Abstract
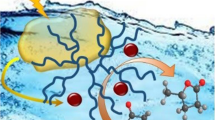
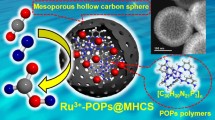
A Ru-decorated porous melamine polymer is found to be an active catalyst upon selective hydrogenation of aqueous 5-hydroxymethylfurfural to 2,5-bis-(hydroxymethyl)furan (yield > 99%) under mild (20 bar H2, 30–90 °C), base/additive free conditions. Owing to its porous structure and unique surface chemistry presenting abundant weakly basic N-sites (amine and triazine), the polymeric catalyst could outperform various benchmark Ru catalysts (viz. Ru/AC, Ru/SBA-15, Ru/Nb2O5, Ru/NbOPO4, Ru/NC, and Ru/g-C3N4) in terms of activity and desired product selectivity. The catalytic material was also found to be reusable and maintained good performance during multiple recycles under kinetic regime in batch mode. Furthermore, the polymeric catalyst also showed good performance for selective HMF hydrogenation under intensified conditions in a fixed-bed reactor achieving constant BHMF yield during 20-h steady-state operation under relatively mild conditions (70 °C, 20 bar, WHSV 0.2 h−1).











Similar content being viewed by others
Data availability
Not applicable.
References
Huber GW, Iborra S, Corma A (2006) Synthesis of transportation fuels from biomass: chemistry, catalysts, and engineering. Chem Rev 106:4044–4098
Alonso DM, Bond JQ, Dumesic JA (2010) Catalytic conversion of biomass to biofuels. Green Chem 12:1493–1513
Dutta S, De S, Saha B (2012) A brief summary of the synthesis of polyester building-block chemicals and biofuels from 5-hydroxymethylfurfural. ChemPlusChem 77:259–272
Kumalaputri AJ, Bottari G, Erne PM, Heeres HJ, Barta K (2014) Tunable and selective conversion of 5-HMF to 2,5-furandimethanol and 2,5-dimethylfuran over copper-doped porous metal oxides. Chemsuschem 7:2266–2275
Nagpure AS, Gogoi P, Lucas N, Chilukuri SV (2020) Novel Ru nanoparticle catalysts for the catalytic transfer hydrogenation of biomass-derived furanic compounds. Sustainable Energy Fuels 4:3654–3667
Gallezot P (2012) Conversion of biomass to selected chemical products. Chem Soc Rev 41:1538–1558
Chatterjee M, Ishizaka T, Kawanami H (2014) Selective hydrogenation of 5-hydroxymethylfurfural to 2,5-bis-(hydroxymethyl)furan using Pt/MCM-41 in an aqueous medium: a simple approach. Green Chem 16:4734–4739
Buntara T, Noel S, Phua PH, Melián-Cabrera I, de Vries JG, Heeres HJ (2011) Caprolactam from renewable resources: catalytic conversion of 5-hydroxymethylfurfural into caprolactone. Angew Chem 123:7221–7225
De S, Burange AS, Luque R (2022) Conversion of biomass-derived feedstocks into value-added chemicals over single-atom catalysts. Green Chem 24:2267–2286
Fujita S, Nakajima K, Yamasaki J, Mizugaki T, Jitsukawa K, Mitsudome T (2020) Unique catalysis of nickel phosphide nanoparticles to promote the selective transformation of biofuranic aldehydes into diketones in water. ACS Catal 10:4261–4267
Wozniak B, Spannenberg A, Li Y, Hinze S, de Vries JG (2018) Cyclopentanone derivatives from 5-hydroxymethylfurfural via 1-hydroxyhexane-2, 5-dione as intermediate. Chemsuschem 11:356–359
Mishra DK, Cho JK, Yi Y, Lee HJ, Kim YJ (2019) Hydroxyapatite supported gold nanocatalyst for base-free oxidative esterification of 5-hydroxymethyl-2-furfural to 2,5-furan dimethylcarboxylate with air as oxidant. J Ind Eng Chem 70:338–345
Zeng C, Seino H, Ren J, Hatanaka K, Yoshie N (2013) Bio-based furan polymers with self-healing abilitY. Macromolecules 46:1794–1802
Hu L, Xu J, Zhou S, He A, Tang X, Lin L, Xu J, Zhao Y (2018) Catalytic advances in the production and application of biomass-derived 2, 5-dihydroxymethylfuran. ACS Catal 8:2959–2980
Cao Q, Liang W, Guan J, Wang L, Qu Q, Zhang X, Wang X, Mu X (2014) Catalytic synthesis of 2, 5-bis-methoxymethylfuran: a promising cetane number improver for diesel. Appl Catal A 481:49–53
Alamillo R, Tucker M, Chia M, Pagan-Torres Y, Dumesic J (2012) The selective hydrogenation of biomass-derived 5-hydroxymethylfurfural using heterogeneous catalysts. Green Chem 14:1413–1419
Che P, Lu F, Zhang J, Huang Y, Nie X, Gao J, Xu J (2012) Catalytic selective etherification of hydroxyl groups in 5-hydroxymethylfurfural over H4SiW12O40/MCM-41 nanospheres for liquid fuel production. Bioresour Technol 119:433–436
Vikanova K, Redina E, Kapustin G, Chernova M, Tkachenko O, Nissenbaum V, Kustov L (2021) Advanced room-temperature synthesis of 2, 5-bis (hydroxymethyl) furan—a monomer for biopolymers—from 5-hydroxymethylfurfural. ACS Sustain Chem Eng 9:1161–1171
Chimentão RJ, Oliva H, Belmar J, Morales K, Mäki-Arvela P, Wärnå J, Murzin DY, Fierro J, Llorca J, Ruiz D (2019) Selective hydrodeoxygenation of biomass derived 5-hydroxymethylfurfural over silica supported iridium catalysts. Appl Catal B 241:270–283
Chimentão RJ, Oliva H, Russo V, Llorca J, Fierro JL, Maki-Arvela P, Murzin DY, Ruiz D (2021) Catalytic transformation of biomass-derived 5-hydroxymethylfurfural over supported bimetallic iridium-based catalysts. The Journal of Physical Chemistry C 125:9657–9678
Fulignati S, Antonetti C, Licursi D, Pieraccioni M, Wilbers E, Heeres HJ, Galletti AMR (2019) Insight into the hydrogenation of pure and crude HMF to furan diols using Ru/C as catalyst. Appl Catal A 578:122–133
Mishra DK, Lee HJ, Truong CC, Kim J, Suh YW, Baek J, Kim YJ (2020) Ru/MnCo2O4 as a catalyst for tunable synthesis of 2,5-bis(hydroxymethyl) furan or 2,5-bis(hydroxymethyl)tetrahydrofuran from hydrogenation of 5-hydroxymethylfurfural. Mol Catal 484:110722
Fulignati S, Antonetti C, Wilbers E, Licursi D, Heeres HJ, Galletti AMR (2021) Tunable HMF hydrogenation to furan diols in a flow reactor using Ru/C as catalyst. J Ind Eng Chem 100:390.e1-390.e9
Wang T, Zhang J, **e W, Tang Y, Guo D, Ni Y (2017) Catalytic transfer hydrogenation of biobased HMF to 2, 5-bis-(hydroxymethyl) furan over Ru/Co3O4. Catalysts 7:92
Chen J, Lu F, Zhang J, Yu W, Wang F, Gao J, Xu J (2013) Immobilized Ru clusters in nanosized mesoporous zirconium silica for the aqueous hydrogenation of furan derivatives at room temperature. ChemCatChem 5:2822–2826
Cerezo-Navarrete C, Mathieu Y, Puche M, Morales C, Concepción P, Martínez-Prieto LM, Corma A (2021) Controlling the selectivity of bimetallic platinum–ruthenium nanoparticles supported on N-doped graphene by adjusting their metal composition. Catal Sci Technol 11:494–505
Ohyama J, Esaki A, Yamamoto Y, Arai S, Satsuma A (2013) Selective hydrogenation of 2-hydroxymethyl-5-furfural to 2,5-bis(hydroxymethyl)furan over gold sub-nano clusters. RSC Adv 3:1033–1036
Kim J, Bathula HB, Yun S, Jo Y, Lee S, Baik JH, Suh Y-W (2021) Hydrogenation of 5-hydroxymethylfurfural into 2, 5-bis (hydroxymethyl) furan over mesoporous Cu–Al2O3 catalyst: From batch to continuous processing. J Ind Eng Chem 102:186–194
Lima S, Chadwick D, Hellgardt K (2017) Towards sustainable hydrogenation of 5-(hydroxymethyl)furfural: a two-stage continuous process in aqueous media over RANEY® catalysts. RSC Adv 7:31401–31407
Feng Y, Yan G, Wang T, Jia W, Zeng X, Sperry J, Sun Y, Tang X, Lei T, Lin L (2019) Cu 1–Cu 0 bicomponent CuNPs@ ZIF-8 for highly selective hydrogenation of biomass derived 5-hydroxymethylfurfural. Green Chem 21:4319–4323
Rao KTV, Hu Y, Yuan Z, Zhang Y, Xu CC (2021) Green synthesis of heterogeneous copper-alumina catalyst for selective hydrogenation of pure and biomass-derived 5-hydroxymethylfurfural to 2, 5-bis (hydroxymethyl) furan. Appl Catal A 609:117892
Subbiah S, Simeonov SP, Esperança JM, Rebelo LPN, Afonso CA (2013) Direct transformation of 5-hydroxymethylfurfural to the building blocks 2, 5-dihydroxymethylfurfural (DHMF) and 5-hydroxymethyl furanoic acid (HMFA) via Cannizzaro reaction. Green Chem 15:2849–2853
Wiesfeld JJ, Kim M, Nakajima K, Hensen EJ (2020) Selective hydrogenation of 5-hydroxymethylfurfural and its acetal with 1, 3-propanediol to 2, 5-bis (hydroxymethyl) furan using supported rhenium-promoted nickel catalysts in water. Green Chem 22:1229–1238
Padilla R, Koranchalil S, Nielsen M (2020) Efficient and selective catalytic hydrogenation of furanic aldehydes using well defined Ru and Ir pincer complexes. Green Chem 22:6767–6772
Bui TQ, Konwar LJ, Samikannu A, Nikjoo D, Mikkola J-P (2020) Mesoporous melamine-formaldehyde resins as efficient heterogeneous catalysts for continuous synthesis of cyclic carbonates from epoxides and gaseous CORu3p. ACS Sustain Chemi Eng 8:12852–12869
Donoeva B, Masoud N, De Jongh PE (2017) Carbon support surface effects in the gold-catalyzed oxidation of 5-hydroxymethylfurfural. ACS Catal 7:4581–4591
Konwar LJ, M J-P (2022) Carbon support effects on metal (Pd, Pt and Ru) catalyzed hydrothermal decarboxylation/deoxygenation of triglycerides. Appl Catal A 638(2022):118611
Shcherban N, Mäki-Arvela P, Aho A, Sergiienko S, Yaremov P, Eränen K, Murzin DY (2018) Melamine-derived graphitic carbon nitride as a new effective metal-free catalyst for Knoevenagel condensation of benzaldehyde with ethylcyanoacetate, Catalysis. Sci Technol 8:2928–2937
Samikannu A, Konwar LJ, Mäki-Arvela P, Mikkola J-P (2019) Renewable N-doped active carbons as efficient catalysts for direct synthesis of cyclic carbonates from epoxides and CO2. Appl Catal B 241:41–51
Gu S, **e J, Li CM (2014) Hierarchically porous graphitic carbon nitride: large-scale facile synthesis and its application toward photocatalytic dye degradation. RSC Adv 4:59436–59439
Teng W, Wu Z, Fan J, Zhang W-X, Zhao D (2015) Amino-functionalized ordered mesoporous carbon for the separation of toxic microcystin-LR. J Mater Chem A 3:19168–19176
Houshang F, Fatemeh H, Rahmatollah R, Ali G (2014) Surfactant-free hydrothermal synthesis of mesoporous niobia samples and their photoinduced decomposition of terephthalic acid (TPA). J Cluster Sci 25:651–666
Kokunešoski M, Gulicovski J, Matović B, Logar M, Milonjić SK, Babić B (2010) Synthesis and surface characterization of ordered mesoporous silica SBA-15. Mater Chem Phys 124:1248–1252
Kosmulski M (2016) Isoelectric points and points of zero charge of metal (hydr) oxides: 50 years after Parks’ review. Adv Coll Interface Sci 238:1–61
Pan C-J, Tsai M-C, Su W-N, Rick J, Akalework NG, Agegnehu AK, Cheng S-Y, Hwang B-J (2017) Tuning/exploiting strong metal-support interaction (SMSI) in heterogeneous catalysis. J Taiwan Inst Chem Eng 74:154–186
Stefano C, Hadi N, Ferenc S, Claudio E, Villa A, Prati L (2018) Ruthenium on carbonaceous materials for the selective hydrogenation of HMF. Molecules 23(8):2007
**a H, Xu S, Hu H, An J, Li C (2018) Efficient conversion of 5-hydroxymethylfurfural to high-value chemicals by chemo-and bio-catalysis. RSC Adv 8:30875–30886
Mohsen-Nia M, Amiri H, Jazi B (2010) Dielectric constants of water, methanol, ethanol, butanol and acetone: measurement and computational study. J Solution Chem 39:701–708
Tucker I, Corbett J, Fatkin J, Jack R, Kaszuba M, MacCreath B, McNeil-Watson F (2015) Laser Doppler Electrophoresis applied to colloids and surfaces. Curr Opin Colloid Interface Sci 20:215–226
Brunner E (1985) Solubility of hydrogen in 10 organic solvents at 298.15, 323.15, and 373.15 K. J Chem Eng Data 30:269–273
De la Iglesia O, Mainar AM, Pardo JI, Urieta JS (2003) Solubilities of nonpolar gases in triethylene glycol dimethyl ether, tetraethylene glycol dimethyl ether, dimethyl carbonate, and diethyl carbonate at 298.15 K and 101.33 kPa partial pressure of gas. J Chem Eng Data 48:657–661
Turkin A, Eyley S, Preegel G, Thielemans W, Makshina E, Sels BF (2021) How trace impurities can strongly affect the hydroconversion of biobased 5-hydroxymethylfurfural? ACS Catal 11:9204–9209
Michel C, Gallezot P (2015) Why is ruthenium an efficient catalyst for the aqueous-phase hydrogenation of biosourced carbonyl compounds? ACS Catal 5:4130–4132
Chen S, Wojcieszak R, Dumeignil F, Marceau E, Royer S, b. (2018) How catalysts and experimental conditions determine the selective hydroconversion of furfural and 5-hydroxymethylfurfural. Chem Rev 118:11023–11117
Acknowledgements
The PRIS registration number of this publication is CSIR-CSMCRI – 13/2022. The authors also thank the Analytical Division and Centralized Instrumental Facilities of this institute for analytical support.
Funding
Financial support from SERB (SRG/2020/000325) and MLP0028 from CSIR, New Delhi.
Author information
Authors and Affiliations
Contributions
M. Mani: data curation, investigation, visualization, writing-original draft; G.K. Govind: investigation, data curation; A.B. Panda: supervision, writing — review and editing; L.J. Konwar: conceptualization, supervision, funding acquisition, writing — review and editing, and project administration.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mani, M., Kadam, G.G., Konwar, L.J. et al. Ru-supported mesoporous melamine polymers as efficient catalysts for selective hydrogenation of aqueous 5-hydroxymethylfurfural to 2,5-bis-(hydroxymethyl)furan. Biomass Conv. Bioref. 14, 6267–6284 (2024). https://doi.org/10.1007/s13399-022-02768-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-022-02768-8