Abstract
Background
There is high mortality rate and poor prognosis in lung cancer, especially non-small-cell lung cancer (NSCLC). Recent study showed that concurrent classic driver oncogene mutation with ROS1 rearrangement was found in NSCLC patients. However, whether this would affect the development and prognosis of NSCLC is still unclear.
Objective
To explore the clinical characteristics and prognosis of NSCLC patients harboring concurrent classic driver oncogene mutation with ROS1 rearrangement.
Methods
A retrospective study was conducted on 220 patients diagnosed with NSCLC. All samples were screened for EGFR and KRAS using amplification-refractory mutation system assay, and for ALK, ROS1 using RT-PCR. The clinical characteristics and clinical outcomes of concurrent gene alterations with ROS1 rearrangement were analyzed.
Results
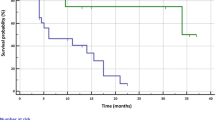
In 220 patients, 12 (5.45%) were ROS1 rearrangement, who tend to be younger, non-smokers. The mutation rates of EGFR, KRAS, ALK and ROS1 in NSCLC were 28.64%, 1.82%, 3.64% and 5.45%, respectively. ROS1 rearrangement was identified to co-occur in 5 (2.27%) NSCLC patients. ROS1/EGFR co-alterations were found in 3.17% of NSCLC patients, 16.67% of ROS1-positive NSCLC patients. Concomitant ROS1/ALK rearrangement constituted 37.50% in ALK-positive patients, and 25.00% in ROS1-positive patients. SDC4-ROS1 was the most common fusion partner in concurrent ROS1 rearrangement patients. The median overall survival of NSCLC with concurrent ROS1 rearrangement group and single ROS1 rearrangement group were 25 months and 14 months.
Conclusion
Concurrent driver oncogenes mutation with ROS1 rearrangement defines a unique subgroup of NSCLC. Patients with concomitant ROS1 rearrangement might have a better prognosis.



Similar content being viewed by others
References
Acquaviva J, Wong R, Charest A (2009) The multifaceted roles of the receptor tyrosine kinase ROS in development and cancer. Biochim Biophys Acta 1795:37–52
Bergethon K, Shaw AT, Ou SH, Katayama R, Lovly CM, McDonald NT, Massion PP, Siwak-Tapp C, Gonzalez A, Fang R et al (2012) ROS1 rearrangements define a unique molecular class of lung cancers. J Clin Oncol 30:863–870
Birchmeier C, Sharma S, Wigler M (1987) Expression and rearrangement of the ROS1 gene in human glioblastoma cells. Proc Natl Acad Sci U S A 84:9270–9274
Cai W, Zhou C, Ren S, Chen X, Gao G, Li X, Li W, Zhao C (2014) Coexistence of Egfr mutations with Alk, Ros1 or Ret rearrangements in lung adenocarcinoma: a clinicopathological analysis from 646 Chinese patients with lung adenocarcinoma. Ann Oncol 25:iv69
Chen YF, Hsieh MS, Wu SG, Chang YL, Shih JY, Liu YN, Tsai MF, Tsai TH, Yu CJ, Yang JC et al (2014) Clinical and the prognostic characteristics of lung adenocarcinoma patients with ROS1 fusion in comparison with other driver mutations in East Asian populations. J Thorac Oncol 9:1171–1179
Cui M, Han Y, Li P, Zhang J, Ou Q, Tong X, Zhao R, Dong N, Wu X, Li W et al (2020) Molecular and clinicopathological characteristics of ROS1-rearranged non-small-cell lung cancers identified by next-generation sequencing. Mol Oncol 14:2787–2795
Davies KD, Le AT, Theodoro MF, Skokan MC, Aisner DL, Berge EM, Terracciano LM, Cappuzzo F, Incarbone M, Roncalli M et al (2012) Identifying and targeting ROS1 gene fusions in non-small cell lung cancer. Clin Cancer Res 18:4570–4579
Davies KD, Le AT, Sheren J, Nijmeh H, Gowan K, Jones KL, Varella-Garcia M, Aisner DL, Doebele RC (2018) Comparison of molecular testing modalities for detection of ROS1 rearrangements in a cohort of positive patient samples. J Thorac Oncol 13:1474–1482
Gainor JF, Shaw AT (2013) Novel targets in non-small cell lung cancer: ROS1 and RET fusions. Oncologist 18:865–875
Gibelin C, Avrillon V, De La Fouchardiere A, Mc Leer-Florin A, Lantuejoul S, Fayette J (2016) Clinical relevance of ROS1 rearrangements detection in advanced squamous cell carcinomas. Lung Cancer 102:42–43
Jao K, Tomasini P, Kamel-Reid S, Korpanty GJ, Mascaux C, Sakashita S, Labbe C, Leighl NB, Liu G, Feld R et al (2018) The prognostic effect of single and multiple cancer-related somatic mutations in resected non-small-cell lung cancer. Lung Cancer 123:22–29
Kim HR, Lim SM, Kim HJ, Hwang SK, Park JK, Shin E, Bae MK, Ou SH, Wang J, Jewell SS et al (2013) The frequency and impact of ROS1 rearrangement on clinical outcomes in never smokers with lung adenocarcinoma. Ann Oncol 24:2364–2370
Li Q, Wu J, Yan LX, Huang JW, Zhang Z, Zhang JE, Gao XL, Luo ZR, Liu J, Yang SF et al (2017) ALK and ROS1 double-rearranged lung squamous cell carcinoma responding to Crizotinib treatment: a case report. J Thorac Oncol 12:e193–e197
Li X, Cai W, Yang G, Su C, Ren S, Zhao C, Hu R, Chen X, Gao G, Guo Z et al (2017) Comprehensive analysis of EGFR-mutant abundance and its effect on efficacy of EGFR TKIs in advanced NSCLC with EGFR mutations. J Thorac Oncol 12:1388–1397
Lin JJ, Ritterhouse LL, Ali SM, Bailey M, Schrock AB, Gainor JF, Ferris LA, Mino-Kenudson M, Miller VA, Iafrate AJ et al (2017) ROS1 fusions rarely overlap with other oncogenic drivers in non-small cell lung cancer. J Thorac Oncol 12:872–877
Liu C, Yu H, Chang J, Chen H, Li Y, Zhao W, Zhao K, Zhu Z, Sun S, Fan M et al (2019) Crizotinib in Chinese patients with ROS1-rearranged advanced nonsmall-cell lung cancer in routine clinical practice. Target Oncol 14:315–323
Lou NN, Zhang XC, Chen HJ, Zhou Q, Yan LX, **e Z, Su J, Chen ZH, Tu HY, Yan HH et al (2016) Clinical outcomes of advanced non-small-cell lung cancer patients with EGFR mutation, ALK rearrangement and EGFR/ALK co-alterations. Oncotarget 7:65185–65195
Lynch TJ, Bell DW, Sordella R, Gurubhagavatula S, Okimoto RA, Brannigan BW, Harris PL, Haserlat SM, Supko JG, Haluska FG et al (2004) Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med 350:2129–2139
Mao Y, Wu S (2017) ALK and ROS1 concurrent with EGFR mutation in patients with lung adenocarcinoma. Onco Targets Ther 10:3399–3404
Nagarajan L, Louie E, Tsujimoto Y, Balduzzi PC, Huebner K, Croce CM (1986) The human c-ros gene (ROS) is located at chromosome region 6q16----6q22. Proc Natl Acad Sci U S A 83:6568–6572
Paez JG, Janne PA, Lee JC, Tracy S, Greulich H, Gabriel S, Herman P, Kaye FJ, Lindeman N, Boggon TJ et al (2004) EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 304:1497–1500
Rikova K, Guo A, Zeng Q, Possemato A, Yu J, Haack H, Nardone J, Lee K, Reeves C, Li Y et al (2007) Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell 131:1190–1203
Sato S, Nagahashi M, Koike T, Ichikawa H, Shimada Y, Watanabe S, Kikuchi T, Takada K, Nakanishi R, Oki E et al (2018) Impact of concurrent genomic alterations detected by comprehensive genomic sequencing on clinical outcomes in east-Asian patients with EGFR-mutated lung adenocarcinoma. Sci Rep 8:1005
Shaw AT, Ou SH, Bang YJ, Camidge DR, Solomon BJ, Salgia R, Riely GJ, Varella-Garcia M, Shapiro GI, Costa DB et al (2014) Crizotinib in ROS1-rearranged non-small-cell lung cancer. N Engl J Med 371:1963–1971
Song Z, Zheng Y, Wang X, Su H, Zhang Y, Song Y (2017) ALK and ROS1 rearrangements, coexistence and treatment in epidermal growth factor receptor-wild type lung adenocarcinoma: a multicenter study of 732 cases. J Thorac Dis 9:3919–3926
Wen YS, Cai L, Zhang XW, Zhu JF, Zhang ZC, Shao JY, Zhang LJ (2014) Concurrent oncogene mutation profile in Chinese patients with stage Ib lung adenocarcinoma. Med (Baltim) 93:e296
Wiesweg M, Eberhardt WEE, Reis H, Ting S, Savvidou N, Skiba C, Herold T, Christoph DC, Meiler J, Worm K et al (2017) High prevalence of concomitant oncogene mutations in prospectively identified patients with ROS1-positive metastatic lung cancer. J Thorac Oncol 12:54–64
Wu J, Lin Y, He X, Yang H, He P, Fu X, Li G, Gu X (2016) Comparison of detection methods and follow-up study on the tyrosine kinase inhibitors therapy in non-small cell lung cancer patients with ROS1 fusion rearrangement. BMC Cancer 16:599
Yan X, Wang H, Li P, Zhang G, Zhang M, Yang J, Zhang X, Zheng X, Ma Z (2019) Efficacy of first-line treatment with epidermal growth factor receptor-tyrosine kinase inhibitor (EGFR-TKI) alone or in combination with chemotherapy for advanced non-small cell lung cancer (NSCLC) with low-abundance mutation. Lung Cancer 128:6–12
Yang JJ, Zhang XC, Su J, Xu CR, Zhou Q, Tian HX, **e Z, Chen HJ, Huang YS, Jiang BY et al (2014) Lung cancers with concomitant EGFR mutations and ALK rearrangements: diverse responses to EGFR-TKI and crizotinib in relation to diverse receptors phosphorylation. Clin Cancer Res 20:1383–1392
Yoshida A, Kohno T, Tsuta K, Wakai S, Arai Y, Shimada Y, Asamura H, Furuta K, Shibata T, Tsuda H (2013) ROS1-rearranged lung cancer: a clinicopathologic and molecular study of 15 surgical cases. Am J Surg Pathol 37:554–562
Zhao W, Choi YL, Song JY, Zhu Y, Xu Q, Zhang F, Jiang L, Cheng J, Zheng G, Mao M (2016) ALK, ROS1 and RET rearrangements in lung squamous cell carcinoma are very rare. Lung Cancer 94:22–27
Zhao Y, Wang S, Zhang B, Qiao R, Xu J, Zhang L, Zhang Y, Han B (2019) Clinical management of non-small cell lung cancer with concomitant EGFR mutations and ALK rearrangements: efficacy of EGFR tyrosine kinase inhibitors and Crizotinib. Target Oncol 14:169–178
Zhou Q, Zhang XC, Chen ZH, Yin XL, Yang JJ, Xu CR, Yan HH, Chen HJ, Su J, Zhong WZ et al (2011) Relative abundance of EGFR mutations predicts benefit from gefitinib treatment for advanced non-small-cell lung cancer. J Clin Oncol 29:3316–3321
Zhu YC, Xu CW, Ye XQ, Yin MX, Zhang JX, Du KQ, Zhang ZH, Hu J (2016) Lung cancer with concurrent EGFR mutation and ROS1 rearrangement: a case report and review of the literature. Onco Targets Ther 9:4301–4305
Acknowledgements
This work was supported by national public health project (201402024). Special thanks will give to the colleges in the laboratory for kindly providing help in the current study.
Author information
Authors and Affiliations
Contributions
DDL, FGJ and LP conceived and designed the study. HJ and LZ analyzed the data. YHX and CML wrote the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Dandan Li, Hua Jiang, Faguang **, Lei Pan, Yonghong **e, Liang Zhang and Chunmei Li declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, D., Jiang, H., **, F. et al. Concurrent classic driver oncogenes mutation with ROS1 rearrangement predicts superior clinical outcome in NSCLC patients. Genes Genom 45, 93–102 (2023). https://doi.org/10.1007/s13258-022-01326-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-022-01326-w