Abstract
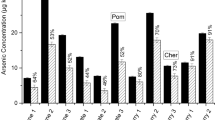
Arnicas Solidago microglossa DC. and Arnica montana L. are easily-accessible plants used as natural medicines by the Brazilian population. These products are susceptible to contamination by elemental impurities that are harmful to the human health. In 2017, the United States Pharmacopeia (USP) published two new chapters for elemental impurities determination. The classical methods were replaced by advanced analytical techniques such as optical emission spectrometry and inductively coupled plasma mass spectrometry (ICP OES and ICP-MS, respectively). However, these are expensive and difficult techniques to implement in small laboratories. Dispersive liquid–liquid microextraction (DLLME) contributes to improve the limit of quantification (LOQ), allowing the use of alternative analytical techniques to ICP such as capillary electrophoresis, atomic absorption and UV–Vis spectrophotometry. This work described the use a design of experiment (DoE) for the optimization of DLLME applied to the determination of metallic impurities in arnica infusions. The replacement of toxic solvents (i.e. chloroform and methanol) for a combination of organic carbonates and ionic liquids, proved to be a promising green alternative as solvent mixture in DLLME technique. The enrichment factor in infusion was between 89 and 127. The method was validated by evaluating the following parameters: selectivity, linearity, precision, accuracy, limit of detection (LOD) and LOQ. The defined range was 5.0–100.0 µg L−1 for cadmium and lead, and 1.5–30.0 µg L−1 for mercury, with accuracy varying from 94 to 107% and RSD from 1.77 to 8.51%. Therefore, DLLME employing green chemistry solvents has proved to be applicable for the analysis of elemental impurities in infusions.




Similar content being viewed by others
References
A.L. Baggio, A. Buskievicz, A. Fornari, T.A. Franke, P. Lucca, Pesquisa fitoquímica dos principais ativos presentes na droga vegetal Solidago microglossa, DC. (Arnica brasileira). Revista Thêma et Scientia 2(2), 149–153 (2012)
R. Thomas, Determining elemental impurities in pharmaceutical materials: how to choose the right technique. Spectroscopy 30(3), 30–42 (2015)
K.-L. Chen, S.-J. Jiang, Y.-L. Chen, Determining lead, cadmium and mercury in cosmetics using swee** via dynamic chelation by capillary electrophoresis. Anal. Bioanal. Chem. 409(9), 2461–2469 (2017)
J.M. Kokosa, Advances in solvent-microextraction techniques. Trends Anal. Chem. 43, 2–13 (2013)
N.S. la Colla, C.E. Domini, J.E. Marcovecchio, S.E. Botté, Latest approaches on green chemistry preconcentration methods for trace metal determination in seawater—a review. J. Environ. Manag. 151, 44–55 (2015)
M. Rezaee et al., Determination of organic compounds in water using dispersive liquid–liquid microextraction. J. Chromatogr. A 1116(1–2), 1–9 (2006)
F.Y. Meng, Y.Q. Wei, H. Lu, X.T. Zhou, J.X. Liu, G. Li, J.Y. Hou, Chelatometric dispersive liquid–liquid microextraction followed by capillary electrophoresis for the analysis of total and water soluble copper in Rhizoma coptidis. Chin. Chem. Lett. 24(6), 506–508 (2013)
M.J. Trujillo-Rodriguez, P. Rocio-Bautista, V. Pino, A.M. Afonso, Ionic liquids in dispersive liquid–liquid microextraction. Trends Anal. Chem. 51, 87–106 (2013)
V. Vičkačkaitė, A. Padarauskas, Ionic liquids in microextraction techniques. Cent. Eur. J. Chem. 10(3), 652–674 (2012)
K. Alfonsi, J. Colberg, P.J. Dunn, T. Fevig, S. Jennings, T.A. Johnson, H.P. Kleine, C. Knight, M.A. Nagy, D.A. Perry, Green chemistry tools to influence a medicinal chemistry and research chemistry based organisation. Green Chem. 10(1), 31–36 (2008). https://doi.org/10.1039/b711717e
R.K. Henderson, C. Jiménez-González, D.J.C. Constable, S.R. Alston, G.G.A. Inglis, G. Fisher, J. Sherwood, S.P. Binks, A.D. Curzons, Expanding GSK’s solvent selection guide-embedding sustainability into solvent selection starting at medicinal chemistry. Green Chem. 13(4), 854 (2011). https://doi.org/10.1039/c0gc00918k
D. Prat, O. Pardigon, H.-W. Flemming, S. Letestu, V. Ducandas, P. Isnard, E. Guntrum, T. Senac, S. Ruisseau, P. Cruciani, Sanofi’s solvent selection guide: a step toward more sustainable processes. Org. Process Res. Dev. 17(12), 1517–1525 (2013). https://doi.org/10.1021/op4002565
S. Mahpishanian, F. Shemirani, Preconcentration procedure using in situ solvent formation microextraction in the presence of ionic liquid for cadmium determination in saline samples by flame atomic absorption spectrometry. Talanta 82(2), 471–476 (2010)
B.R. Oliveira, C.C. Silva, J.C.P. Calado, W.L. Batista, F.A. Siqueira, L.S.L. Junior, Preparação de α-acetiloxi-n-cicloexilamidas através da reação de Passerini utilizando dimetilcarbonato como solvente ambientalmente amigável. Quim. Nova 41(1), 92–99 (2018)
B. Schäffner, F. Schäffner, S.P. Verevkin, A. Börner, Organic carbonates as solvents in synthesis and catalysis. Chem. Rev. 110(8), 4554–4581 (2010). https://doi.org/10.1021/cr900393d
M.L. de França, L. Separovic, L.S. Longo Jr., D.C. de Oliveira, F.R. Lourenço, L.A. Calixto, Determining uncertainty in a simple UV–Vis spectrometry method employing dimethyl carbonate as green solvent for lead determination in water. Measurement 173, 108581 (2021). https://doi.org/10.1016/j.measurement.2020.108581
M. Tobiszewski, W. Zabrocka, M. Bystrzanowska, Diethyl carbonate as a green extraction solvent for chlorophenol determination with dispersive liquid–liquid microextraction. Anal. Methods 11(6), 844–850 (2019). https://doi.org/10.1039/c8ay02683a
R. Coutinho, M.T.G. Vianna, M. Marques, Optimisation of the conditions of dispersive liquid–liquid microextraction for environmentally friendly determination of bisphenols and benzophenone in complex water matrices by LC-MS/MS. Microchem. J. 180, 107636 (2022). https://doi.org/10.1016/j.microc.2022.107636
H.T. Elbalkiny, A.M. Yehia, Artificial networks for spectral resolution of antibiotic residues in bovine milk; solidification of floating organic droplet in dispersive liquid–liquid microextraction for sample treatment. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 266, 120449 (2022). https://doi.org/10.1016/j.saa.2021.120449
A.Ö. Altunay, A. Elik, A novel sonication assisted dispersive liquid–liquid microextraction method for methylparaben in cosmetic samples using deep eutectic solvent. Sustain. Chem. Pharm. 9, 100781 (2022). https://doi.org/10.1016/j.scp.2022.100781
X. **g, H. Xue, X. Sang, X. Wang, L. Jia, Magnetic deep eutectic solvent-based dispersive liquid–liquid microextraction for enantioselectively determining chiral mefentrifluconazole in cereal samples via ultra-high-performance liquid chromatography. Food Chem. 391, 133220 (2022). https://doi.org/10.1016/j.foodchem.2022.133220
L. Mousavi, Z. Tamiji, M.R. Khoshayand, Applications and opportunities of experimental design for the dispersive liquid-liquid microextraction method—a review. Talanta 190, 335–356 (2018). https://doi.org/10.1016/j.talanta.2018.08.002
M. Shamsipur, B. Hemmateenejad, N.J. Jahani, K.M. Maleki, Liquid chromatographic-mass spectrometric monitoring of photodegradation of diphenylamine using experimental design methodology. J. Photochem. Photobiol. A: Chem. 299, 210–217 (2015). https://doi.org/10.1016/j.jphotochem.2014.12.002
ANVISA. Agencia Nacional de Vigilância Sanitária, Formulário de Fitoterápicos da Farmacopeia Brasileira, 1ª Ed. Brasilia (2018)
ANVISA. Agencia Nacional de Vigilância Sanitária, Guia para tratamento estatístico da validação analítica 1ª versão. Brasilia (2017)
G. Chatel, C. Goux-Henry, A. Mirabaud, T. Rossi, N. Kardos, B. Andrioletti, M. Draye, H2O2/NaHCO3-mediated enantioselective epoxidation of olefins in NTf2-based ionic liquids and under ultrasound. J. Catal. 291, 127–132 (2012). https://doi.org/10.1016/j.jcat.2012.04.016
K. Goossens, K. Lava, P. Nockemann, K. Van|Hecke, L. Van|Meervelt, K. Driesen, C. Görller-Walrand, K. Binnemans, T. Cardinaels, Pyrrolidinium ionic liquid crystals. Chem. A Eur. J. 15(3), 656–674 (2009). https://doi.org/10.1002/chem.200801566
L.S. Longo, E.F. Smith, P. Licence, Study of the stability of 1-Alkyl-3-methylimidazolium Hexafluoroantimonate(V) based ionic liquids using x-ray photoelectron spectroscopy. ACS Sustain. Chem. Eng. 4(11), 5953–5962 (2016). https://doi.org/10.1021/acssuschemeng.6b00919
M. Potangale, A. Das, S. Kapoor, S. Tiwari, Effect of anion and alkyl chain length on the structure and interactions of N-alkyl pyridinium ionic liquids. J. Mol. Liq. 240, 694–707 (2017). https://doi.org/10.1016/j.molliq.2017.05.036
I.M. Fukuda et al., Design of experiments (DoE) applied to pharmaceutical and analytical quality by design (QbD). Braz. J. Pharm. Sci. 54, 1–16 (2018)
ANVISA. Agencia Nacional de Vigilância Sanitária, Farmacopeia Brasileira, 6ª Ed. Brasilia (2019)
ANVISA. Agencia Nacional de Vigilância Sanitária, RDC Nº 166, DE 24 DE JULHO DE 2017. Dispõe sobre a validação de métodos analíticose dá outras providências. Agência Nacional de Vigilância Sanitária. D.O.U. Poder Executivo, Brasília (2017)
USP (US Pharmacopeial Convention). <232> Elemental Impurities – Limits; <233> Elemental Impurities – Procedures; <2332> Elemental Contaminants in Dietary Supplements. USP 43 NF-37 (2020)
AOAC Official Methods of Analysis, Guidelines for standard method performance requirements (2016)
ICH Q3D (R1), International conference on harmonisation of technical requirements for registration of pharmaceuticals for human use—guideline for elemental impurities (2019)
A.R.M. de Oliveira, I.R.D.O.S.S. Magalhães, F.J.M. de Santana, P.S. Bonato, Microextração em fase líquida (LPME): fundamentos da técnica e aplicações na análise de fármacos em fluidos biológicos. Quim. Nova 31(3), 637–644 (2008). https://doi.org/10.1590/s0100-40422008000300031
A. Asghari, M. Ghazaghi, M. Rajabi, M. Behzad, M. Ghaedi, Ionic liquid-based dispersive liquid–liquid microextraction combined with high performance liquid chromatography-UV detection for simultaneous preconcentration and determination of Ni Co, Cu and Zn in water samples. J. Serb. Chem. Soc. 79(1), 63–76 (2014)
P. Berton, R.G. Wuilloud, An online ionic liquid-based microextraction system coupled to electrothermal atomic absorption spectrometry for cobalt determination in environmental samples and pharmaceutical formulations. Anal. Methods 3(3), 664 (2011)
S. Li, S. Cai, W. Hu, H. Chen, H. Liu, Ionic liquid-based ultrasound-assisted dispersive liquid–liquid microextraction combined with electrothermal atomic absorption spectrometry for a sensitive determination of cadmium in water samples. Spectrochim. Acta Part B At. Spectrosc. 64(7), 666–671 (2009)
P. Berton, R.G. Wuilloud, Highly selective ionic liquid-based microextraction method for sensitive trace cobalt determination in environmental and biological samples. Anal. Chim. Acta 662(2), 155–162 (2010)
M. Baghdadi, F. Shemirani, In situ solvent formation microextraction based on ionic liquids: a novel sample preparation technique for determination of inorganic species in saline solutions. Anal. Chim. Acta 634(2), 186–191 (2009)
M.L. Martins et al., Microextração Líquido-Líquido Dispersiva (DLLME): Fundamentos e aplicações. Scientia Chromatographica 4(1), 38 (2012)
Acknowledgements
The authors are grateful to the grant #2016/25256-6, São Paulo Research Foundation (FAPESP).
Author information
Authors and Affiliations
Contributions
GBG: Writing—Original Draft, Conceptualization, Investigation, Validation, Formal analysis KFA: Validation LSLJ: Resources MAA: Methodology, Resources BLB: Methodology, Resources FRL: Writing—Review and Editing, Formal analysis, Data Curation LAC: Funding acquisition, Supervision, Writing—Review and Editing, Conceptualization.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Grecco, G.B., Albini, K.F., Longo, L.S. et al. Application of experimental design for dispersive liquid–liquid microextraction optimization for metallic impurities determination in arnica infusion employing green solvents. J IRAN CHEM SOC 20, 371–380 (2023). https://doi.org/10.1007/s13738-022-02674-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-022-02674-w