Abstract
We analyzed catalase's function, structure, and thermal stability in two basic amino acid-based deep eutectic solvents due to the importance of studying the enzymatic behavior in suitable non-aqueous media. We used various UV-visible spectroscopic methods, fluorescence, and circular dichroism (CD). Combining arginine with glycerol and lysine with glycerol was applied to form two distinct amino acid-based deep eutectic solvents.
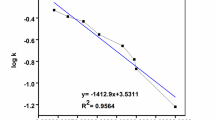
Kinetic studies showed that forming an intricate hydrogen bond network between glycerol and amino acid components in arginine or lysine-based aqueous deep eutectic solvents of 45% and 40%, respectively, keeps the catalase activity near 50%. Our outcomes demonstrated that the intrinsic fluorescence of the catalase is quenched by increasing the concentration of these solvents via a static mechanism. Also, CD results have further represented remarkable changes in arginine-glycerol and lysine-glycerol solvents with a diminished α-helical content and an increase in β-pleated sheets content. Studies of the thermal stability of catalase in the presence of these solvents showed that with increasing concentration of these solvents, the thermal stability of enzyme (Tm and ΔG° values) decreases.
Finally, the above data suggested that arginine and lysine with chaotropic factors of guanidine and ammonium in their functional group, respectively, act as influential factors in the activity and structural stability of the enzyme.








Similar content being viewed by others
References
A.R. Harifi-Mood, R. Ghobadi, A. Divsalar, Int. J. Biol. Macromol. 95, 115–120 (2017)
H. Vanda et al., Comptes Rendus Chim. 21, 628–638 (2018)
A. Paiva et al., ACS Sustain. Chem. Eng.. 2, 1063–1071 (2014)
A. Shishov et al., Microchem. J. 135, 33–38 (2017)
Y.P. Mbous et al., Biotechnol. Adv.. 35, 105–134 (2017)
S. Mouden et al., Phytochem. Rev. 16, 935–951 (2017)
B.B. Hansen et al., Chem. Rev. 121, 1232–1285 (2020)
M.S. Rahman et al., J. Mol. Liq. 321, 114745 (2021)
Y. Liu et al., J. Nat. Prod. 81, 679–690 (2018)
B. Koohshekan et al., J. Mol. Liq. 218, 8–15 (2016)
Vitolo, M., World J, Pharm. Pharm. Sci. 10, 47 (2021)
C. Glorieux, P.B. Calderon, Biol. Chem. 398, 1095–1108 (2017)
M. Galasso et al., Free Radic. Biol. Med. 172, 264–272 (2021)
O. Abazari, A. Divsalar, R. Ghobadi, J. Biomol. Struct. Dyn. 38, 609–615 (2020)
M.M. Goyal, A. Basak, Protein Cell 1, 888–897 (2010)
L. Góth, T. Nagy, Arch. Biochem. Biophys. 525, 195–200 (2012)
A. Goulas et al., Neurosci. Lett. 330, 210–212 (2002)
M. Asaduzzaman Khan et al., Chin. J. Cancer Res. 22, 87–92 (2010)
J. Kaushal et al., Biocatal. Agric. Biotechnol. 16, 192–199 (2018)
K. Czyzewska, A. Trusek, Pol. J. Chem. Technol. 20, 39–43 (2018)
R. Ghobadi, A. Divsalar, J. Mol. Liq. 310, 113207 (2020)
X. Tian, S. Zhang, L. Zheng, J. Microbiol. Biotechnol. 26, 80–88 (2016)
J.T. Gorke, F. Srienc, R.J. Kazlauskas, Chem. Commun. (2008). https://doi.org/10.1039/b716317g
Y. Huang et al., Food Chem. 221, 1400–1405 (2017)
M.J. Lopez, S.S. Mohiuddin, Biochemistry, Essential Amino Acids (StatPearls Publishing, Treasure Island (FL), 2022)
G. Wu, Amino Acids 37, 1–17 (2009)
Z. Yang, J. Biotechnol. 144, 12–22 (2009)
H. Ren et al., J. Clean. Prod. 193, 802–810 (2018)
H. Aebi, [13] Catalase in vitro, in Methods in enzymology. (Elsevier, Netherlands, 1984), pp.121–126
Y. Dai et al., Food Chem. 187, 14–19 (2015)
F. Gabriele et al., J. Mol. Liq. 291, 111301 (2019)
Y. **a et al., Int. J. Biol. Macromol. 40, 437–443 (2007)
K. Prakash et al., Protein Sci. 11, 46–57 (2002)
G. Savelli, N. Spreti, P. Di Profio, Current Opin. Colloid Interface Sci. 5, 111–117 (2000)
M.N. Jones, A. Finn, A. Mosavi-Movahedi, B.J. Waller, Biochim. Biophys. Acta (BBA) Protein Struct. Mol. Enzymol. 913(3), 395–398 (1987)
B. Srinivasan, Words Adv. Teach. Enzym. Kinet. 288, 2068–2083 (2021)
L. Lomba et al., Appl. Sci. 11, 10156 (2021)
E.M. Elnemma, Bull. Korean Chem. Soc. 25, 127–129 (2004)
A.A. Saboury, J. Iran. Chem. Soc. 6, 219–229 (2009)
A.M. Eberhardt et al., Appl. Catal. B Environ. 47, 153–163 (2004)
A. Cornish-Bowden, Perspect. Sci. 1, 121–125 (2014)
Y. Hu, L. Da, Spectrochim Acta Part A Mol Biomol Spectrosc. 121, 230–237 (2014)
H. L., S. R., Chem. Lett. 30, 844–845(2001)
S.M. Kelly, T.J. Jess, N.C. Price, Biochim. Biophys. Acta (BBA) Proteins Proteomics 1751, 119–139 (2005)
J. Lloyd, Nat. Phys. Sci. 231, 64–65 (1971)
F.M. Najjar et al., Int. J. Biol. Macromol. 95, 550–556 (2017)
A.A. Saboury, A.A. Moosavi-Movahedi, Biochem. EduC. 23, 164–167 (1995)
A. Moosavi-Movahedi, Pure Appl. Chem. 66, 71–75 (1994)
Acknowledgments
The Research Council of Kharazmi University has supported our research. We gratefully acknowledge their support.
Funding
Funding was provided by Kharazmi University, (Grant Number 1402).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Assar, E., Divsalar, A., Jafari, T. et al. Evaluation of the enzymatic activity of catalase in aqueous mediums of basic amino acids based deep eutectic solvents. J IRAN CHEM SOC 20, 2879–2888 (2023). https://doi.org/10.1007/s13738-023-02884-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-023-02884-w