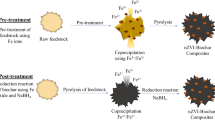
Abstract
The reductive removal of redox-reactive contaminants, including 2,4-dinitrotoluene, 4-chlorophenol, chromate, and selenate, using biochar-coated metals and the relationship between reduction kinetics and electrochemical properties of biochar-coated metals were examined. To investigate the role of electrochemical properties, one-electron-transfer electrochemical reduction and oxidation mediators were used to determine electron-accepting capacities (EACs) and electron-donating capacities (EDCs) of biochar, zero-valent iron [Fe(0)], FeS, zero-valent zinc [Zn(0)], biochar-coated Fe(0), biochar-coated FeS, and biochar-coated Zn(0). Kinetic analysis showed that biochar coating resulted in decreasing the reductive transformation of contaminants by biochar-coated metals, suggesting that the sorbed molecules were further reduced to reduction products. Biochar, metals, and biochar-coated metal EDCs were an order of magnitude higher than their EACs, ranging between 11.01 and 38.61 mmol e−/g. The linear relationship between the EDCs and kinetic rates revealed that electron transfer from the core metal to sorbed contaminant oxidation is responsible for the reductive removal. A significant and irreversible EDC decrease generates electrons for the reductive transformation. To our knowledge, it is the first attempt to examine the electrochemical properties of metal-biochar composites and their relationship with reduction kinetics of various contaminants. Our results indicate that biochar-coated metals can be used as a sorbent and reductant for redox-reactive contaminants in subsurface environments.






Similar content being viewed by others
References
Aeschbacher M, Sander M, Schwarzenbach RP 2010 Novel electrochemical approach to assess the redox properties of humic substances Environ Sci Technol 44 87 93
Aeschbacher M, Graf C, Schwarzenbach RP, Sander M 2012 Antioxidant properties of humic substances Environ Sci Technol 46 4916 4925
Devi P, Saroha AK 2014 Synthesis of the magnetic biochar composites for use as an adsorbent for the removal of pentachlorophenol from the effluent Bioresource Technol 169 525 531
Dolfing J, Eekert M Van, Seech A, Vogan J, Mueller J 2007 In situ chemical reduction (ISCR) technologies: significance of low Eh reactions Soil Sed Contam 17 63 74
Dong H, Deng J, **e Y, Zhang C, Jiang Z, Cheng Y, Hou K, Zeng G 2017 Stabilization of nanoscale zero-valent iron (nZVI) with modified biochar for Cr(VI) removal from aqueous solution J Hazard Mater 332 79 86
Gorski CA , Aeschbacher M, Soltermann D, Voegelin A, Baeyens B, Fernandes MM, Hofstetter TB, Sander M 2012 Redox properties of structural Fe in clay minerals. 1. Electrochemical quantification of electron-donating and –accepting capacities of smectites Environ Sci Technol 46 9360 9368
Gorski CA, Klüpfel L, Voegelin A, Sander M, Hofstetter TB, 2012 Redox properties of structural Fe in clay minerals. 2. Electrochemical and spectroscopic characterization of electron transfer irreversibility in ferruginous smectite, SWa-1 Environ Sci Technol 46 9369 9377
Huling SG, Pivetz BE (2006) In situ chemical oxidation (ISCO). EPA/600/R-06/072, United States Environmental Protection Agency, Cincinnati.
Klüpfel L, Keiluweit M, Kleber M, Sander M 2014 Redox properties of plant biomass-derived black carbon (biochar) Environ Sci Technol 48 5601 5611
Klüpfel L, Piepenbrock A, Kappler A, Sander M 2014 Humic substances as fully regenerable electron acceptors in recurrently anoxic environments Nature Geosci 7 195 200
Lehmann J, Joseph S 2015 Biochar for environmental management: science, technology and implementation 2 Routledge New York
Lv J, Han R, Huang Z, Luo L, Cao D, Zhang S 2018 Relationship between molecular components and reducing capacities of humic substances ACS Earth Space Chem 2 330 339
Oh SY, Seo YD 2014 Sorptive removal of nitro explosives and metals using biochar J Environ Qual 43 1663 1671
Oh SY, Seo YD, Ryu KS 2016 Reductive removal of 2,4-dinitrotoluene and 2,4-dichlorophenol with zero-valent iron-included biochar Bioresource Technol 216 1014 1021
Oh SY, Seo YD, Ryu KS, DY Park, Lee SH 2017 Redox and catalytic properties of biochar-coated zero-valent iron for the removal of nitro explosives and halogenated phenols Environ Sci Processes Impacts 19 711 719
Oh SY, Seo TC 2019 Upgrading biochar via co-pyrolyzation of agricultural biomass and polyethylene terephthalate wastes RSC Adv 9 28284 28290
Oh SY, Seo YD, Rajagopal R, Ryu KS 2021 Removal of chromate and selenate in natural water using iron-bearing mineral-biochar composites Environ Earth Sci 80 240
Prévoteau A, Ronsse F, Cid I, Boeckx P, Rabaey K 2016 The electron donating capacity of biochar is dramatically underestimated Sci Rep 6 32870
Seo YD, Oh SY, Rajagopal R, Ryu KS 2020 FeS-biochar and Zn(0)-biochar for remediation of redox-reactive contaminants RSC Adv 10 30203 30213
Su H, Fang Z, Tsang PE, Zheng L, Cheng W, Fang J, Zhao D 2016 Remediation of hexavalent chromium contaminated soil by biochar-supported zero-valent iron nanoparticles J Hazard Mater 318 533 540
Sun H, Zhou G, Liu S, Ang HM, Tadé MO, Wang S 2012 Nano-Fe0 encapsulated in microcarbon spheres: synthesis, characterization, and environmental applications ACS Appl Mater Interfaces 4 6235 6241
Sunkara B, Zhan J, He J, McPherson GL, Piringer G, John VT 2010 Nanoscale zerovalent iron supported on uniform carbon mesopheres for the in situ remediation of chlorinated hydrocarbons ACS Appl Mater Interfaces 2 2854 2862
Suthersan SS, Horst J, Schnobrich M, Welty N, McDonough J 2016 Remediation engineering: design concepts 2 CRC Press Boca Raton
Tseng HH, Su JG, Liang C 2011 Synthesis of granular activated carbon/zero valent iron composites for simultaneous adsorption/dechlorination of trichloroethylene J Hazard Mater 192 500 506
Wu X, Yang Q, Xu D, Zhong Y, Luo K, Li X, Chen H, Zeng G 2013 Simultaneous adsorption/reduction of bromate by nanoscale zerovalent iron supported on modified activated carbon Ind Eng Chem Res 52 12574 12581
Zhang C 2020 Soil and groundwater remediation: fundamentals, practices, and sustainability Wiley New York
Zheng X, Liu Y, Fu H, Qu X, Yan M, Zhang S, Zhu D 2019 Comparing electron donating/accepting capacities (EDC/EAC) between crop residue-derived dissolved black carbon and standard humic substances Sci Total Environ 673 29 35
Zhou Y, Gao B, Zimmerman AR, Chen H, Zhang M, Cao X 2014 Biochar-supported zero-valent iron for removal of various contaminants from aqueous solutions Bioresource Technol 152 538 542
Acknowledgments
This work was supported by a National Research Foundation (NRF) of Korea grant funded by the Korean government (MSIT) (2020R1A2C1010855). This work was also supported in part by a Korea Institute for Advanced of Technology (KIAT) grant funded by the Korea Government (MOTIE) (P0008421).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Additional information
Editorial responsibility: Samareh Mirkia.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Seo, Y.D., Oh, S.Y., Rajagopal, R. et al. Redox-reactive contaminant removal using biochar-coated metals: the role of electrochemical properties. Int. J. Environ. Sci. Technol. 19, 4209–4220 (2022). https://doi.org/10.1007/s13762-021-03452-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-021-03452-6