Abstract
Aims
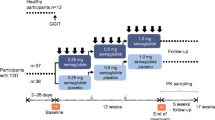
This randomized, open-label, parallel-group, controlled trial compared the effects of dulaglutide and trelagliptin on beta-cell function in patients with type 2 diabetes.
Materials and methods
For 24 weeks, participants received dulaglutide (0.75 mg/week) or trelagliptin (100 mg/week), after which beta-cell function was evaluated using a glucagon stimulation test-based disposition index. The primary endpoint was the change in disposition index over the 24-week treatment period.
Results
Fifty patients with type 2 diabetes who received metformin with or without basal insulin were randomized to receive dulaglutide or trelagliptin. Forty-eight patients completed the 24-week dulaglutide (n = 23) or trelagliptin (n = 25) treatment. The dulaglutide group reduced HbA1c levels more than the trelagliptin group (dulaglutide: − 0.77% ± 0.07% vs. trelagliptin: − 0.57% ± 0.07%; p = 0.04). Change in disposition index during the 24 weeks did not differ between the groups (dulaglutide: − 0.07 ± 1.08 vs. trelagliptin: + 0.59 ± 1.04; p = 0.66), but the dulaglutide group increased HOMA2-%β levels more than the trelagliptin group (dulaglutide: + 26.2 ± 4.3% vs. trelagliptin: + 5.4 ± 4.1%; p = 0.001). The dulaglutide group showed greater body fat mass reduction than the trelagliptin group (dulaglutide: − 1.2 ± 0.3 kg vs. trelagliptin: − 0.3 ± 0.2 kg; p = 0.02) without skeletal muscle mass loss.
Conclusion
Dulaglutide and trelagliptin had similar effects on beta-cell function according to the glucagon stimulation test-based disposition index. However, dulaglutide promoted improved HOMA2-%β levels compared to trelagliptin and body fat mass was reduced without loss of skeletal muscle mass (UMIN-CTR 000024164).




Similar content being viewed by others
Data availability
In order to protect the privacy of the participants in this study, and because permission to share data with third parties was not obtained in the informed consent form, data from this study cannot be provided.
References
U.K. Prospective Diabetes Study Group. U.K. Prospective diabetes study 16: overview of 6 years' therapy of type II Diabetes: a progressive disease. Diabetes. 1995;44(11):1249–1258. https://doi.org/10.2337/diab.44.11.1249.
Bo S, Gentile L, Castiglione A, Prandi V, Canil S, Ghigo E, et al. C-peptide and the risk for incident complications and mortality in type 2 diabetic patients: a retrospective cohort study after a 14-year follow-up. Eur J Endocrinol. 2012;167(2):173–80. https://doi.org/10.1530/EJE-12-0085.
Jendle J, Grunberger G, Blevins T, Giorgino F, Hietpas RT, Botros FT. Efficacy and safety of dulaglutide in the treatment of type 2 diabetes: a comprehensive review of the dulaglutide clinical data focusing on the AWARD phase 3 clinical trial program. Diabetes Metab Res Rev. 2016;32(8):776–90. https://doi.org/10.1002/dmrr.2810.
Seino Y, Yabe D. Alogliptin benzoate for the treatment of type 2 diabetes. Expert Opin Pharmacother. 2014;15(6):851–63. https://doi.org/10.1517/14656566.2014.898750.
McKeage K. Trelagliptin: first global approval. Drugs. 2015;75(10):1161–4. https://doi.org/10.1007/s40265-015-0431-9.
Gerstein HC, Colhoun HM, Dagenais GR, Diaz R, Lakshmanan M, Pais P, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet. 2019;394(10193):121–30. https://doi.org/10.1016/S0140-6736(19)31149-3.
Retnakaran R, Kramer CK, Choi H, Swaminathan B, Zinman B. Liraglutide and the preservation of pancreatic beta-cell function in early type 2 diabetes: the LIBRA trial. Diabetes Care. 2014;37(12):3270–8. https://doi.org/10.2337/dc14-0893.
Kondo Y, Satoh S, Osada UN, Terauchi Y. Early liraglutide treatment improves beta-cell function in patients with type 2 diabetes: a retrospective cohort study. Endocr J. 2015;62(11):971–80. https://doi.org/10.1507/endocrj.EJ15-0206.
Xu L, Man CD, Charbonnel B, Meninger G, Davies MJ, Williams-Herman D, et al. Effect of sitagliptin, a dipeptidyl peptidase-4 inhibitor, on beta-cell function in patients with type 2 diabetes: a model-based approach. Diabetes Obes Metab. 2008;10(12):1212–20. https://doi.org/10.1111/j.1463-1326.2008.00887.x.
Alsalim W, Goransson O, Carr RD, Bizzotto R, Tura A, Pacini G, et al. Effect of single-dose DPP-4 inhibitor sitagliptin on beta-cell function and incretin hormone secretion after meal ingestion in healthy volunteers and drug-naive, well-controlled type 2 diabetes subjects. Diabetes Obes Metab. 2018;20(4):1080–5. https://doi.org/10.1111/dom.13192.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53(6):982–92. https://doi.org/10.1053/j.ajkd.2008.12.034.
Levy JC, Matthews DR, Hermans MP. Correct homeostasis model assessment (HOMA) evaluation uses the computer program. Diabetes Care. 1998;21(12):2191–2. https://doi.org/10.2337/diacare.21.12.2191.
Funakoshi S, Fujimoto S, Hamasaki A, Fujiwara H, Fujita Y, Ikeda K, et al. Utility of indices using C-peptide levels for indication of insulin therapy to achieve good glycemic control in Japanese patients with type 2 diabetes. J Diabetes Investig. 2011;2(4):297–303. https://doi.org/10.1111/j.2040-1124.2010.00096.x.
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28(7):412–9. https://doi.org/10.1007/BF00280883.
Terauchi Y, Satoi Y, Takeuchi M, Imaoka T. Monotherapy with the once weekly GLP-1 receptor agonist dulaglutide for 12 weeks in Japanese patients with type 2 diabetes: dose-dependent effects on glycaemic control in a randomised, double-blind, placebo-controlled study. Endocr J. 2014;61(10):949–59. https://doi.org/10.1507/endocrj.ej14-0147.
Inagaki N, Onouchi H, Maezawa H, Kuroda S, Kaku K. Once-weekly trelagliptin versus daily alogliptin in Japanese patients with type 2 diabetes: a randomised, double-blind, phase 3, non-inferiority study. Lancet Diabetes Endocrinol. 2015;3(3):191–7. https://doi.org/10.1016/S2213-8587(14)70251-7.
Harder H, Nielsen L, Tu DT, Astrup A. The effect of liraglutide, a long-acting glucagon-like peptide 1 derivative, on glycemic control, body composition, and 24-h energy expenditure in patients with type 2 diabetes. Diabetes Care. 2004;27(8):1915–21. https://doi.org/10.2337/diacare.27.8.1915.
Zinman B, Gerich J, Buse JB, Lewin A, Schwartz S, Raskin P, et al. Efficacy and safety of the human glucagon-like peptide-1 analog liraglutide in combination with metformin and thiazolidinedione in patients with type 2 diabetes (LEAD-4 Met+TZD). Diabetes Care. 2009;32(7):1224–30. https://doi.org/10.2337/dc08-2124.
Linn T, Schneider K, Goke B, Federlin K. Glucagon-like-peptide-1 (7–36) amide improves glucose sensitivity in beta-cells of NOD mice. Acta Diabetol. 1996;33(1):19–24. https://doi.org/10.1007/BF00571935.
Moens K, Flamez D, Van Schravendijk C, Ling Z, Pipeleers D, Schuit F. Dual glucagon recognition by pancreatic beta-cells via glucagon and glucagon-like peptide 1 receptors. Diabetes. 1998;47(1):66–72. https://doi.org/10.2337/diab.47.1.66.
Holst JJ. The physiology of glucagon-like peptide 1. Physiol Rev. 2007;87(4):1409–39. https://doi.org/10.1152/physrev.00034.2006.
Drucker DJ, Holst JJ. The expanding incretin universe: from basic biology to clinical translation. Diabetologia. 2023;66(10):1765–79. https://doi.org/10.1007/s00125-023-05906-7.
Kondo Y, Satoh S, Nagakura J, Kimura M, Nezu U, Terauchi Y. Defining criteria for the introduction of liraglutide using the glucagon stimulation test in patients with type 2 diabetes. J Diabetes Investig. 2013;4(6):571–5. https://doi.org/10.1111/jdi.12082.
Usui R, Yabe D, Kuwata H, Fujiwara S, Watanabe K, Hyo T, et al. Retrospective analysis of safety and efficacy of insulin-to-liraglutide switch in Japanese type 2 diabetes: A caution against inappropriate use in patients with reduced beta-cell function. J Diabetes Investig. 2013;4(6):585–94. https://doi.org/10.1111/jdi.12111.
Ohbatake A, Yagi K, Karashima S, Shima Y, Miyamoto Y, Asaka H, et al. C-peptide area under the curve at glucagon stimulation test predicts glucose improvements by GLP-1 receptor analogue: a retrospective observational study. Diabetes Ther. 2019;10(2):673–81. https://doi.org/10.1007/s13300-019-0586-6.
Wu S, Gao L, Cipriani A, Huang Y, Yang Z, Yang J, et al. The effects of incretin-based therapies on beta-cell function and insulin resistance in type 2 diabetes: A systematic review and network meta-analysis combining 360 trials. Diabetes Obes Metab. 2019;21(4):975–83. https://doi.org/10.1111/dom.13613.
Guglielmi C, Del Toro R, Lauria A, Maurizi AR, Fallucca S, Cappelli A, et al. Effect of GLP-1 and GIP on C-peptide secretion after glucagon or mixed meal tests: Significance in assessing B-cell function in diabetes. Diabetes Metab Res Rev. 2017. https://doi.org/10.1002/dmrr.2899.
Shah M, Vella A. Effects of GLP-1 on appetite and weight. Rev Endocr Metab Disord. 2014;15(3):181–7. https://doi.org/10.1007/s11154-014-9289-5.
Da Porto A, Casarsa V, Colussi G, Catena C, Cavarape A, Sechi L. Dulaglutide reduces binge episodes in type 2 diabetic patients with binge eating disorder: A pilot study. Diabetes Metab Syndr. 2020;14(4):289–92. https://doi.org/10.1016/j.dsx.2020.03.009.
Funakoshi S, Fujimoto S, Hamasaki A, Fujiwara H, Fujita Y, Ikeda K, et al. Analysis of factors influencing pancreatic beta-cell function in Japanese patients with type 2 diabetes: association with body mass index and duration of diabetic exposure. Diabetes Res Clin Pract. 2008;82(3):353–8. https://doi.org/10.1016/j.diabres.2008.09.010.
Saisho Y, Tanaka K, Abe T, Shimada A, Kawai T, Itoh H. Effect of obesity on declining beta cell function after diagnosis of type 2 diabetes: a possible link suggested by cross-sectional analysis. Endocr J. 2012;59(3):187–95. https://doi.org/10.1507/endocrj.ej11-0206.
Donath MY, Shoelson SE. Type 2 diabetes as an inflammatory disease. Nat Rev Immunol. 2011;11(2):98–107. https://doi.org/10.1038/nri2925.
Eizirik DL, Cardozo AK, Cnop M. The role for endoplasmic reticulum stress in diabetes mellitus. Endocr Rev. 2008;29(1):42–61. https://doi.org/10.1210/er.2007-0015.
Poitout V, Amyot J, Semache M, Zarrouki B, Hagman D, Fontes G. Glucolipotoxicity of the pancreatic beta cell. Biochim Biophys Acta. 2010;1801(3):289–98. https://doi.org/10.1016/j.bbalip.2009.08.006.
Bunck MC, Diamant M, Corner A, Eliasson B, Malloy JL, Shaginian RM, et al. One-year treatment with exenatide improves beta-cell function, compared with insulin glargine, in metformin-treated type 2 diabetic patients: a randomized, controlled trial. Diabetes Care. 2009;32(5):762–8. https://doi.org/10.2337/dc08-1797.
van Raalte DH, Bunck MC, Smits MM, Hoekstra T, Corner A, Diamant M, et al. Exenatide improves beta-cell function up to 3 years of treatment in patients with type 2 diabetes: a randomised controlled trial. Eur J Endocrinol. 2016;175(4):345–52. https://doi.org/10.1530/EJE-16-0286.
Ozcan U, Cao Q, Yilmaz E, Lee AH, Iwakoshi NN, Ozdelen E, et al. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science. 2004;306(5695):457–61. https://doi.org/10.1126/science.1103160.
Vilas-Boas EA, Almeida DC, Roma LP, Ortis F, Carpinelli AR. Lipotoxicity and beta-cell failure in type 2 diabetes: Oxidative stress linked to NADPH oxidase and ER stress. Cells. 2021. https://doi.org/10.3390/cells10123328.
Magkos F, Fraterrigo G, Yoshino J, Luecking C, Kirbach K, Kelly SC, et al. Effects of moderate and subsequent progressive weight loss on metabolic function and adipose tissue biology in humans with obesity. Cell Metab. 2016;23(4):591–601. https://doi.org/10.1016/j.cmet.2016.02.005.
Acknowledgements
This study was presented in part as a general poster session at the 78th Scientific Sessions of the American Diabetes Association, Orlando, Florida, June 22-26, 2018, and the 54th European Association for the Study of Diabetes Annual Meeting, Berlin, Germany, October 1-5, 2018.
Funding
This work was self-funded.
Author information
Authors and Affiliations
Contributions
All authors participated in study design, data analysis, interpretation of study results, drafting, revision, and approval of the final version of the manuscript. All authors accept responsibility for all aspects of this work.
Corresponding author
Ethics declarations
Conflict of interest
Yoshinobu Kondo received honoraria for lectures from Novo Nordisk Pharma. Shinobu Satoh received honoraria for lectures from Novo Nordisk Pharma and Eli Lilly. Yasuo Terauchi received honoraria for lectures from MSD; Ono Pharmaceutical; Nippon Boehringer Ingelheim; Mitsubishi Tanabe Pharma; Daiichi Sankyo; Kowa; Novo Nordisk Pharma; Eli Lilly; Sanofi; Sumitomo Pharma; Bayer, and AstraZeneca, and has obtained research support from Nippon Boehringer Ingelheim; Mitsubishi Tanabe Pharma; Daiichi Sankyo; Eli Lilly; Sanofi; and Sumitomo Pharma.
Human rights statement
The protocol for this study was approved by the Institutional Review Board of Yokohama City University (date of approval: November 24, 2016, approval No. B161110001) and Chigasaki Municipal Hospital (date of approval: November 18, 2016, approval No. 2016-09), and this study was also registered in the clinical trial registry (University hospital Medical Information Network Clinical Trials Registry 000024164). All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and/or with the Helsinki Declaration of 1964 and later versions.
Informed consent
All participants were fully informed of the aims and conduct of the study. Written informed consent was obtained from all participants prior to enrollment.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Kondo, Y., Satoh, S. & Terauchi, Y. Effects of dulaglutide and trelagliptin on beta-cell function in patients with type 2 diabetes: a randomized controlled study: DUET-beta study. Diabetol Int (2024). https://doi.org/10.1007/s13340-024-00717-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13340-024-00717-6