Abstract
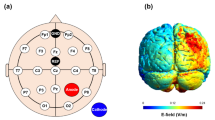
Purpose: The purpose of this study was to investigate the neuromodulatory effects of transauricular vagus nerve stimulation (taVNS) and determine optimal taVNS duration to induce the meaningful neuromodulatroty effects using resting-state electroencephalography (EEG). Method: Fifteen participants participated in this study and taVNS was applied to the cymba conchae for a duration of 40 min. Resting-state EEG was measured before and during taVNS application. EEG power spectral density (PSD) and brain network indices (clustering coefficient and path length) were calculated across five frequency bands (delta, theta, alpha, beta and gamma), respectively, to assess the neuromodulatory effect of taVNS. Moreover, we divided the whole brain region into the five regions of interest (frontal, central, left temporal, right temporal, and occipital) to confirm the neuromodulation effect on each specific brain region. Result: Our results demonstrated a significant increase in EEG frequency powers across all five frequency bands during taVNS. Furthermore, significant changes in network indices were observed in the theta and gamma bands compared to the pre-taVNS measurements. These effects were particularly pronounced after approximately 10 min of stimulation, with a more dominant impact observed after approximately 20–30 min of taVNS application. Conclusion: The findings of this study indicate that taVNS can effectively modulate the brain activity, thereby exerting significant effects on brain characteristics. Moreover, taVNS duration of approximately 20–30 min was considered appropriate for inducing a stable and efficient neuromodulatory effects. Consequently, these findings have the potential to contribute to research aimed at enhancing cognitive and motor functions through the modulation of EEG using taVNS.








Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
References
Sellaro R, de Gelder B, Finisguerra A, Colzato LS. Transcutaneous vagus nerve stimulation (tVNS) enhances recognition of emotions in faces but not bodies. Cortex. 2018;99:213–23.
Yuan H, Silberstein SD. Vagus nerve and vagus nerve stimulation, a comprehensive review: part I. Headache. 2016;56(1):71–8.
Meng F-G, Jia F-M, Ren X-H, Ge Y, Wang K-L, Ma Y-S, Ge M, Zhang K, Hu W-H, Zhang X. Vagus nerve stimulation for pediatric and adult patients with pharmaco-resistant epilepsy. Chin Med J-Peking. 2015;128(19):2599–604.
Ben-Menachem E. Vagus-nerve stimulation for the treatment of epilepsy. Lancet Neurol. 2002;1(8):477–82.
Austelle CW, O’Leary GH, Thompson S, Gruber E, Kahn A, Manett AJ, Short B, Badran BW. A comprehensive review of vagus nerve stimulation for depression. Neuromodulation. 2022;25(3):309–15.
Grimm S, Bajbouj M. Efficacy of vagus nerve stimulation in the treatment of depression. Expert Rev Neurother. 2010;10(1):87–92.
Stegeman I, Velde H, Robe P, Stokroos R, Smit A. Tinnitus treatment by vagus nerve stimulation: a systematic review. PLoS ONE. 2021;16(3):e0247221.
Broncel A, Bocian R, Kłos-Wojtczak P, Kulbat-Warycha K, Konopacki J. Vagal nerve stimulation as a promising tool in the improvement of cognitive disorders. Brain Res Bull. 2020;155:37–47.
Merrill CA, Jonsson MA, Minthon L, Ejnell H, Silander HC-s, Blennow K, Karlsson M, Nordlund A, Rolstad S, Warkentin S. Vagus nerve stimulation in patients with Alzheimer’s disease: additional follow-up results of a pilot study through 1 year. J Clin Psychiat. 2006;67(8):1171–8.
Yap JY, Keatch C, Lambert E, Woods W, Stoddart PR, Kameneva T. Critical review of transcutaneous vagus nerve stimulation: challenges for translation to clinical practice. Front Neurosci-Switz. 2020;284.
Bauer S, Baier H, Baumgartner C, Bohlmann K, Fauser S, Graf W, Hillenbrand B, Hirsch M, Last C, Lerche H. Transcutaneous vagus nerve stimulation (tVNS) for treatment of drug-resistant epilepsy: a randomized, double-blind clinical trial (cMPsE02). Brain Stimul. 2016;9(3):356–63.
Fang J, Egorova N, Rong P, Liu J, Hong Y, Fan Y, Wang X, Wang H, Yu Y, Ma Y. Early cortical biomarkers of longitudinal transcutaneous vagus nerve stimulation treatment success in depression. Neuroimage-Clin. 2017;14:105–11.
Liu J, Fang J, Wang Z, Rong P, Hong Y, Fan Y, Wang X, Park J, ** Y, Liu C. Transcutaneous vagus nerve stimulation modulates amygdala functional connectivity in patients with depression. J Affect Disorder. 2016;205:319–26.
Yakunina N, Kim SS, Nam E-C. BOLD fMRI effects of transcutaneous vagus nerve stimulation in patients with chronic tinnitus. PLoS ONE. 2018;13(11):e0207281.
Shim HJ, Kwak MY, An Y-H, Kim DH, Kim YJ, Kim HJ. Feasibility and safety of transcutaneous vagus nerve stimulation paired with notched music therapy for the treatment of chronic tinnitus. J Audil Otol. 2015;19(3):159.
De Ridder D, Vanneste S, Engineer ND, Kilgard MP. Safety and efficacy of vagus nerve stimulation paired with tones for the treatment of tinnitus: a case series. Neuromodulation. 2014;17(2):170–9.
Li R, Yang D, Fang F, Hong K-S, Reiss AL, Zhang Y. Concurrent fNIRS and EEG for brain function investigation: a systematic, methodology-focused review. Sensors-Basel. 2022;22(15):5865.
He B, Yang L, Wilke C, Yuan H. Electrophysiological imaging of brain activity and connectivity—challenges and opportunities. IEEE T Bio-Med Neg. 2011;58(7):1918–31.
Pihlaja M, Failla L, Peräkylä J, Hartikainen KM. Reduced frontal Nogo-N2 with uncompromised response inhibition during transcutaneous vagus nerve stimulation—more efficient cognitive control? Front Hum Neurosci. 2020;14:561780.
Fischer R, Ventura-Bort C, Hamm A, Weymar M. Transcutaneous vagus nerve stimulation (tVNS) enhances conflict-triggered adjustment of cognitive control. Cog Affect Behav Ne. 2018;18:680–93.
Sharon O, Fahoum F, Nir Y. Transcutaneous vagus nerve stimulation in humans induces pupil dilation and attenuates alpha oscillations. J Neurosci. 2021;41(2):320–30.
Ricci L, Croce P, Lanzone J, Boscarino M, Zappasodi F, Tombini M, Di Lazzaro V, Assenza G. Transcutaneous vagus nerve stimulation modulates EEG microstates and delta activity in healthy subjects. Brain Sci. 2020;10(10):668.
Bullmore E, Sporns O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci. 2009;10(3):186–98.
Rubinov M, Sporns O. Complex network measures of brain connectivity: uses and interpretations. NeuroImage. 2010;52(3):1059–69.
Latora V, Marchiori M. Efficient behavior of small-world networks. Phys Rev Lett. 2001;87(19):198701.
Smit DJ, Stam CJ, Posthuma D, Boomsma DI, De Geus EJ. Heritability of small-world networks in the brain: a graph theoretical analysis of resting‐state EEG functional connectivity. Hum Brain Mapp. 2008;29(12):1368–78.
Bastos AM, Schoffelen J-M. A tutorial review of functional connectivity analysis methods and their interpretational pitfalls. Front Syst Neurosci. 2016;9:175.
Gianlorenco ACL, de Melo PS, Marduy A, Kim AY, Kim CK, Choi H, Song J-J, Fregni F. Electroencephalographic patterns in taVNS: a systematic review. Biomedicines. 2022;10(9):2208.
Farmer AD, Strzelczyk A, Finisguerra A, Gourine AV, Gharabaghi A, Hasan A, Burger AM, Jaramillo AM, Mertens A, Majid A. International consensus based review and recommendations for minimum reporting standards in research on transcutaneous vagus nerve stimulation (version 2020). Front Hum Neurosci. 2021;14:568051.
Chen L, Zhang J, Wang Z, Zhang X, Zhang L, Xu M, Liu S, Ming D. Effects of transcutaneous vagus nerve stimulation (tVNS) on action planning: a behavioural and EEG study. IEEE T Neur SYS REH. 2021;30:1675–83.
Clancy JA, Mary DA, Witte KK, Greenwood JP, Deuchars SA, Deuchars J. Non-invasive vagus nerve stimulation in healthy humans reduces sympathetic nerve activity. Brain Stimul. 2014;7(6):871–7.
Kreuzer PM, Landgrebe M, Husser O, Resch M, Schecklmann M, Geisreiter F, Poeppl TB, Prasser SJ, Hajak G, Langguth B. Transcutaneous vagus nerve stimulation: retrospective assessment of cardiac safety in a pilot study. Front Psychiatry. 2012;3:70.
Keute M, Wienke C, Ruhnau P, Zaehle T. Effects of transcutaneous vagus nerve stimulation (tVNS) on beta and gamma brain oscillations. Cortex. 2021;140:222–31.
Chan H-L, Tsai Y-T, Meng L-F, Wu T. The removal of ocular artifacts from EEG signals using adaptive filters based on ocular source components. Ann Biomed Eng. 2010;38:3489–99.
Donaldson PH, Kirkovski M, Rinehart NJ, Enticott PG. A double-blind HD-tDCS/EEG study examining right temporoparietal junction involvement in facial emotion processing. Soc Neurosci-UK. 2019;14(6):681–96.
Oostenveld R, Fries P, Maris E, Schoffelen J-M. FieldTrip. Open source software for advanced analysis of MEG, EEG, and invasive electrophysiological data. Comput Intel Neurosc. 2011;2011:1–9.
Friedman M. The use of ranks to avoid the assumption of normality implicit in the analysis of variance. J Am Stat Assoc. 1937;32(200):675–701.
Berger A, Pixa NH, Steinberg F, Doppelmayr M. Brain oscillatory and hemodynamic activity in a bimanual coordination task following transcranial alternating current stimulation (tACS): a combined EEG-fNIRS study. Front Behav Neurosci. 2018;12:67.
Spitoni GF, Cimmino RL, Bozzacchi C, Pizzamiglio L, Di Russo F. Modulation of spontaneous alpha brain rhythms using low-intensity transcranial direct-current stimulation. Front Hum Neurosci. 2013;7:529.
Mangia AL, Pirini M, Cappello A. Transcranial direct current stimulation and power spectral parameters: a tDCS/EEG co-registration study. Front Hum Neurosci. 2014;8:601.
Keatch C, Lambert E, Woods W, Kameneva T. Measuring brain response to transcutaneous vagus nerve stimulation (tVNS) using simultaneous magnetoencephalography (MEG). J Neural Eng. 2022;19(2):026038.
Rings T, von Wrede R, Bröhl T, Schach S, Helmstaedter C, Lehnertz K. Impact of transcutaneous auricular vagus nerve stimulation on large-scale functional brain networks: from local to global. Front Physiol. 2021; 1328.
von Wrede R, Rings T, Schach S, Helmstaedter C, Lehnertz K. Transcutaneous auricular vagus nerve stimulation induces stabilizing modifications in large-scale functional brain networks: towards understanding the effects of taVNS in subjects with epilepsy. Sci Rep-UK. 2021;11(1):7906.
Sauseng P, Griesmayr B, Freunberger R, Klimesch W. Control mechanisms in working memory: a possible function of EEG theta oscillations. Neurosci Biobehav R. 2010;34(7):1015–22.
Jensen O, Tesche CD. Frontal theta activity in humans increases with memory load in a working memory task. Eur J Neurosci. 2002;15(8):1395–9.
Howard MW, Rizzuto DS, Caplan JB, Madsen JR, Lisman J, Aschenbrenner-Scheibe R, Schulze-Bonhage A, Kahana MJ. Gamma oscillations correlate with working memory load in humans. Cereb Cortex. 2003;13(12):1369–74.
Roux F, Wibral M, Mohr HM, Singer W, Uhlhaas PJ. Gamma-band activity in human prefrontal cortex codes for the number of relevant items maintained in working memory. J Neurosci. 2012;32(36):12411–20.
Sun J-B, Cheng C, Tian Q-Q, Yuan H, Yang X-J, Deng H, Guo X-Y, Cui Y-P, Zhang M-K, Yin Z-X. Transcutaneous auricular vagus nerve stimulation improves spatial working memory in healthy young adults. Front Neurosci-Switz. 2021;15:790793.
Jacobs HI, Riphagen JM, Razat CM, Wiese S, Sack AT. Transcutaneous vagus nerve stimulation boosts associative memory in older individuals. Neurobiol Aging. 2015;36(5):1860–7.
Rufener KS, Geyer U, Janitzky K, Heinze HJ, Zaehle T. Modulating auditory selective attention by non-invasive brain stimulation: Differential effects of transcutaneous vagal nerve stimulation and transcranial random noise stimulation. Eur J Neurosci. 2018;48(6):2301–9.
Gadeyne S, Mertens A, Van den Carrette E, Boon P, Raedt R, Vonck K. Transcutaneous auricular vagus nerve stimulation cannot modulate the P3b event-related potential in healthy volunteers. Clin Neurophysiol. 2022;135:22–9.
Imburgio MJ, Orr JM. Effects of prefrontal tDCS on executive function: methodological considerations revealed by meta-analysis. Neuropsychologia. 2018;117:156–66.
Wu L, Liu T, Wang J. Improving the effect of transcranial alternating current stimulation (tACS): a systematic review. Front Hum Neurosci. 2021;15:652393.
Lee TL, Lee H, Kang N. A meta-analysis showing improved cognitive performance in healthy young adults with transcranial alternating current stimulation. Npj Sci Learn. 2023;8(1):1.
Funding
This work was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by Ministry of Education (No. 2019R1I1A3A01060732), the National Research Foundation of Korea(NRF) grant funded by the Korea government(MSIT) (No. IITP-2024-RS-2023-00258971), and the Neurive Co., Ltd (Q2129191).
Author information
Authors and Affiliations
Contributions
All authors designed the initial study. Analyzed the data, and wrote the manuscript by [Yun-Sung Lee] and [Woo-** Kim]. Wrote the manuscript by [Miseon Shim], [Hyuk Choi], and [Jae-Jun Song]. Provided the study instrumentation by [Ki Hwan Hong], [Hyuk Choi] and [Jae-Jun Song]. Supervised data analysis, and revised the manuscript by [Han-Jeong Hwang]. All authors had full access to all data in the study and responsibility for the integrity of the data and accuracy of the data analysis.
Corresponding author
Ethics declarations
Ethical approval
The protocol of this study was approved by the Institutional Review Board (IRB) of Korea University (KUIRB-2022-0077-01).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, YS., Kim, WJ., Shim, M. et al. Investigating neuromodulatory effect of transauricular vagus nerve stimulation on resting-state electroencephalography. Biomed. Eng. Lett. 14, 677–687 (2024). https://doi.org/10.1007/s13534-024-00361-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13534-024-00361-8