Abstract
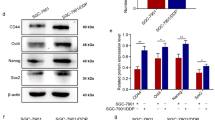
This work aims to evaluate the impact of 2-morpholino-8-phenyl-4H-chromen-4-one (LY294002) combined 5-fluorouracil (5-FU) for the activity of CD90+ liver cancer cells derived from the human liver cancer cell line MHCC97H. MHCC97H sphere-forming cells (MSFCs) were amplified in serum-free medium and CD90+ cells were isolated from bulk MSFCs using flow cytometry. The phenotype of these CD90+ cells which show liver cancer stem cells (LCSCs) behavior was validated in vitro and in a xenograft model in nude mice. MSFCs, CD90+ liver cancer cells (CD90+ LCCs), and parental MHCC97H cells were treated with no drug, LY294002 alone, 5-FU alone, or both drugs together and then compared in terms of stem cell-related gene expression, proliferation, and invasion. Stem cell phenotype increased with increasing proportion of CD90+ cells, in ascending order: parental MHCC97H cells, MSFCs, and CD90+ liver cancer cells. LY294002 reduced the expression of CD90, Nanog, SALL4, and SHP2 in a concentration-dependent manner in CD90+ LCCs and MSFCs, but not in parental cells. LY294002 blocked AKT phosphorylation via the PI3K/AKT signaling pathway and inhibited CD90+ LCCs proliferation and tumorigenicity in vitro and in vivo. CD90+ liver cancer cells can express liver cancer stem cell phenotype. LY294002 inhibits the proliferation and invasion of MHCC97H-derived CD90+ LCCs and sensitized CD90+ LCCs-derived tumors to 5-FU in the current study which may provide insight into the association between the LY294002 combined 5-FU and liver cancer stem cell (LCSCs).













Similar content being viewed by others
References
Dudeck O, Ricke J. Advances in regional chemotherapy of the liver. Expert Opin Drug Deliv. 2011;8(8):1057–69.
Zhong JH, Rodriguez AC, Ke Y, Wang YY, Wang L, Li LQ. Hepatic resection as a safe and effective treatment for hepatocellular carcinoma involving a single large tumor, multiple tumors, or macrovascular invasion. Medicine (Baltimore). 2015;94(3), e396.
Zhong JH, Ke Y, Gong WF, et al. Hepatic resection associated with good survival for selected patients with intermediate and advanced-stage hepatocellular carcinoma. Ann Surg. 2014;260(2):329–40.
Erhardt A, Kolligs F, Dollinger M, et al. TACE plus sorafenib for the treatment of hepatocellular carcinoma: results of the multicenter, phase II SOCRATES trial. Cancer Chemother Pharmacol. 2014;74(5):947–54.
**ao GQ, Song JL, Shen S, Yang JY, Yan LN. Living donor liver transplantation does not increase tumor recurrence of hepatocellular carcinoma compared to deceased donor transplantation. World J Gastroenterol. 2014;20(31):10953–9.
Fu QH, Zhang Q, Bai XL, et al. Sorafenib enhances effects of transarterial chemoembolization for hepatocellular carcinoma: a systematic review and meta-analysis. J Cancer Res Clin Oncol. 2014;140(8):1429–40.
Li X, Luo Q, Song G. Novel therapeutic strategies for treatment of hepatocellular carcinoma: targeting intervention on liver cancer stem cells. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2013;30(4):894–8.
Fornari C, Beccuti M, Lanzardo S, et al. A mathematical-biological joint effort to investigate the tumor-initiating ability of cancer stem cells. PLoS One. 2014;9(9):e106193.
Shafee N, Smith CR, Wei S, et al. Cancer stem cells contribute to cisplatin resistance in Brca1/p53-mediated mouse mammary tumors. Cancer Res. 2008;68(9):3243–50.
Svendsen A, Verhoeff JJ, Immervoll H, et al. Expression of the progenitor marker NG2/CSPG4 predicts poor survival and resistance to ionising radiation in glioblastoma. Acta Neuropathol. 2011;122(4):495–510.
Ignatova TN, Kukekov VG, Laywell ED, Suslov ON, Vrionis FD, Steindler DA. Human cortical glial tumors contain neural stem-like cells expressing astroglial and neuronal markers in vitro. Glia. 2002;39(3):193–206.
Ponti D, Costa A, Zaffaroni N, et al. Isolation and in vitro propagation of tumorigenic breast cancer cells with stem/progenitor cell properties. Cancer Res. 2005;65(13):5506–11.
Haley EM, Kim Y. The role of basic fibroblast growth factor in glioblastoma multiforme and glioblastoma stem cells and in their in vitro culture. Cancer Lett. 2014;346(1):1–5.
Yang ZF, Ho DW, Ng MN, et al. Significance of CD90+ cancer stem cells in human liver cancer. Cancer Cell. 2008;13(2):153–66.
Zhou J, Yi L, Ouyang Q, Xu L, Gui H, Xu M. Neurotensin signaling regulates stem-like traits of glioblastoma stem cells through activation of IL-8/CXCR1/STAT3 pathway. Cell Signal. 2014;26(12):2896–902.
Hajifathali A, Saba F, Atashi A, Soleimani M, Mortaz E, Rasekhi M. The role of catecholamines in mesenchymal stem cell fate. Cell Tissue Res. 2014;358(3):651–65.
Chen Q, Cao HZ, Zheng PS. LGR5 promotes the proliferation and tumor formation of cervical cancer cells through the Wnt/beta-catenin signaling pathway. Oncotarget. 2014;5(19):9092–105.
Bartman CM, Egelston J, Kattula S, et al. Gene expression profiling in mouse embryonic stem cells reveals glycogen synthase kinase-3-dependent targets of phosphatidylinositol 3-kinase and Wnt/beta-catenin signaling pathways. Front Endocrinol (Lausanne). 2014;5:133.
Sapey E, Greenwood H, Walton G, et al. Phosphoinositide 3-kinase inhibition restores neutrophil accuracy in the elderly: toward targeted treatments for immunosenescence. Blood. 2014;123(2):239–48.
Hua HW, Jiang F, Huang Q, Liao ZJ, Ding G. Re-sensitization of 5-FU resistance by SPARC through negative regulation of glucose metabolism in hepatocellular carcinoma. Tumour Biol. 2015;36(1):303–13.
Sukowati CH, Anfuso B, Torre G, Francalanci P, Crocè LS, Tiribelli C. The expression of CD90/Thy-1 in hepatocellular carcinoma: an in vivo and in vitro study. PLoS One. 2013;8(10), e76830.
**ang Q, Ma Y, Dong J, Shen R. Carnosic acid induces apoptosis associated with mitochondrial dysfunction and Akt inactivation in HepG2 cells. Int J Food Sci Nutr. 2014:1–9.
Deonarain MP, Kousparou CA, Epenetos AA. Antibodies targeting cancer stem cells: a new paradigm in immunotherapy? MAbs. 2009;1(1):12–25.
Singh BN, Kumar D, Shankar S, Srivastava RK. Rottlerin induces autophagy which leads to apoptotic cell death through inhibition of PI3K/Akt/mTOR pathway in human pancreatic cancer stem cells. Biochem Pharmacol. 2012;84(9):1154–63.
Torigoe T, Hirohashi Y, Yasuda K, Sato N. Constitutive expression and activation of stress response genes in cancer stem-like cells/tumour initiating cells: potent targets for cancer stem cell therapy. Int J Hyperthermia. 2013;29(5):436–41.
Zhang XL, Jia Q, Lv L, Deng T, Gao J. Tumorspheres derived from HCC cells are enriched with cancer stem cell-like cells and present high chemoresistance dependent on the Akt pathway. Anticancer Agents Med Chem. 2015.
Yan X, Luo H, Zhou X, Zhu B, Wang Y, Bian X. Identification of CD90 as a marker for lung cancer stem cells in A549 and H446 cell lines. Oncol Rep. 2013;30(6):2733–40.
Tang KH, Dai YD, Tong M, et al. A CD90(+) tumor-initiating cell population with an aggressive signature and metastatic capacity in esophageal cancer. Cancer Res. 2013;73(7):2322–32.
Guo Z, Li LQ, Jiang JH, Ou C, Zeng LX, **ang BD. Cancer stem cell markers correlate with early recurrence and survival in hepatocellular carcinoma. World J Gastroenterol. 2014;20(8):2098–106.
Yang J, Gao C, Chai L, Ma Y. A novel SALL4/OCT4 transcriptional feedback network for pluripotency of embryonic stem cells. PLoS One. 2010;5(5), e10766.
Oishi N, Yamashita T, Kaneko S. Molecular biology of liver cancer stem cells. Liver Cancer. 2014;3(2):71–84.
Aceto N, Sausgruber N, Brinkhaus H, et al. Tyrosine phosphatase SHP2 promotes breast cancer progression and maintains tumor-initiating cells via activation of key transcription factors and a positive feedback signaling loop. Nat Med. 2012;18(4):529–37.
Bard-Chapeau EA, Li S, Ding J, et al. Ptpn11/Shp2 acts as a tumor suppressor in hepatocellular carcinogenesis. Cancer Cell. 2011;19(5):629–39.
Lim CY, Tam WL, Zhang J, et al. Sall4 regulates distinct transcription circuitries in different blastocyst-derived stem cell lineages. Cell Stem Cell. 2008;3(5):543–54.
Zhao K, Zhu BS, Gong W, et al. SN50 enhances the effects of LY294002 on cell death induction in gastric cancer cell line SGC7901. Arch Med Sci. 2013;9(6):990–8.
Luo X, Dong Z, Chen Y, Yang L, Lai D. Enrichment of ovarian cancer stem-like cells is associated with epithelial to mesenchymal transition through an miRNA-activated AKT pathway. Cell Prolif. 2013;46(4):436–46.
Zeng YP, Yang ZR, Guo XF, Jun W, Dong WG. Synergistic effect of puerarin and 5-fluorouracil on hepatocellular carcinoma. Oncol Lett. 2014;8(6):2436–42.
Huang XW, Tang ZY, Lawrence TS, Zhang M. 5-Fluorouracil and hydroxyurea enhance adenovirus-mediated transgene expression in colon and hepatocellular carcinoma cells. J Cancer Res Clin Oncol. 2005;131(3):184–90.
Kim JS, Kim BS, Kim J, Park CS, Chung IY. The phosphoinositide-3-kinase/Akt pathway mediates the transient increase in Nanog expression during differentiation of F9 cells. Arch Pharm Res. 2010;33(7):1117–25.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant No. 81160262 and 81260088) and the Guangxi Cancer Institute and Key Laboratory of High-Incidence-Tumor early Prevention and Treatment, Ministry of Education.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
None
Ethical standards
This study was approved by the Ethics Committee of the Affiliated Tumor Hospital of Guangxi Medical University; it was performed according to the Declaration of Helsinki 2013 edition. Animal experiments were carried out in compliance with standard protocols approved by the Ethical Committee of Guangxi Medical University and the Committee of Experimental Animal Feeding and Management.
Additional information
Yu-Chong Peng, Shi-Dong Lu and Jian-Hong Zhong contributed equally to this work.
About this article
Cite this article
Peng, YC., Lu, SD., Zhong, JH. et al. Combination of 5-fluorouracil and 2-morphilino-8-phenyl-4H-chromen-4-one may inhibit liver cancer stem cell activity. Tumor Biol. 37, 10943–10958 (2016). https://doi.org/10.1007/s13277-016-4915-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-016-4915-3