Abstract
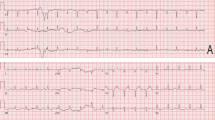
Entrectinib, a multikinase inhibitor of ROS1 and tropomyosin receptor kinases, is recommended to treat ROS1-positive metastatic non-small cell lung cancer (NSCLC). In a previous study, entrectinib-related cardiotoxicity occurred in 2% of patients; however, lethal arrhythmias remain understudied. We encountered a case of fatal arrhythmia due to drug-induced Brugada syndrome caused by entrectinib. An 81-year-old Japanese male with lung adenocarcinoma harboring ROS1-fusion gene was treated with entrectinib. The patient developed lethal arrhythmias three days after drug initiation, including ventricular tachycardia with Brugada-like electrocardiogram changes. Echocardiography and coronary angiography revealed no evidence of acute coronary syndrome or myocarditis. Following the termination of entrectinib, the electrocardiogram abnormality improved within 12 days. Hence, paying special attention to and monitoring electrocardiogram changes is necessary. In addition, it is also necessary to consider early therapeutic interventions and discontinuation of the drug in cases of drug-induced Brugada syndrome.



Similar content being viewed by others
Data availability
All data included in this report are available upon reasonable request from the corresponding author.
References
Kris MG, Johnson BE, Berry LD et al (2014) Using multiplexed assays of oncogenic drivers in lung cancers to select targeted drugs. JAMA 311:1998–2006. https://doi.org/10.1001/jama.2014.3741
Drilon A, Siena S, Dziadziuszko R et al (2020) Entrectinib in ROS1 fusion-positive non-small-cell lung cancer: integrated analysis of three phase 1–2 trials. Lancet Oncol 21:261–270. https://doi.org/10.1016/s1470-2045(19)30690-4
Fonseca M, Chen DH, Walker JM et al (2021) Entrectinib-related myocarditis in a young female patient with metastatic non-small cell lung cancer. BMJ Case Reports. 14:e243946. https://doi.org/10.1136/bcr-2021-243946
Martineau C, Turcotte M-K, Otis N et al (2022) Management of adverse events related to first-generation tyrosine receptor kinase inhibitors in adults: a narrative review. Support Care Cancer 30:10471–10482. https://doi.org/10.1007/s00520-022-07401-y
Tisdale JE, Chung MK, Campbell KB et al (2020) Drug-induced arrhythmias: a scientific statement from the american heart association. Circulation 142:e214–e233. https://doi.org/10.1161/cir.0000000000000905
Konigstein M, Rosso R, Topaz G et al (2016) Drug-induced brugada syndrome: clinical characteristics and risk factors. Heart Rhythm 13:1083–1087. https://doi.org/10.1016/j.hrthm.2016.03.016
Sgherza N, Rossi AVR, Colonna P et al (2013) Use of tyrosine kinase inhibitors in a patient with Brugada syndrome and chronic myeloid leukemia. Int J Hematol 98:483–486. https://doi.org/10.1007/s12185-013-1395-8
Nardin C, Colas M, Badoz M et al (2017) Brugada syndrome induced by BRAF and MEK inhibitors in a melanoma patient. Eur Heart J 38:2151–2151. https://doi.org/10.1093/eurheartj/ehx133
Tang CPS, Lip GYH, McCormack T et al (2022) Management of cardiovascular complications of bruton tyrosine kinase inhibitors. Brit J Haematol 196:70–78. https://doi.org/10.1111/bjh.17788
Turker I, Ai T, Itoh H et al (2017) Drug-induced fatal arrhythmias: acquired long QT and Brugada syndromes. Pharmacol Therapeut 176:48–59. https://doi.org/10.1016/j.pharmthera.2017.05.001
Otsu Y, Kata Y, Takayasu H et al (2022) Entrectinib-induced heart failure in a patient with metastatic lung adenocarcinoma a case report. Cureus. 14:e32174. https://doi.org/10.7759/cureus.32174
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
Funding
This case report was not supported by any funds, grants, or other means of support.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of the study. KF and TH wrote the first draft of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
T. Hase received personal fees and research funding from Chugai Pharmaceutical Co. outside the submitted work. All remaining authors have no conflicts of interest to declare.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Inform consent
The patient gave written informed consent as a part of an observational study approved by the Ethics Review Committee of Nagoya University Graduate School of Medicine (No. 2018–0386).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Futamura, K., Hase, T., Tanaka, A. et al. Lethal ventricular arrhythmia due to entrectinib-induced Brugada syndrome: a case report and literature review. Int Canc Conf J 12, 299–304 (2023). https://doi.org/10.1007/s13691-023-00620-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13691-023-00620-y