Abstract
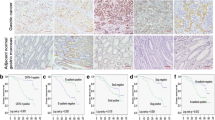
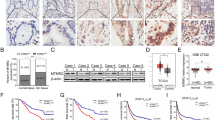
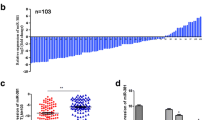
Membrane type 1-matrix metalloproteinase (MT1-MMP) has been identified to play a significant role in several types of cancers, but little is known about the significance of MT1-MMP in gastric cancer patients. The purpose of this study is to investigate the involvement of MT1-MMP in tumor progression of gastric cancer. MT1-MMP expression levels were examined in gastric cancer tissues and cells, and normal gastric tissues and cells. The effects and molecular mechanisms of MT1-MMP expression on cell proliferation, migration, and invasion were also explored. In our results, MT1-MMP messenger RNA (mRNA) and protein expression levels were significantly increased in gastric cancer tissue. Moreover, the overexpression of MT1-MMP was positively associated with the status of clinical stage and lymph node metastasis through real-time PCR. Furthermore, knocking down MT1-MMP expression significantly suppressed the cell migration and invasion in vitro and regulated the expression of MMPs and epithelial-mesenchymal transition (EMT)-associated genes. In conclusions, our study demonstrates that MT1-MMP was overexpressed in gastric cancer tissue, and reduced expression of MT1-MMP suppressed cell migration, invasion, and through regulating the expression of MMPs and the process of EMT in gastric cancer.



Similar content being viewed by others
References
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64:9–29.
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: Globocan 2008. Int J Cancer J Int Cancer. 2010;127:2893–917.
Jang BG, Kim WH. Molecular pathology of gastric carcinoma. Pathobiol J Immunopathol Mol Cell Biol. 2011;78:302–10.
Ohtsu A. Chemotherapy for metastatic gastric cancer: past, present, and future. J Gastroenterol. 2008;43:256–64.
Pahwa S, Stawikowski MJ, Fields GB. Monitoring and inhibiting MT1-MMP during cancer initiation and progression. Cancers. 2014;6:416–35.
Suojanen J, Salo T, Koivunen E, Sorsa T, Pirila E. A novel and selective membrane type-1 matrix metalloproteinase (MT1-MMP) inhibitor reduces cancer cell motility and tumor growth. Cancer Biol Ther. 2009;8:2362–70.
Wang X, Wilson MJ, Slaton JW, Sinha AA, Ewing SL, Pei D. Increased aggressiveness of human prostate PC-3 tumor cells expressing cell surface localized membrane type-1 matrix metalloproteinase (MT1-MMP). J Androl. 2009;30:259–74.
Akanuma N, Hoshino I, Akutsu Y, Murakami K, Isozaki Y, Maruyama T, et al. MicroRNA-133a regulates the mRNAs of two invadopodia-related proteins, FSCN1 and MMP14, in esophageal cancer. Br J Cancer. 2014;110:189–98.
Xu M, Wang YZ. miR-133a suppresses cell proliferation, migration and invasion in human lung cancer by targeting MMP-14. Oncol Rep. 2013;30:1398–404.
Liu Z, Yang H, Luo W, Jiang Q, Mai C, Chen Y, et al. Loss of cytoplasmic KLF4 expression is correlated with the progression and poor prognosis of nasopharyngeal carcinoma. Histopathology. 2013;63:362–70.
Ulasov I, Yi R, Guo D, Sarvaiya P, Cobbs C. The emerging role of MMP14 in brain tumorigenesis and future therapeutics. Biochim Biophys Acta. 1846;2014:113–20.
Zhou H, Wu A, Fu W, Lv Z, Zhang Z. Significance of semaphorin-3A and MMP-14 protein expression in non-small cell lung cancer. Oncol Lett. 2014;7:1395–400.
Atkinson JM, Pennington CJ, Martin SW, Anikin VA, Mearns AJ, Loadman PM, et al. Membrane type matrix metalloproteinases (MMPs) show differential expression in non-small cell lung cancer (NSCLC) compared to normal lung: correlation of MMP-14 mRNA expression and proteolytic activity. Eur J Cancer (Oxford, England : 1990). 2007;43:1764–71.
Haage A, Nam DH, Ge X, Schneider IC. Matrix metalloproteinase-14 is a mechanically regulated activator of secreted MMPs and invasion. Biochem Biophys Res Commun. 2014;450:213–8.
Udayakumar TS, Chen ML, Bair EL, Von Bredow DC, Cress AE, Nagle RB, et al. Membrane type-1-matrix metalloproteinase expressed by prostate carcinoma cells cleaves human laminin-5 beta3 chain and induces cell migration. Cancer Res. 2003;63:2292–9.
Munaut C, Noel A, Hougrand O, Foidart JM, Boniver J, Deprez M. Vascular endothelial growth factor expression correlates with matrix metalloproteinases MT1-MMP, MMP-2 and MMP-9 in human glioblastomas. Int J Cancer J Int Cancer. 2003;106:848–55.
Wang YZ, Wu KP, Wu AB, Yang ZC, Li JM, Mo YL, et al. MMP-14 overexpression correlates with poor prognosis in non-small cell lung cancer. Tumour Biol J Int Soc Oncodev Biol Med. 2014.
He L, Chu D, Li X, Zheng J, Liu S, Li J, et al. Matrix metalloproteinase-14 is a negative prognostic marker for patients with gastric cancer. Dig Dis Sci. 2013;58:1264–70.
Peng CW, Wang LW, Fang M, Yang GF, Li Y, Pang DW. Combined features based on MT1-MMP expression, CD11b + immunocytes density and LNR predict clinical outcomes of gastric cancer. J Transl Med. 2013;11:153.
Tetu B, Brisson J, Wang CS, Lapointe H, Beaudry G, Blanchette C, et al. The influence of MMP-14, TIMP-2 and MMP-2 expression on breast cancer prognosis. Breast Cancer Res BCR. 2006;8:R28.
Wang L, Yuan J, Tu Y, Mao X, He S, Fu G, et al. Co-expression of MMP-14 and MMP-19 predicts poor survival in human glioma. Clin Transl Oncol Off Publ Fed Spanish Oncol Soc Natl Cancer Inst Mexico. 2013;15:139–45.
Yang B, Gao J, Rao Z, Shen Q. Clinicopathological and prognostic significance of α5β1-integrin and MMP-14 expressions in colorectal cancer. Neoplasma. 2013;60:254–61.
Trudel D, Desmeules P, Turcotte S, Plante M, Gregoire J, Renaud MC, et al. Visual and automated assessment of matrix metalloproteinase-14 tissue expression for the evaluation of ovarian cancer prognosis. Mod Pathol Off J US Can Acad Pathol Inc. 2014;27:1394–404.
Trudel D, Fradet Y, Meyer F, Harel F, Tetu B. Membrane-type-1 matrix metalloproteinase, matrix metalloproteinase 2, and tissue inhibitor of matrix proteinase 2 in prostate cancer: identification of patients with poor prognosis by immunohistochemistry. Hum Pathol. 2008;39:731–9.
Wu KP, Li Q, Lin FX, Li J, Wu LM, Li W, et al. MT1-MMP is not a good prognosticator of cancer survival: evidence from 11 studies. Tumour Biol J Int Soc Oncodev Biol Med. 2014.
Nishida Y, Miyamori H, Thompson EW, Takino T, Endo Y, Sato H. Activation of matrix metalloproteinase-2 (MMP-2) by membrane type 1 matrix metalloproteinase through an artificial receptor for proMMP-2 generates active MMP-2. Cancer Res. 2008;68:9096–104.
Sato H, Takino T, Okada Y, Cao J, Shinagawa A, Yamamoto E, et al. A matrix metalloproteinase expressed on the surface of invasive tumour cells. Nature. 1994;370:61–5.
Sato H, Takino T, Miyamori H. Roles of membrane-type matrix metalloproteinase-1 in tumor invasion and metastasis. Cancer Sci. 2005;96:212–7.
McGowan PM, Duffy MJ. Matrix metalloproteinase expression and outcome in patients with breast cancer: analysis of a published database. Ann Oncol Off J Eur Soc Med Oncol / ESMO. 2008;19:1566–72.
Chellaiah MA, Ma T. Membrane localization of membrane type 1 matrix metalloproteinase by CD44 regulates the activation of pro-matrix metalloproteinase 9 in osteoclasts. BioMed Res Int. 2013;2013:302392.
Knauper V, Will H, Lopez-Otin C, Smith B, Atkinson SJ, Stanton H, et al. Cellular mechanisms for human procollagenase-3 (MMP-13) activation. Evidence that MT1-MMP (MMP-14) and gelatinase a (MMP-2) are able to generate active enzyme. J Biol Chem. 1996;271:17124–31.
Yang CC, Zhu LF, Xu XH, Ning TY, Ye JH, Liu LK. Membrane type 1 matrix metalloproteinase induces an epithelial to mesenchymal transition and cancer stem cell-like properties in SCC9 cells. BMC Cancer. 2013;13:171.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Wenfeng Li and Shouzhi Li are co-first authors.
Rights and permissions
About this article
Cite this article
Li, W., Li, S., Deng, L. et al. Decreased MT1-MMP in gastric cancer suppressed cell migration and invasion via regulating MMPs and EMT. Tumor Biol. 36, 6883–6889 (2015). https://doi.org/10.1007/s13277-015-3381-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3381-7