Abstract
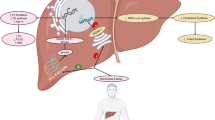
It is important to prevent not only diabetic complications but also diabetic comorbidities in diabetes care. We have elucidated multifaceted insulin action in various tissues mainly by means of model mice, and it was revealed that insulin regulates endoplasmic reticulum (ER) stress response during feeding, whereas ER stress ‘response failure’ contributes to the development of steatohepatitis, one of the major diabetic comorbidities. Insulin regulates gluconeogenesis not only in the liver but also in the proximal tubules of the kidney, which is also suppressed by reabsorbed glucose in the latter. In skeletal muscle, another important insulin-targeted tissue, impaired insulin/IGF-1 signaling leads not only to sarcopenia, an aging-related disease, but also to bone loss and shorter longevity. Aging is regulated by adipokines as well, and it is deemed to be accelerated by ‘imbalanced adipokines’ in combination with genetic background of progeria. Moreover, we reported effects of intensive multifactorial intervention on diabetic complications and mortality in patients with type 2 diabetes in a large-scale clinical trial, the J-DOIT3, followed by reports of subsequent sub-analyses of renal events and fracture events. Various approaches to elucidate the mechanisms underlying the development of diabetes and how it should be treated are expected to help us improve diabetes management.





Similar content being viewed by others
Data availability
Data sharing is not applicable to this article as no new data were created or analyzed in this review.
References
The Japan Diabetes Society Group. Treatment guide for diabetes 2022–2023. Tokyo: Bunkodo; 2022.
Kubota N, Kubota T, Itoh S, et al. Dynamic functional relay between insulin receptor substrate 1 and 2 in hepatic insulin signaling during fasting and feeding. Cell Metab. 2008;8(1):49–64.
Kadowaki T, Ueki K, Yamauchi T, Kubota N. SnapShot: insulin signaling pathways. Cell. 2012;148(3):624,e1.
Rinella ME, Lazarus JV, Ratziu V, et al. A multi-society Delphi consensus statement on new fatty liver disease nomenclature. J Hepatol. 2023. https://doi.org/10.1016/j.aohep.2023.101133. (In press).
Sasako T, Ohsugi M, Kubota N, et al. Hepatic Sdf2l1 controls feeding-induced ER stress and regulates metabolism. Nat Commun. 2019;10(1):947.
Park SW, Zhou Y, Lee J, et al. The regulatory subunits of PI3K, p85alpha and p85beta, interact with XBP-1 and increase its nuclear translocation. Nat Med. 2010;16(4):429–37.
Winnay JN, Boucher J, Mori MA, Ueki K, Kahn CR. A regulatory subunit of phosphoinositide 3-kinase increases the nuclear accumulation of X-box-binding protein-1 to modulate the unfolded protein response. Nat Med. 2010;16(4):438–45.
Fukuda S, Sumii M, Masuda Y, et al. Murine and human SDF2L1 is an endoplasmic reticulum stress-inducible gene and encodes a new member of the Pmt/rt protein family. Biochem Biophys Res Commun. 2001;280(1):407–14.
Day CP, James OF. Steatohepatitis: a tale of two “hits”? Gastroenterology. 1998;114(4):842–5.
Sasako T, Ueki K. ER stress response failure and steatohepatitis comorbid with diabetes. In: Psychology and patho-physiological outcomes of eating. IntechOpen Ltd; 2021. p. 69–83.
Tateishi R, Matsumura T, Okanoue T, et al. Hepatocellular carcinoma development in diabetic patients: a nationwide survey in Japan. J Gastroenterol. 2021;56(3):261–73.
Soeda K, Sasako T, Enooku K, et al. Gut insulin action protects from hepatocarcinogenesis in diabetic mice comorbid with nonalcoholic steatohepatitis. Nat Commun. 2023;14(1):6584.
Bhattarai KR, Chaudhary M, Kim HR, Chae HJ. Endoplasmic reticulum (ER) stress response failure in diseases. Trends Cell Biol. 2020;30(9):672–5.
Yagil C, Varadi-Levi R, Ifrach C, Yagil Y. Dysregulated UPR and ER stress related to a mutation in the Sdf2l1 gene are involved in the pathophysiology of diet-induced diabetes in the Cohen diabetic RAT. Int J Mol Sci. 2023;24(2):1355.
Sazonovs A, Stevens CR, Venkataraman GR, et al. Large-scale sequencing identifies multiple genes and rare variants associated with Crohn’s disease susceptibility. Nat Genet. 2022;54(9):1275–83.
Mather A, Pollock C. Glucose handling by the kidney. Kidney Int Suppl. 2011;120:S1-6.
Butlen D, Vadrot S, Roseau S, Morel F. Insulin receptors along the rat nephron: [125I] insulin binding in microdissected glomeruli and tubules. Pflugers Arch. 1988;412(6):604–12.
Tiwari S, Singh RS, Li L, et al. Deletion of the insulin receptor in the proximal tubule promotes hyperglycemia. J Am Soc Nephrol. 2013;24(8):1209–14.
Sasaki M, Sasako T, Kubota N, et al. Dual regulation of gluconeogenesis by insulin and glucose in the proximal tubules of the kidney. Diabetes. 2017;66(9):2339–50.
Rubera I, Poujeol C, Bertin G, et al. Specific Cre/Lox recombination in the mouse proximal tubule. J Am Soc Nephrol. 2004;15(8):2050–6.
Bingham M. In this issue of Diabetes. Diabetes. 2017;66(9):2325–6.
Packer M. Role of deranged energy deprivation signaling in the pathogenesis of cardiac and renal disease in states of perceived nutrient overabundance. Circulation. 2020;141(25):2095–105.
Liu H, Sridhar VS, Lovblom LE, et al. Markers of kidney injury, inflammation, and fibrosis associated with ertugliflozin in patients with CKD and diabetes. Kidney Int Rep. 2021;6(8):2095–104.
Sasako T, Tanaka T, Yamauchi T. Adaptive response as a potential key link between SGLT2 inhibition and renoprotection. Kidney Int Rep. 2021;6(8):2022–4.
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al. Sarcopenia: European consensus on definition and diagnosis: report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39(4):412–23.
Cruz-Jentoft AJ, Bahat G, Bauer J, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48(1):16–31.
Chen LK, Woo J, Assantachai P, et al. Asian working group for sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J Am Med Dir Assoc. 2020;21(3):300–7.
Campisi J, Kapahi P, Lithgow GJ, Melov S, Newman JC, Verdin E. From discoveries in ageing research to therapeutics for healthy ageing. Nature. 2019;571(7764):183–92.
Lee CG, Boyko EJ, Barrett-Connor E, et al. Insulin sensitizers may attenuate lean mass loss in older men with diabetes. Diabetes Care. 2011;34(11):2381–6.
Schiaffino S, Mammucari C. Regulation of skeletal muscle growth by the IGF1-Akt/PKB pathway: insights from genetic models. Skelet Muscle. 2011;1(1):4.
Jaiswal N, Gavin M, Loro E, et al. AKT controls protein synthesis and oxidative metabolism via combined mTORC1 and FOXO1 signalling to govern muscle physiology. J Cachexia Sarcopenia Muscle. 2022;13(1):495–514.
Sasako T, Umehara T, Soeda K, et al. Deletion of skeletal muscle Akt1/2 causes osteosarcopenia and reduces lifespan in mice. Nat Commun. 2022;13(1):5655.
Kirk B, Al Saedi A, Duque G. Osteosarcopenia: a case of geroscience. Aging Med (Milton). 2019;2(3):147–56.
[Press Release: Elucidating impact of imparied insulin action in diabetes on aging of skeletal muscle and systemic lonevity]. https://www.h.u-tokyo.ac.jp/participants/research/saishinkenkyu/__icsFiles/afieldfile/2022/10/03/release_20221005.pdf. Accessed 31 Oct 2023.
Tian J, Chung HK, Moon JS, et al. Skeletal muscle mitoribosomal defects are linked to low bone mass caused by bone marrow inflammation in male mice. J Cachexia Sarcopenia Muscle. 2022;13(3):1785–99.
Hosoi T, Yakabe M, Sasakawa H, et al. Sarcopenia phenotype and impaired muscle function in male mice with fast-twitch muscle-specific knockout of the androgen receptor. Proc Natl Acad Sci USA. 2023;120(4): e2218032120.
Sasako T, Ueki K. Sarcopenia: loss of mighty armor against frailty and aging. J Diabetes Investig. 2023;14(10):1145–7.
Yamauchi T, Kadowaki T. Adiponectin receptor as a key player in healthy longevity and obesity-related diseases. Cell Metab. 2013;17(2):185–96.
Sasako T, Kadowaki H, Fujiwara T, et al. Severe aortic stenosis during leptin replacement therapy in a patient with generalized lipodystrophy-associated progeroid syndrome due to an LMNA variant: a case report. J Diabetes Investig. 2022;13(9):1636–8.
Hosoe J, Miya F, Kadowaki H, et al. Clinical usefulness of multigene screening with phenotype-driven bioinformatics analysis for the diagnosis of patients with monogenic diabetes or severe insulin resistance. Diabetes Res Clin Pract. 2020;169: 108461.
Hussain I, Patni N, Ueda M, et al. A novel generalized lipodystrophy-associated progeroid syndrome due to recurrent heterozygous LMNA p.T10I mutation. J Clin Endocrinol Metab. 2018;103(3):1005–14.
[Case report of severe aortic stenosis develo** during leptin replacement therapy for a rare disease of lipidystrophy]. https://www.h.u-tokyo.ac.jp/participants/research/saishinkenkyu/20220708-1.html. Accessed 31 Oct 2023.
Seki Y, Yamada T, Kiyosue A, et al. Asymptomatic myocardial infarction in a patient with myotonic dystrophy type 1. J Cardiol Cases. 2022;26(4):248–51.
Nakagawa-Nagahama Y, Igarashi M, Miura M, Sasako T, Yamauchi T. A case of variant hemoglobin Hb Hoshida [β 43 Glu (GAG) → Gln (CAG) ] diagnosed at the onset of acute-onset type 1 diabetes mellitus. J Japan Diab Soc. 2022;65(9):505–11.
Sasako T, Yamauchi T. Clinical trials in participants with type 2 diabetes undertaken in Japan. J Japan Diab Soc. 2022;65(10):518–21.
Yazaki Y, Kadowaki T. Combating diabetes and obesity in Japan. Nat Med. 2006;12(1):73–4.
Japan Diabetes Optimal Integrated Treatment study for 3 major risk factors of cardiovascular diseases (J-DOIT3). https://clinicaltrials.gov/ct2/show/NCT00300976. Accessed 31 Oct 2023.
Ueki K, Sasako T, Kato M, et al. Design of and rationale for the Japan Diabetes Optimal Integrated Treatment study for 3 major risk factors of cardiovascular diseases (J-DOIT3): a multicenter, open-label, randomized, parallel-group trial. BMJ Open Diabetes Res Care. 2016;4(1): e000123.
Ueki K, Sasako T, Okazaki Y, et al. Effect of an intensified multifactorial intervention on cardiovascular outcomes and mortality in type 2 diabetes (J-DOIT3): an open-label, randomised controlled trial. Lancet Diabetes Endocrinol. 2017;5(12):951–64.
Study results. http://www.jdoit3.jp/en/result.html. Accessed 31 Oct 2023.
Sone H, Tanaka S, Iimuro S, et al. Long-term lifestyle intervention lowers the incidence of stroke in Japanese patients with type 2 diabetes: a nationwide multicentre randomised controlled trial (the Japan Diabetes Complications Study). Diabetologia. 2010;53(3):419–28.
Griffin SJ, Rutten G, Khunti K, et al. Long-term effects of intensive multifactorial therapy in individuals with screen-detected type 2 diabetes in primary care: 10-year follow-up of the ADDITION-Europe cluster-randomised trial. Lancet Diabetes Endocrinol. 2019;7(12):925–37.
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359(15):1577–89.
Sasako T, Kadowaki T, Ueki K. ADDITION-Europe: the first decade and beyond. Lancet Diabetes Endocrinol. 2019;7(12):891–3.
Ueki K, Sasako T, Okazaki Y, et al. Multifactorial intervention has a significant effect on diabetic kidney disease in patients with type 2 diabetes. Kidney Int. 2021;99(1):256–66.
Sasako T, Yamauchi T. Addressing screams for evidence on renoprotection by GLP-1 receptor agonists. Kidney Int. 2022;101(2):222–4.
Sasako T, Ueki K, Miyake K, et al. Effect of a multifactorial intervention on fracture in patients with type 2 diabetes: subanalysis of the J-DOIT3 study. J Clin Endocrinol Metab. 2021;106(5):e2116–28.
Chan JCN. How can we optimise diabetes care in real-world practice? Lancet Diabetes Endocrinol. 2017;5(12):927–9.
Gaede P, Lund-Andersen H, Parving HH, Pedersen O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med. 2008;358(6):580–91.
Sasako T, Yamauchi T, Ueki K. Intensified multifactorial intervention in patients with type 2 diabetes mellitus. Diabetes Metab J. 2023;47(2):185–97.
Acknowledgements
This review is a summary of my presentation in the Lilly Award Lecture at the 66th annual meeting of the Japan Diabetes Society, Kagoshima, Japan. I would like to express sincere gratitude to Dr. Takashi Kadowaki, Dr. Kazuyuki Tobe, Dr. Kohjiro Ueki, Dr. Naoto Kubota, and Dr. Toshimasa Yamauchi for their mentoring and to my colleagues and collaborators for their support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
I declare that I have no conflicts of interest.
Research involves human and animal participants
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Sasako, T. Exploring mechanisms underlying diabetes comorbidities and strategies to prevent vascular complications. Diabetol Int 15, 34–40 (2024). https://doi.org/10.1007/s13340-023-00677-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-023-00677-3