contaminated soil and Cd uptake by Lolium perenne. Environ Geochem Health 41:2067–2080. https://doi.org/10.1007/s10653-019-00257-y
Lian G, Wang B, Lee X, Li L, Liu T, Lyu W (2019) Enhanced removal of hexavalent chromium by engineered biochar composite fabricated from phosphogypsum and distillers grains. Sci Total Environ 697:134119. https://doi.org/10.1016/j.scitotenv.2019.134119
Liang M, Ding Y, Zhang Q, Wang D, Li H, Lu L (2020) Removal of aqueous Cr (VI) by magnetic biochar derived from bagasse. Sci Rep 10:21473. https://doi.org/10.1038/s41598-020-78142-3
Memon JR, Memon SQ, Bhanger MI, El-Turki A, Hallam KR, Allen GC (2009) Banana peel: a green and economical sorbent for the selective removal of Cr (VI) from industrial wastewater. Colloids Surf B 70:232–237. https://doi.org/10.1016/j.colsurfb.2008.12.032
Mergbi M, Galloni MG, Aboagye D, Elimian E, Su P, Ikram BM, Nabgan W, Bedia J, Amor HB, Contreras S, Medina F (2023) Valorization of lignocellulosic biomass into sustainable materials for adsorption and photocatalytic applications in water and air remediation. Environ Sci Pollut Res 30:74544–74574. https://doi.org/10.1007/s11356-023-27484-2
Mohapatra RK, Parhi PK, Pandey S, Bindhani BK, Thatoi H, Panda CR (2019) Active and passive biosorption of Pb (II) using live and dead biomass of marine bacterium Bacillus xiamenensis PbRPSD202: kinetics and isotherm studies. J Environ Manage 247:121–134. https://doi.org/10.1016/j.jenvman.2019.06.073
Moussavi G, Barikbin B (2010) Biosorption of chromium (VI) from industrial wastewater onto pistachio hull waste biomass. J Chem Eng 162:893–900. https://doi.org/10.1016/j.cej.2010.06.032
Munoz AJ, Espinola F, Moya M, Ruiz E (2015) Biosorption of Pb (II) ions by Klebsiella sp. 3S1 isolated from a wastewater treatment plant: kinetics and mechanisms studies. Biomed Res Int 719060:1–15. https://doi.org/10.1155/2015/719060
Murad HA, Mahtab A, Bundschuh J, Hashimoto Y, Zhang M, Sarkar B, Ok YS (2022) A remediation approach to chromium-contaminated water and soil using engineered biochar derived from peanut shell. Environ Res 204:112125. https://doi.org/10.1016/j.envres.2021.112125
Naik BR, Suresh C, Kumar NS, Seshaiah K, Reddy AVR (2017) Biosorption of Pb (II) and Ni (II) ions by chemically modified Eclipta alba stem powder: kinetics and equilibrium studies. Sep Sci Technol 52:1717–1732. https://doi.org/10.1080/01496395.2017.1298614
Ning P, Yang G, Hu L, Sun J, Shi L, Zhou Y, Wang Z, Yang J (2021) Recent advances in the valorization of plant biomass. Biotechnol Biofuels 14:102. https://doi.org/10.1186/s13068-021-01949-3
Pan R, Bu J, Ren G, Zhang Z, Li K, Ding A (2022) Mechanism of removal of hexavalent chromium from aqueous solution by Fe-modified biochar and its application. Appl Sci 12:1238. https://doi.org/10.3390/app12031238
Peng Z, Liu X, Chen H, Liu Q, Tang J (2019) Characterization of ultraviolet-modified biochar from different feedstocks for enhanced removal of hexavalent chromium from water. Water Sci Technol 79:1705–1716. https://doi.org/10.2166/wst.2019.170
Primo JDO, Bittencourt C, Acosta S, Sierra-Castillo A, Colomer JF, Jaerger S, Teixeira VC, Anaissi FJ (2020) Synthesis of zinc oxide nanoparticles by ecofriendly routes: adsorbent for copper removal from wastewater. Front Chem 8:571790. https://doi.org/10.3389/fchem.2020.571790
Puglla EP, Guaya D, Tituana C, Osorio F, García-Ruiz MJ (2020) Biochar from agricultural by-products for the removal of lead and cadmium from drinking water. Water 12:2933. https://doi.org/10.3390/w12102933
Qasem NA, Mohammed RH, Lawal DU (2021) Removal of heavy metal ions from wastewater: A comprehensive and critical review. NPJ Clean Water 4:36. https://doi.org/10.1038/s41545-021-00127-0
Rajapaksha AU, Alam MS, Chen N, Alessi DS, Igalavithana AD, Tsang DCW, Ok YS (2018) Removal of hexavalent chromium in aqueous solutions using biochar: chemical and spectroscopic investigations. Sci Total Environ 625:1567–1573. https://doi.org/10.1016/j.scitotenv.2017.12.195
Rama CM (2016) Facile synthesis of zero valent iron mag netic biochar composites for Pb(II) removal from the aqueous medium. Alexandria Eng. J. 55:619–625. https://doi.org/10.1016/j.aej.2015.12.015
Ramrakhiani L, Majumder R, Khowala S (2011) Removal of hexavalent chromium by heat inactivated fungal biomass of Termitomyces clypeatus: surface characterization and mechanism of biosorption. J Chem Eng 171:1060–1068. https://doi.org/10.1016/j.cej.2011.05.002
Rangabhashiyam S, Balasubramanian P (2019) The potential of lignocellulosic biomass precursors for biochar production: performance, mechanism and wastewater application-a review. Ind Crops Prod 128:405–423. https://doi.org/10.1016/j.indcrop.2018.11.041
Rather RA, Singh S, Pal B (2017) Photocatalytic degradation of methylene blue by plasmonic metal-TiO2 nanocatalysts under visible light irradiation. J Nanosci Nanotechnol 17:1210–1216. https://doi.org/10.1166/jnn.2017.12658
Ren G, ** Y, Zhang C, Gu H, Qu J (2015) Characteristics of Bacillus sp. PZ-1 and its biosorption to Pb (II). Ecotoxicol 117:141–148. https://doi.org/10.1016/j.ecoenv.2015.03.033
Renu AM, Singh K (2017) Heavy metal removal from wastewater using various adsorbents: a review. J Water Reuse Desalin 7:387–419. https://doi.org/10.2166/wrd.2016.104
Ronsse F, Van Hecke S, Dickinson D, Prins W (2013) Production and characterization of slow pyrolysis biochar: influence of feedstock type and pyrolysis conditions. Gcb Bioenergy 5:104–115. https://doi.org/10.1111/gcbb.12018
Scharf B, Clement CC, Zolla V, Perino G, Yan B, Elci SG, Purdue E, Goldring S, Macaluso F, Cobelli N, Vachet RW, Santambrogio L (2014) Molecular analysis of chromium and cobalt-related toxicity. Sci Rep 4:1–12
Sekar M, Mathimani T, Alagumalai A, Chi NTL, Duc PA, Bhatia SK, Brindhadevi K, Pugazhendhi A (2021) A review on the pyrolysis of algal biomass for biochar and bio-oil–Bottlenecks and scope. Fuel 283:119190. https://doi.org/10.1016/j.fuel.2020.119190
Shibo D, Wei M, Yuzhen P, Fanqing M, Shuangen Y, Lei W (2017) Synthesis of magnetic biochar from iron sludge for the enhancement of Cr (VI) removal from solution. J Taiwan Inst Chem Eng 80:835–841. https://doi.org/10.1016/j.jtice.2017.07.002
Simón D, Palet C, Costas A, Cristóbal A (2022) Agro-industrial waste as potential heavy metal adsorbents and subsequent safe disposal of spent adsorbents. Water 14:3298. https://doi.org/10.3390/w14203298
Sinha S, Behera SS, Das S, Basu A, Mohapatra RK, Murmu BM, Parhi PK (2018) Removal of congo red dye from aqueous solution using amberlite IRA-400 in batch and fixed bed reactors. Chem Eng Commun 4:432–444. https://doi.org/10.1080/00986445.2017.1399366
Su W, Zhou L, Zhou Y (2003) Preparation of microporous activated carbon from coconut shells without activating agents. Carbon 41:861–863
Suliman W, Harsh JB, Abu-Lail NI, Fortuna AM, Dallmeyer I, Garcia-Perez M (2016) Influence of feedstock source and pyrolysis temperature on biochar bulk and surface properties. Biomass Bioenerg 84:37–48. https://doi.org/10.1016/j.biombioe.2015.11.010
Tan X, Liu Y, Zeng G, Wang X, Hu X, Gu Y, Yang Z (2015) Application of biochar for the removal of pollutants from aqueous solutions. Chemosphere 125:70–85. https://doi.org/10.1016/j.chemosphere.2014.12.058
Ullah I, Nadeem R, Iqbal M, Manzoor Q (2013) Biosorption of chromium onto native and immobilized sugarcane bagasse waste biomass. Ecol Eng 60:99–107. https://doi.org/10.1016/j.ecoleng.2013.07.028
Venkatraman Y, Arunkumar P, Kumar NS, Osman AI, Muthiah M, Al-Fatesh AS, Koduru JR (2023) Exploring modified rice straw biochar as a sustainable solution for simultaneous Cr (VI) and Pb (II) removal from wastewater: characterization, mechanism insights, and application feasibility. ACS Omega 8:38130–38147. https://doi.org/10.1021/acsomega.3c04271
Verbinnen B, Block C, Caneghem JV, Vandecasteele C (2015) Recycling of spent adsorbents for oxyanions and heavy metal ions in the production of ceramics. Waste Manag 45:407–411. https://doi.org/10.1016/j.wasman.2015.07.006
Verma R, Maji PK, Sarkar S (2021) Comprehensive investigation of the mechanism for Cr (VI) removal from contaminated water using coconut husk as a biosorbent. J Clean Prod 314:128117. https://doi.org/10.1016/j.jclepro.2021.128117
Vidhya L, Dhandapani M, Shanthi K, Kamala-Kannan S (2018) Removal of Cr (VI) from aqueous solution using coir pith biochar–An eco-friendly approach. Indian J Chem Technol 25:266–273
Volesky B (2007) Biosorption and me. Water Res 41:4017–4029. https://doi.org/10.1016/j.watres.2007.05.062
Wang XS, Li ZZ, Tao SR (2009) Removal of chromium (VI) from aqueous solution using walnut hull. J Environ Manage 90:721–729. https://doi.org/10.1016/j.jenvman.2008.01.011
Wang S, Gao B, Li Y, Mosa A, Zimmerman AR, Ma LQ, Migliaccio KW (2015) Manganese oxide-modified biochars: preparation, characterization, and sorption of arsenate and lead. Bioresour Technol 181:13–17. https://doi.org/10.1016/j.biortech.2015.01.044
Wang H, Zhang M, Lv Q (2019a) Removal efficiency and mechanism of Cr (VI) from aqueous solution by maize straw biochars derived at different pyrolysis temperatures. Water 11:781. https://doi.org/10.3390/w11040781
Wang T, Liu Y, Wang J, Wang X, Liu B, Wang Y (2019b) In-situ remediation of hexavalent chromium contaminated groundwater and saturated soil using stabilized iron sulfide nanoparticles. J Environ Manage 231:679–686. https://doi.org/10.1016/j.jenvman.2018.10.085
Xu Y, Bai T, Li Q, Yang H, Yan Y, Sarkar B, Lam SS, Bolan N (2021) Influence of pyrolysis temperature on the characteristics and lead (II) adsorption capacity of phosphorus-engineered poplar sawdust biochar. J Anal Appl Pyrolysis 154:105010. https://doi.org/10.1016/j.jaap.2020.105010
Yang W, Lu C, Liang B, Yin C, Lei G, Wang B, Zhou X, Zhen J, Quan S, **g Y (2023) Removal of Pb (II) from aqueous solution and adsorption kinetics of corn stalk biochar. Separations 10:438. https://doi.org/10.3390/separations10080438
Yang W, Lei G, Quan S, Zhang L, Wang B, Hu H, Li L, Ma H, Yin C, Feng F, **g Y (2022) The Removal of Cr(VI) from Aqueous Solutions with Corn Stalk Biochar. Int J Environ Res Public Health 19:14188. https://doi.org/10.3390/ijerph192114188
Zhang J, Lü F, Luo C, Shao L, He P (2014) Humification characterization of biochar and its potential as a composting amendment. J Environ Sci 26:390–397. https://doi.org/10.1016/S1001-0742(13)60421-0
Zhao J, Yang L, Li F, Yu R, ** C (2009) Structural evolution in the graphitization process of activated carbon by high-pressure sintering. Carbon 47:744–751. https://doi.org/10.1016/j.carbon.2008.11.006
Zhao N, Yin Z, Liu F, Zhang M, Lv Y, Hao Z, Pan G, Zhang J (2018) Environmentally persistent free radicals mediated removal of Cr(VI) from highly saline water by corn straw biochars. Bioresour Technol 260:294–301. https://doi.org/10.1016/j.biortech.2018.03.116
Zhao N, Zhao C, Tsang DC, Liu K, Zhu L, Zhang W, Zhang J, Tang Y, Qiu R (2021) Microscopic mechanism about the selective adsorption of Cr(VI) from salt solution on O-rich and N-rich biochars. J Hazard Mater 404:124162. https://doi.org/10.1016/j.jhazmat.2020.124162
Zinicovscaia I, Grozdov D, Yushin N, Abdusamadzoda D, Gundorina S, Rodlovskaya E, Kristavchuk O (2020) Metal removal from chromium containing synthetic effluents by Saccharomyces cerevisiae. Desalination 178:254–270. https://doi.org/10.5004/dwt.2020.24987
Zornoza R, Moreno-Barriga F, Acosta JA, Muñoz MA, Faz A (2016) Stability, nutrient availability and hydrophobicity of biochars derived from manure, crop residues, and municipal solid waste for their use as soil amendments. Chemosphere 144:122–130. https://doi.org/10.1016/j.chemosphere.2015.08.046
Acknowledgements
The authors would like to thankfully acknowledge the Director, CSIR-Institute of Minerals and Materials Technology, Bhubaneswar, India, for providing the necessary instrumental facilities and support to carry out this research work and publish the research findings.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Editorial responsibility: P.F. Rupani.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Acharya, C., Mohapatra, R.K., Sasmal, A. et al. Effective remediation of Cr(VI) using coconut coir-derived porous biochar: application of kinetics and isotherm approaches. Int. J. Environ. Sci. Technol. 21, 7249–7268 (2024). https://doi.org/10.1007/s13762-024-05460-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-024-05460-8