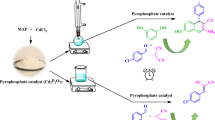
Abstract
A simple and efficient one-pot synthesis of tetrahydrobenzo[b]pyrans and dihydropyrano[4,3-b]pyrans derivatives was described. These reactions were realized through a three-component condensation of aromatic aldehyde, malononitrile, and 5,5-dimethyl-1,3-cyclohexanedione or 4-hydroxy-6-methylpyran-2-one using phosphate fertilizers monoammonium phosphate, NH4(H2PO4)2, (MAP), diammonium phosphate, (NH4)2(HPO4), (DAP), and triple superphosphate, Ca(H2PO4)2.H2O, (TSP) as heterogeneous catalysts. Under optimal reaction conditions, these fertilizers show an excellent catalytic activity. Indeed, the tetrahydrobenzo[b]pyrans and dihydropyrano[4,3-b]pyrans derivatives were obtained with good yields in short reaction times. Also, these fertilizers can be reused at least four times without significant loss of their catalytic activity. Therefore, their application can be contributing to the development of green chemistry.









Similar content being viewed by others
References
D. Rodríguez-Padrón, A.R. Puente-Santiago, A.M. Balu, M.J. Muñoz-Batista, R. Luque, Chem. Cat. Chem. 11, 18 (2019)
S. Bhaskaruni, S. Maddila, W. van Zyl, S. Jonnalagadda, Molecules 23, 1648 (2018)
V.S. Konkala, P.K. Dubey, Chin. Chem. Lett. 28, 1571 (2017)
S.A. Jehbez, H.R. Safaei, J. Iran. Chem. Soc. 15, 1041 (2018)
G.H. Sayed, M.E. Azab, K.E. Anwer, J. Heterocycl. Chem. 56, 2121 (2019)
P. Nazari, A. Bazi, S.A. Ayatollahi, H. Dolati, S.M. Mahdavi, L. Rafighdoost, M. Amirmostofian, Iran. J. Pharm. Res. 16, 943 (2017)
D. Kumar, P. Sharma, H. Singh, K. Nepali, G.K. Gupta, S.K. Jain, F. Ntie-Kang, RSC Adv. 7, 36977 (2017)
Z. Tashrifi, M. Mohammadi-Khanaposhtani, H. Hamedifar, B. Larijani, S. Ansari, M. Mahdavi, Mol. Divers. 1, 47 (2019)
M. Amirnejad, M.R. Naimi-Jamal, H. Tourani, H. Ghafuri, Monatsh Chem. 144, 1219 (2013)
S. Rostamnia, A. Morsali, Inorganica Chim. Acta 411, 113 (2014)
V.M. Joshi, R.L. Magar, P.B. Throat, S.U. Tekale, B.R. Patil, M.P. Kale, R.P. Pawar, Chin. Chem. Lett. 25, 455 (2014)
S. Balalaie, M. Bararjanian, M. Sheikh-Ahmadi, S. Hekmat, P. Salehi, Synth. Commun. 37, 1097 (2007)
B. Maleki, N. Nasiri, R. Tayebee, A. Khojastehnezhad, H.A. Akhlaghi, RSC Adv. 6, 79128 (2016)
S.F. Hojati, A. Amiri, N. MoeiniEghbali, S. Mohamadi, Appl Organometal Chem 32, e4235 (2018)
H. Naeimi, M.F. Zarabi, Res. Chem. Intermed. 44, 3227 (2018)
A.V. Borhade, J.A. Agashe, D.R. Tope, Russ. J. Appl. Chem. 90, 1005 (2017)
H.R. Saadati-Moshtaghin, F.M. Zonoz, Mater. Chem. Phys. 199, 159 (2017)
E. Stoyanov, I. Ivanov, D. Heber, Mol. 5, 19 (2000)
N.G. Khaligh, Chin. Chem. Lett. 26, 26 (2015)
M.A. Zolfigol, M. Yarie, S. Baghery, A. Khoshnood, D.A. Alonso, Res. Chem. Intermed. 43, 3291 (2017)
M. Abaszadeh, M. Seifi, Acta Chem. Iasi 25, 38 (2017)
N.G. Khaligh, S.B.A. Hamid, S.J.J. Titinchi, Polycycl. Aromat. Comp. 37, 31 (2017)
A. Zarei, M. Yarie, M.A. Zolfigol, K. Niknam, J. Chin. Chem. Soc. 67, 1120 (2019)
A.R. Kiasat, S. Hamid, S.J. Saghanezhad, Nanochem. Res. 1, 157 (2016)
N.D. Vala, H.H. Jardosh, M.P. Patel, Chinese Chem. Lett. 27, 168 (2016)
I. Bahammou, A. Esaady, S. Boukhris, R. Ghailane, N. Habbadi, A. Hassikou, A. Souizi, Med. J. Chem. 5, 61 (2016)
S. Chehab, Y. Merroun, T. Ghailane, R. Ghailane, S. Boukhris, A. Souizi, J. Turk. Chem. Soc. Sect A Chem. 5, 355 (2018)
Y. Merroun, S. Chehab, T. Ghailane, S. Boukhris, R. Ghailane, N. Habbadi, A. Hassikou, B. Lakhrissi, A. Souizi, J. Turk. Chem. Soc. Sect A Chem. 5, 303 (2018)
S. Chehab, Y. Merroun, T. Ghailane, R. Ghailane, S. Boukhris, M. Akhazzane, A. Kerbal, A. Souizi, Russ. J. Org. Chem. 55, 1380 (2019)
S. Chehab, Y. Merroun, T. Ghailane, N. Habbadi, S. Boukhris, A. Hassikou, R. Ghailane, M. Akhazzane, A. Kerbal, A. Daich, A. Souizi, Med. J. Chem. 7, 56 (2018)
Y. Merroun, S. Chehab, T. Ghailane, M. Akhazzane, A. Souizi, R. Ghailane, React. Kinet. Mech. Catal. 126, 249 (2019)
A. El Hallaoui, S. Chehab, B. Malek, O. Zimou, T. Ghailane, S. Boukhris, A. Souizi, R. Ghailane, ChemistrySelect 4, 3062 (2019)
A. El Hallaoui, S. Chehab, T. Ghailane, B. Malek, O. Zimou, S. Boukhriss, A. Souizi, R. Ghailane, Polycycl. Aromat. Compd. 40, 1 (2020)
Office chérifien des phosphates” to be found under https://www.ocpgroup.ma/fr/nos-produits/les-engrais, 2020
M. Matteini, S. Rescic, F. Fratini, G. Botticelli, Int. J. Archit. Heritage. 5, 717 (2011)
S. Balalaie, S. Nikoo, S. Haddadi, Synth. Commun. 38, 2521 (2008)
P. Salehi, M. Dabiri, A.R. Khosropour, P. Roozbehniya, J Iran Chem Soc. 3, 98 (2006)
M.G. Dekamin, M. Eslami, A. Maleki, Tetrahedron 69, 1074 (2013)
N. Hazeri, M.T. Maghsoodlou, F. Mir, M. Kangani, H. Saravani, E. Molashahi, Chin. J. Catal. 35, 391 (2014)
D. Rajguru, B.S. Keshwal, S. Jain, Chin. Chem. Lett. 24, 1033 (2013)
R. Mozafari, F. Heidarizadeh, Polyhedron 162, 263 (2019)
H.F. Niya, N. Hazeri, M.R. Kahkhaie, M.T. Maghsoodlou, Res. Chem. Intermed. 46, 1685 (2020)
U.V. Mane, M.S. Chavan, B.R. Choudhari, D.V. Mane, J Pharm Chem Biol Sci. 6, 311 (2019)
M. Hajjami, F. Gholamian, R.H.E. Hudson, A.M. Sanati, Catal. Lett. 149, 228 (2019)
S.S. Pourpanah, S.M. Habibi-Khorassani, M. Shahraki, Chin. J. Catal. 36, 757 (2015)
A.A. Mohammadi, M.R. Asghariganjeh, A. Hadadzahmatkesh, Arab. J. Chem. 10, S2213 (2017)
D. Azarifar, Y. Abbasi, Synth. Commun. 46, 745 (2016)
S.M. Khatat, B. Sadeghi, M. Ghasemi, Inorg Nano-Met Chem. 49, 56 (2019)
X. Fan, D. Feng, Y. Qu, X. Zhang, J. Wang, P.M. Loiseau, G. Andrei, R. Snoeck, E.D. Clercq, Bioorg. Med. Chem. Lett. 20, 809 (2010)
G. Brahmachari, B. Banerjee, A.C.S. Sustain, Chem. Eng. 2, 411 (2014)
N. G. Khaligh, T. Mihankhah, M. R. Johan, Org. Prep. Proced. Int. 1945 (2020)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chehab, S., Merroun, Y., Ghailane, T. et al. A facile and efficient synthesis of tetrahydrobenzo[b]pyrans and dihydropyrano[4,3-b]pyrans derivatives using phosphate fertilizers MAP, DAP, and TSP as heterogeneous catalysts. J IRAN CHEM SOC 18, 2665–2678 (2021). https://doi.org/10.1007/s13738-021-02223-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-021-02223-x