Abstract
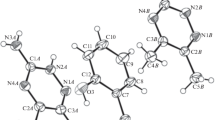
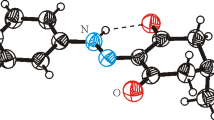
The title compound, C10H10FNO3, also known as [4-((4-fluorophenyl)amino)-4-oxobutanoic acid] (BFAOB), is synthesized efficiently, and its structure is confirmed by the SC-XRD technique, which indicates that two crystallographically different molecules are present in the asymmetric unit. The fluorobenzene ring is oriented at a dihedral angle of 76.09 (9)° and 23.7 (2)° with respect to acetamide moiety in the first and second types of molecule, respectively. A molecular overlay plot is employed to explore the difference between two types of molecules. Strong hydrogen bonding of type N–H…O, O–H…O and comparatively weak hydrogen bonding of type C-H…O stabilize the crystal packing. The titled compound is also characterized by powder XRD. Hirshfeld surface inspection is carried out to explore the non-covalent interactions that are responsible for crystal packing. DFT calculations demonstrate the high stability of BFAOB crystal compound.











Similar content being viewed by others
References
A.G. Hadi, K. Jawad, D.S. Ahmed, E. Yousif, Syst. Rev. Pharm. 10, 26–31 (2019)
S. Shoaib Ahmad Shah, M. Ashfaq, A. Waseem, M. MehboobAhmed, T. Najam, S. Shaheen, G. Rivera, Mini-Rev. Med. Chem. 15, 406–426 (2015)
X.-X. **e, Y.-C. Yang, B.-H. Dou, Z.-F. Li, G. Li, Coord. Chem. Rev. 403, 213100 (2020)
T. Hidalgo, M. Alonso-Nocelo, B. Bouzo, S. Reimondez-Troitiño, C. Abuin-Redondo, M. de la Fuente, P. Horcajada, Nanoscale 12, 4839–4845 (2020)
B. Godlewska-Żyłkiewicz, R. Świsłocka, M. Kalinowska, A. Golonko, G. Świderski, Ż Arciszewska, E. Nalewajko-Sieliwoniuk, M. Naumowicz, W. Lewandowski, Mater. 13, 4454 (2020)
S.S. Panda, S.C. Jain, Bioorg. Med. Chem. Lett. 23, 3225–3229 (2013)
C. Magnani, V.L.B. Isaac, M.A. Correa, H.R.N. Salgado, Anal. Methods 6, 3203 (2014)
R. Moore, S. Derry, P. Wiffen, S. Straube, D. Aldington, Eur. J. Pain 19, 1213–1223 (2015)
C.L. Ventola, Pharm. Ther. 40, 277 (2015)
I.A. Rather, B.-C. Kim, V.K. Bajpai, Y.-H. Park, Saudi. J. Biol. Sci. 24, 808 (2017)
C.B. Aakeröy, S. Forbes, J. Desper, J. Am. Chem. Soc. 131, 17048–17049 (2009)
N.J. Babu, A. Nangia, Cryst. Growth Des. 11, 2662–2679 (2011)
M.R. Chierotti, K. Gaglioti, R. Gobetto, D. Braga, F. Grepioni, L. Maini, CrystEngComm 15, 7598–7605 (2013)
A. Dalpiaz, B. Pavan, V. Ferretti, Drug Discov. Today 22, 1134–1138 (2017)
A.O. Surov, A.P. Voronin, N.A. Vasilev, A.B. Ilyukhin, G.L. Perlovich, New J. Chem. 45, 3034–3047 (2021)
M.N. Tahir, M. Ashfaq, F. Alexander, J. Caballero, E.W. Hernández-Rodríguez, A. Ali, J. Mol. Struct. 1193, 185–194 (2019)
M. Ashfaq, M.N. Tahir, A. Kuznetsov, S.H. Mirza, M. Khalid, A. Ali, J. Mol. Struct. 1199, 127041 (2020)
A. Ali, M. Khalid, M.F.U. Rehman, S. Haq, A. Ali, M.N. Tahir, M. Ashfaq, F. Rasool, A.A.C. Braga, ACS Omega 5, 15115–15128 (2020)
G.M. Sheldrick, Acta Crystallogr. A 64, 112 (2008)
A.L. Spek, Acta Crystallogr. Sect. D: Biol. Crystallogr. 65, 148–155 (2009)
F. Javed, S. Ali, S. Shahzadi, S. Sharma, K. Qanungo, K. Munawar, I. Khan, Russ. J. Gen. Chem. 87, 2409–2420 (2017)
S.K. Wolff, D.J. Grimwood, J.J. McKinnon, M.J. Turner, D. Jayatilaka, and M.A. Spackman, (2012).
M.J. Frisch, G.W. Trucks, H.B. Schlegel, D.J. Fox, Gaussian, Inc., Wallingford CT, 2016.
R. Dennington, T.A. Keith, J.M. Millam, Semichem Inc., Shawnee Mission, KS, 2016.
T. Lu, F. Chen, J. Comput. Chem. 33, 580–592 (2012)
M.A. Spackman, D. Jayatilaka, Cryst. Eng. Comm. 11, 19–32 (2009)
M. Ashfaq, G. Bogdanov, V. Glebov, A. Ali, M.N. Tahir, S. Abdullah, J. Mol. Struct. 1224, 129309 (2021)
H. Kargar, R. Behjatmanesh-Ardakani, M. Fallah-Mehrjardi, V. Torabi, K.S. Munawar, M. Ashfaq, M.N. Tahir, J. Mol. Struct. 1233, 130105 (2021)
H. Kargar, M. Fallah-Mehrjardi, R. Behjatmanesh-Ardakani, V. Torabi, K.S. Munawar, M. Ashfaq, M.N. Tahir, J. Mol. Struct. 1243, 130782 (2021)
J.J. McKinnon, M.A. Spackman, A.S. Mitchell, Acta Crystallogr. B 60, 627–668 (2004)
M. Ashfaq, K.S. Munawar, M.N. Tahir, N. Dege, M. Yaman, S. Muhammad, S.S. Alarfaji, H. Kargar, M.U. Arshad, ACS Omega 6(34), 22357 (2021)
A. Ali, M. Khalid, S. Abid, M.N. Tahir, J. Iqbal, M. Ashfaq, F. Kanwal, C. Lu, Cryst. 10, 778 (2020)
M. Ashfaq, G. Bogdanov, A. Ali, M.N. Tahir, S. Abdullah, J. Mol. Struct. 1235, 130215 (2021)
M. Khalid, A. Ali, S. Haq, M.N. Tahir, J. Iqbal, A.A. Braga, M. Ashfaq, S.U.H. Akhtar, J. Mol. Struct. 1224, 129308 (2021)
A. Ali, A. Kuznetsov, M.U. Khan, M.N. Tahir, M. Ashfaq, A.R. Raza, S. Muhammad, J. Mol. Struct. 1230, 129893 (2021)
A. Ali, M. Khalid, M.N. Tahir, M. Imran, M. Ashfaq, R. Hussain, M.A. Assiri, I. Khan, ACS Omega 6(10), 7047–7057 (2021)
H. Kargar, M. Fallah-Mehrjardi, R. Behjatmanesh-Ardakani, K.S. Munawar, M. Ashfaq, M.N. Tahir, Polyhedron. 115428, (2021).
H. Kargar, M. Fallah-Mehrjardi, R. Behjatmanesh-Ardakani, K.S. Munawar, M. Ashfaq, M.N. Tahir, Polyhedron. 115428 (2021).\
M. Turner, J.J. Mckinnon, D. Jayatilaka, M. Spackman, CrystEngComm 13, 1804–1813 (2011)
H. Kargar, M. Fallah-Mehrjardi, R. Behjatmanesh-Ardakani, K.S. Munawar, M. Ashfaq, M.N. Tahir, J. Mol. Struct. 1241, 130653 (2021)
H. Kargar, M. Fallah-Mehrjardi, R. Behjatmanesh-Ardakani, K.S. Munawar, M. Ashfaq, M.N. Tahir, Inorganica Chim. Acta. 120568 (2021).
Acknowledgements
M. M. A. and MI express appreciation to the Deanship of Scientific Research at King Khalid University, Saudi Arabia for funding through the research groups program under grant number R.G.P. 2/109/42.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ashfaq, M., Munawar, K.S., Bogdanov, G. et al. Single crystal inspection, Hirshfeld surface investigation and DFT study of a novel derivative of 4-fluoroaniline: 4-((4-fluorophenyl)amino)-4-oxobutanoic acid (BFAOB). J IRAN CHEM SOC 19, 1953–1961 (2022). https://doi.org/10.1007/s13738-021-02432-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-021-02432-4