Abstract
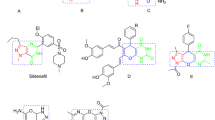
Ag–TiO2 nanocomposite as an efficient catalyst was synthesized and characterized by AFM analysis. This nanocomposite acts as a heterogeneous and recyclable catalyst for the room temperature synthesis of 1,2,4,5-tetrasubstituted imidazoles from aldehydes, benzil, ammonium acetate and several anilines or aliphatic amines via one-pot four-component condensation reaction in H2O as a green solvent. Ag–TiO2 nanocomposite was simply recovered by filtration and was reused up to three times.



Similar content being viewed by others
References
C. Lucarelli, A. Vaccari, Green Chem. 13, 1941 (2011)
B.C. Gates, Faraday Discuss. 188, 591 (2016)
P. Lanzafame, S. Perathoner, G. Centi, S. Gross, E.J.M. Hensen, Catal. Sci. Technol. 7, 5182 (2017)
M. Bowker, Catal. Lett. 142, 1411 (2012)
J. Akbari, A. Heydari, H.R. Kalhor, S.A. Kohan, J. Comb. Chem. 12, 137 (2010)
P. Wasserscheid, T. Welton, Ionic Liquid in Synthesis (Wiley-VCH, Weinheim, 2007).
K. De, P. Bhanja, A. Bhaumik, C. Mukhopadhyay, ChemCatChem 10, 590 (2018)
L. Yin, J. Liebscher, Chem. Rev. 107, 133 (2007)
S. Shylesh, V. Schnemann, W.R. Thiel, Angew. Chem. 122, 3504 (2010)
M. Lamblin, L. Nassar-Hardy, J.C. Hierso, E. Fouquet, F.X. Felpin, Adv. Synth. Catal. 352, 33 (2010)
D. Astruc, E. Boisselier, C. Ornelas, Chem. Rev. 110, 1857 (2010)
P. Das, A. Dutta, A. Bhaumik, C. Mukhopadhyay, Green Chem. 16, 1426 (2014)
A.V. Biradar, S.B. Umbarkar, M.K. Dongare, Appl. Catal. A 285, 190 (2005)
S.J. Singh, R.V. Jayaram, Tetrahedron Lett. 49, 4249 (2008)
M.B. Gawande, R.V. Jayaram, Catal. Commun. 7, 931 (2006)
E. Rafiee, S. Eavania, Green Chem. 13, 2116 (2011)
S. Brauch, S.S. van Berkel, B. Westermann, Chem. Soc. Rev. 42, 4948 (2013)
R.C. Cioc, E. Ruijter, R. Orru, Green Chem. 16, 2958 (2014)
S. Sharma, R.A. Maurya, K.-I. Min, G.-Y. Jeong, D.-P. Kim, Angew. Chem. Int. Ed. Engl. 52, 7564 (2013)
S. Shaabani, A. Doemling, Angew. Chem. Int. Ed. 57, 16266 (2018)
M. Shen, T.G. Driver, Org. Lett. 10, 3367 (2008)
K. Bahrami, M.M. Khodaei, F. Naali, J. Org. Chem. 73, 6835 (2008)
R. Rossi, G. Angelici, G. Casotti, C. Manzini, M. Lessi, Adv. Synth. Catal. 361, 2737 (2019)
M.R. Grimmett, in Comprehensive Heterocyclic Chemistry II, ed. by A.R. Katritsky, E.F.V. Scriven (Pergamon, Oxford, 1996), pp. 77–220
M. Misono, Chem. Commun. 13, 1141 (2001)
J.W. Black, G.J. Durant, J.C. Emmett, C.R. Ganellin, Nature 248, 65 (1974)
U. Ucucu, N.G. Karaburun, I. Iskdag, Farmaco 56, 285 (2001)
M. Shekouhy, A. Moaddeli, A. KhalafiNezhad, J. Porous Mater. 25, 75 (2018)
M.G. Dekamin, E. Arefi, A. Yaghoubi, RSC Adv. 6, 86982 (2016)
R. Bansal, P.K. Soni, A.K. Halve, J. Heterocyclic Chem. 55, 1308 (2018)
M.A. Zolfigol, S. Baghery, A.R. Moosavi-Zare, S.M. Vahdat, RSC Adv. 5, 32933 (2015)
Z. Zarnegar, J. Safari, New J. Chem. 38, 4555 (2014)
B. Maleki, H. Eshghi, A. Khojastehnezhad, R. Tayebee, S. SedighAshrafi, G. EsmailianKahoo, F. Moeinpour, RSC Adv. 5, 64850 (2015)
A.R. Karimi, Z. Alimohammadi, M.M. Amini, Mol. Divers. 14, 635 (2010)
M. Hajjami, F. Ghorbani, Z. Yousofvand, Appl. Organometal. Chem. 31, e3843 (2017)
Z. Taherinia, A. Ghorbani-Choghamarani, M. Hajjami, Catal. Lett. 149, 151 (2019)
M. Hajjami, A. Ghorbani-Choghamarani, Z. Yousofvand, M. Norouzi, J. Chem. Sci. 127, 1221 (2015)
M.A. Zolfigol, S. Baghery, A.R. Moosavi-Zare, S.M. Vahdat, J. Mol. Catal. A: Chem. 409, 216 (2015)
B. Maleki, M. Baghayeri, S.M. Vahdat, A. Mohammadzadeh, S. Akhoondi, RSC Adv. 5, 46545 (2015)
S. Asghari, S. Ramezani, M. Ahmadipour, M. Hatami, DEC MONOMERS POLYM. 16(4), 349 (2013)
S.M. Vahdat, S. GhafouriRaz, S. Baghery, J. Chem. Sci. 126, 579 (2014)
S.M. Vahdat, F. Chekin, M. Hatami, M. Khavarpour, S. Baghery, Chin. J. Catal. 34, 758 (2013)
F. Chekin, S.M. Vahdat, M.J. Asadi, Russian. J. Appl. Chem. 89, 816 (2016)
S. Yazdani, M. Hatami, S.M. Vahdat, Turk J. Chem. 38, 388 (2014)
S.M. Vahdat, M. Khavarpour, F. Mohanazadeh, J. Appl. Chem. 9, 41 (2015)
H.R. Shaterian, M. Ranjbar, K. Azizi, J. Iran. Chem. Soc. 8, 1120 (2011)
M.A. Zolfigol, A. Khazaei, A.R. Moosavi-Zare, A. Zare, Z. Asgari, V. Khakyzadeh, A. Hasaninejad, J. Ind. Eng. Chem. 19, 721 (2013)
S.S. Pandit, S.K. Bhalerao, U.S. Aher, G.L. Adhav, V.U. Pandit, J. Chem. Sci. 123, 421 (2011)
S. Balalaie, M.M. Hashemi, M. Akhbari, Tetrahedron Lett. 44, 1709 (2003)
S.D. Sharma, P. Hazarika, D. Konwar, Tetrahedron Lett. 49, 2216 (2008)
B.F. Mirjalili, A.H. Bamoniri, L. Zamani, ScientiaIranica C. 19, 565 (2012)
D.S. Raghuvanshi, K.N. Singh, Indian J. Chem. 49B, 1394 (2010)
M.R. Mohammadizadeh, A. Hasaninejad, M. Bahramzadeh, Synth. Commun. 39, 3232 (2009)
C. Kurumurthy, G.S. Kumar, G.M. Reddy, P. Nagender, P.S. Rao, B. Narsaiah, Res. Chem. Intermed. 38, 359 (2012)
C.-Y. Chen, W.-P. Hu, P.-C. Yan, G.C. Senadi, J.-J. Wang, Org. Lett. 15, 6116 (2013)
B. Das, J. Kashanna, R.A. Kumar, P. Jangili, Monatsh. Chem. 144, 223 (2013)
F. Nemati, A. Elhampour, M. Bagheri, Natanzi 47, 666 (2017)
S.U. Bhat, R.A. Naikoo, R. Tomar, Int. Res. J. Pure Appl. Chem. 11, 1 (2016)
J. Safari, S. Gandomi-Ravandi, Z. Akbari, J. Adv. Res. 4, 509 (2013)
M. Haghighi, K. Nikoofar, IJC. 5(1), 57 (2015)
A. Khazaei, A.R. Moosavi-zare, F. Gholami, V. Khakyzadeh, Appl. Organomet. Chem. 30(8), 691 (2016)
M. Nejatianfar, B. Akhlaghinia, R. Jahanshahi, Appl. Organomet. Chem. 32(2), 4095 (2018)
M. Safaiee, M.A. Zolfigol, M. Tavasoli, M. Mokhlesi, J. Iran. Chem. Soc. 11, 1593 (2014)
M.A. Zolfigol, A. Khazaei, M. Safaiee, M. Mokhlesi, R. Rostamian, M. Bagheri, M. Shiri, H.G. Kruger, J. Mol. Catal. A: Chem. 370, 80 (2013)
A. Khazaei, M.A. Zolfigol, M. Safaiee, M. Mokhlesi, E. Donyadari, M. Shiri, H.G. Kruger, Catal. Commun. 26, 34 (2012)
M.A. Zolfigol, M. Safaiee, F. Afsharnadery, N. Bahrami-Nejad, S. Baghery, S. Salehzadeh, F. Maleki, RSC Adv. 5, 100546 (2015)
Acknowledgements
The authors are thankful for the facilities provided to carry out research in chemistry research laboratory at Ayatollah Amoli Branch, Islamic Azad University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Alipour, S., Vahdat, S.M. & Chekin, F. Ag–TiO2 nanocomposite-catalyzed one-pot synthesis of 1,2,4,5-tetrasubstituted imidazoles: a green and benign approach. J IRAN CHEM SOC 18, 2315–2321 (2021). https://doi.org/10.1007/s13738-021-02192-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-021-02192-1