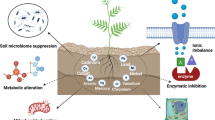
Abstract
The translocation of heavy metals (HMs) from the rhizosphere to plant systems constitutes a fundamental mechanism governing HM uptake. Microbial augmentation has emerged as a promising strategy to enhance this process. The study investigates the mechanism of enhanced translocation of heavy metals (HMs) from artificially polluted soil to Chrysopogon zizanioides, facilitated by Bacillus xiamenensis VITMSJ3. Pb, Ni, and Cd translocation to the roots and shoots of C. zizanioides was examined, revealing a significant increase of over 15% in HM uptake upon treatment with Bacillus xiamenensis VITMSJ3 (Accession number MT822866). VITMSJ3 exhibited biofilm formation capabilities, attributed to quorum sensing molecule production, and demonstrated resistance to Pb and Ni upto 4000 ppm and Cd upto 450 ppm, respectively. Moreover, VITMSJ3 displayed plant growth-promoting bacterial (PGPB) traits such as, indole-3-acetic acid (IAA), phosphate, ammonia, siderophore, and hydrogen cyanide (HCN) production. Amplification of candidate genes responsible for HM resistance (pbr for Pb, ncc for Ni, cadA for Cd) corroborated the genetic basis of resistance. SEM-EDAX micrographs confirmed HM uptake and translocation along with the presence of VITMSJ3. Enzymatic analysis revealed the synthesis of superoxide dismutase (SOD), catalase (CAT), glutathione-S-transferase (GST), peroxidase (POD), and ascorbate peroxidase (APX), implicating their involvement in ROS detoxification. Overall, the study underscores the efficacy of B. xiamenensis VITMSJ3 in enhancing HM translocation, thereby elucidating its potential for phytoremediation applications.











Similar content being viewed by others
Data availability
Supporting data for the experimental study is mentioned in the supplementary file.
References
Abd El-Rahman AF, Shaheen HA, Abd El-Aziz RM, Ibrahim DSS (2019) Influence of hydrogen cyanide-producing rhizobacteria in controlling the crown gall and root-knot nematode, Meloidogyne incognita. Egypt J Biol Pest Control. https://doi.org/10.1186/s41938-019-0143-7
Abou-Shanab RAI, van Berkum P, Angle JS (2007) Heavy metal resistance and genotypic analysis of metal resistance genes in gram-positive and gram-negative bacteria present in Ni-rich serpentine soil and in the rhizosphere of Alyssum murale. Chemosphere 68:360–367. https://doi.org/10.1016/j.chemosphere.2006.12.051
Aebi H (1984) [13] Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Afridi MS, Amna S et al (2019) Induction of tolerance to salinity in wheat genotypes by plant growth promoting endophytes: Involvement of ACC deaminase and antioxidant enzymes. Plant Physiol Biochem 139:569–577. https://doi.org/10.1016/j.plaphy.2019.03.041
Ali A, Guo D, Li Y et al (2021) Streptomyces pactum addition to contaminated mining soils improved soil quality and enhanced metals phytoextraction by wheat in a green remediation trial. Chemosphere 273:2. https://doi.org/10.1016/j.chemosphere.2021.129692
Amna XY, Farooq MA et al (2020) Multi-stress tolerant PGPR Bacillus xiamenensis PM14 activating sugarcane (Saccharum officinarum L.) red rot disease resistance. Plant Physiol Biochem 151:640–649. https://doi.org/10.1016/j.plaphy.2020.04.016
Arnon DI, Physiology SP, Jan N (2015) Copper enzymes in isolated chloroplasts . Polyphenoloxidase in Beta vulgaris Published by : American Society of Plant Biologists ( ASPB ). 24:1–15
Ashraf U, Tang X (2017) Yield and quality responses, plant metabolism and metal distribution pattern in aromatic rice under lead (Pb) toxicity. Chemosphere 176:141–155. https://doi.org/10.1016/j.chemosphere.2017.02.103
Baig KS, Arshad M, Khalid A et al (2014) Improving growth and yield of maize through bioinoculants carrying auxin production and phosphate solubilizing activity. Soil Environ 33:159–168
Borremans B, Hobman JL, Provoost A et al (2001) Cloning and functional analysis of the pbr lead resistance determinant of Ralstonia metallidurans CH34. J Bacteriol 183:5651–5658. https://doi.org/10.1128/JB.183.19.5651-5658.2001
Brown RW, Chadwick DR, Bending GD et al (2022) Nutrient (C, N and P) enrichment induces significant changes in the soil metabolite profile and microbial carbon partitioning. Soil Biol Biochem 172:108779. https://doi.org/10.1016/j.soilbio.2022.108779
Buragohain T, Dey P, Osborne WJ (2023) Food Bioscience In vitro studies on the inhibition of microbial pathogens by PPDHMP synthesized by Bacillus sp an endophyte of Citrus lemon (Kaji nemu). Food Biosci 55:103003. https://doi.org/10.1016/j.fbio.2023.103003
Cenkci S, Ciǧerci IH, Yildiz M et al (2010) Lead contamination reduces chlorophyll biosynthesis and genomic template stability in Brassica rapa L. Environ Exp Bot 67:467–473. https://doi.org/10.1016/j.envexpbot.2009.10.001
Chen T, Zhang B (2016) Measurements of proline and malondialdehyde content and antioxidant enzyme activities in leaves of drought stressed cotton. Bio Protocol 6:2. https://doi.org/10.21769/bioprotoc.1913
Chen Y, Shen Z, Li X (2004) The use of vetiver grass (Vetiveria zizanioides) in the phytoremediation of soils contaminated with heavy metals. Appl Geochem 19:1553–1565. https://doi.org/10.1016/j.apgeochem.2004.02.003
Daraz U, Li Y, Sun Q et al (2021) Inoculation of Bacillus spp. modulate the soil bacterial communities and available nutrients in the rhizosphere of vetiver plant irrigated with acid mine drainage. Chemosphere 263:128345. https://doi.org/10.1016/j.chemosphere.2020.128345
Das A, Osborne JW (2018a) Monitoring the stress resistance of Pennisetum purpureum in Pb (II) contaminated soil bioaugmented with Enterobacter cloacae as defence strategy. Chemosphere 210:495–502. https://doi.org/10.1016/j.chemosphere.2018.07.050
Das A, Osborne JW (2018b) Enhanced lead uptake by an association of plant and earthworm bioaugmented with bacteria. Pedosphere 28:311–322. https://doi.org/10.1016/S1002-0160(18)60021-9
Das J, Cross G, Qu C et al (2015) Devices, systems, and methods for automated monitoring enabling precision agriculture. IEEE Int Conf Autom Sci Eng 2015:462–469. https://doi.org/10.1109/CoASE.2015.7294123
Das A, Belgaonkar P, Raman AS et al (2017) Bioremoval of lead using Pennisetum purpureum augmented with Enterobacter cloacae-VITPASJ1: a pot culture approach. Environ Sci Pollut Res 24:15444–15453. https://doi.org/10.1007/s11356-017-8988-3
Demiral T, Türkan I (2005) Comparative lipid peroxidation, antioxidant defense systems and proline content in roots of two rice cultivars differing in salt tolerance. Environ Exp Bot 53:247–257. https://doi.org/10.1016/j.envexpbot.2004.03.017
Dereeper A, Guignon V, Blanc G et al (2008) Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res 36:2. https://doi.org/10.1093/nar/gkn180
Dey P, Buragohain T, Osborne WJ (2023) Inhibition of microbial pathogens and toxicity assessment of noformicin synthesized by Psychrobacter faecalis: An endophyte of Averrhoa carambola. Process Biochem 134:329–340. https://doi.org/10.1016/j.procbio.2023.10.015
Dimkpa CO, Merten D, Svatoš A et al (2009) Siderophores mediate reduced and increased uptake of cadmium by Streptomyces tendae F4 and sunflower (Helianthus annuus), respectively. J Appl Microbiol 107:1687–1696. https://doi.org/10.1111/j.1365-2672.2009.04355.x
Din BU, Amna RM et al (2020) Assisted phytoremediation of chromium spiked soils by Sesbania Sesban in association with Bacillus xiamenensis PM14: a biochemical analysis. Plant Physiol Biochem 146:249–258. https://doi.org/10.1016/j.plaphy.2019.11.010
Fernández-Fuego D, Keunen E, Cuypers A et al (2017) Mycorrhization protects Betula pubescens Ehr. from metal-induced oxidative stress increasing its tolerance to grow in an industrial polluted soil. J Hazard Mater 336:119–127. https://doi.org/10.1016/j.jhazmat.2017.04.065
Fiodor A, Ajijah N, Dziewit L, Pranaw K (2023) Biopriming of seed with plant growth-promoting bacteria for improved germination and seedling growth. Front Microbiol 14:2. https://doi.org/10.3389/fmicb.2023.1142966
Fiske CH, Subbarow Y (1925) The colorimetric determination of phosphorus. J Biol Chem 66:375–400. https://doi.org/10.1016/s0021-9258(18)84756-1
Gall JE, Boyd RS, Rajakaruna N (2015) Transfer of heavy metals through terrestrial food webs: a review. Environ Monit Assess 187:2. https://doi.org/10.1007/s10661-015-4436-3
Gao N, Wang H, Yang EH (2010) An experimental study on ferromagnetic nickel nanowires functionalized with antibodies for cell separation. Nanotechnology 21:2. https://doi.org/10.1088/0957-4484/21/10/105107
Geetha K, Venkatesham E, Hindumathi A, Bhadraiah B (2014) Isolation, screening and characterization of plant growth promoting bacteria and their effect on Vigna Radita (L.) R.Wilczek. Int J Curr Microbiol Appl Sci 3:799–809
Genchi G, Sinicropi MS, Lauria G et al (2020) The effects of cadmium toxicity. Int J Environ Res Public Health 17:1–24. https://doi.org/10.3390/ijerph17113782
Genova Genova Bradford Protein Assay Protocol : P09–003A. i:5–7
Gulati A, Sharma N, Vyas P et al (2010) Organic acid production and plant growth promotion as a function of phosphate solubilization by Acinetobacter rhizosphaerae strain BIHB 723 isolated from the cold deserts of the trans-Himalayas. Arch Microbiol 192:975–983. https://doi.org/10.1007/s00203-010-0615-3
Gupta S, Pandey S (2019) ACC deaminase producing bacteria with multifarious plant growth promoting traits alleviates salinity stress in French Bean (Phaseolus vulgaris) plants. Front Microbiol 10:1–17. https://doi.org/10.3389/fmicb.2019.01506
Gupta V, Jatav PK, Verma R et al (2017) Nickel accumulation and its effect on growth, physiological and biochemical parameters in millets and oats. Environ Sci Pollut Res 24:23915–23925. https://doi.org/10.1007/s11356-017-0057-4
Gupta DK, Chatterjee S, Walther C (2020) Lead in plants and the environment
Hemeda HM, Klein BP (1990) Effects of naturally occurring antioxidants on peroxidase activity of vegetable extracts—HEMEDA—2006—Journal of Food Science—Wiley Online Library. J Food Sci 55:184–186
Henderson RG, Durando J, Oller AR et al (2012) Acute oral toxicity of nickel compounds. Regul Toxicol Pharmacol 62:425–432. https://doi.org/10.1016/j.yrtph.2012.02.002
Isenberg HD (2003) Clinical microbiology: past, present, and future. J Clin Microbiol 41:917–918. https://doi.org/10.1128/JCM.41.3.917-918.2003
Islam S, Akanda AM, Prova A et al (2016) Isolation and identification of plant growth promoting rhizobacteria from cucumber rhizosphere and their effect on plant growth promotion and disease suppression. Front Microbiol 6:1–12. https://doi.org/10.3389/fmicb.2015.01360
Itusha A, Osborne WJ, Vaithilingam M (2019) Enhanced uptake of Cd by biofilm forming Cd resistant plant growth promoting bacteria bioaugmented to the rhizosphere of Vetiveria zizanioides. Int J Phytoremediation 21:487–495. https://doi.org/10.1080/15226514.2018.1537245
Jeevanandam V, Osborne J (2022) Understanding the fundamentals of microbial remediation with emphasize on metabolomics. Prep Biochem Biotechnol 52:351–363. https://doi.org/10.1080/10826068.2021.1946694
Kadam AA, Kulkarni AN, Lade HS, Govindwar SP (2014) Exploiting the potential of plant growth promoting bacteria in decolorization of dye Disperse Red 73 adsorbed on milled sugarcane bagasse under solid state fermentation. Int Biodeterior Biodegrad 86:364–371. https://doi.org/10.1016/j.ibiod.2013.10.012
Kai K, Kasamatsu K, Hayashi H (2012) (Z)-N-(4-Decenoyl) homoserine lactone, a new quorum-sensing molecule, produced by endobacteria associated with Mortierella alpina A-178. Tetrahedron Lett 53:5441–5444. https://doi.org/10.1016/j.tetlet.2012.07.133
Khan MS, Zaidi A, Musarrat J (2014) Phosphate solubilizing microorganisms: principles and application of microphos technology
Koch LK, Akhtar N, Ilyas N et al (2021) Role of Bacillus cereus in Improving the growth and Phytoextractability of Brassica nigra (L.) K. Koch Chrom Contam Soil Mol 26:1–20
Kumar S, Prasad S, Yadav KK et al (2019) Hazardous heavy metals contamination of vegetables and food chain: Role of sustainable remediation approaches—a review. Environ Res 179:108792. https://doi.org/10.1016/j.envres.2019.108792
Li J, McDonald-Gillespie J (2020) Airborne lead (Pb) from abandoned mine waste in Northeastern Oklahoma, USA. GeoHealth 4:2. https://doi.org/10.1029/2020GH000273
MacWilliams MP (2009) Indole test protocol. Am Soc Microbiol Microbiol 2:1–9
Marchiol L, Sacco P, Assolari S, Zerbi G (2004) Reclamation of polluted soil: phytoremediation potential of crop-related Brassica species. Water Air Soil Pollut 158:345–356. https://doi.org/10.1023/B:WATE.0000044862.51031.fb
Masalha J, Kosegarten H, Elmaci Ö, Mengel K (2000) The central role of microbial activity for iron acquisition in maize and sunflower. Biol Fertil Soils 30:433–439. https://doi.org/10.1007/s003740050021
Mcdevitt S (2009) Methyl red and voges-proskauer test protocols. Am Soc Microbiol 2:1–9
Mehta S, Nautiyal CS (2001) An efficient method for qualitative screening of phosphate-solubilizing bacteria. Curr Microbiol 43:51–56. https://doi.org/10.1007/s002840010259
Mowlick S, Yasukawa H, Inoue T et al (2013) Suppression of spinach wilt disease by biological soil disinfestation incorporated with Brassica juncea plants in association with changes in soil bacterial communities. Crop Prot 54:185–193. https://doi.org/10.1016/j.cropro.2013.08.012
Mukherjee P, Roychowdhury R, Roy M (2017) Phytoremediation potential of rhizobacterial isolates from Kans grass (Saccharum spontaneum) of fly ash ponds. Clean Technol Environ Policy 19:1373–1385. https://doi.org/10.1007/s10098-017-1336-y
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Nazir R, Khan M, Masab M et al (2015) Accumulation of heavy metals (Ni, Cu, Cd, Cr, Pb, Zn, Fe) in the soil, water and plants and analysis of physico-chemical parameters of soil and water collected from Tanda Dam Kohat. J Pharm Sci Res 7:89–97
Nemat H, Shah AA, Akram W et al (2020) Ameliorative effect of co-application of Bradyrhizobium japonicum EI09 and Se to mitigate chromium stress in Capsicum annum L. Int J Phytoremediation 22:1396–1407. https://doi.org/10.1080/15226514.2020.1780412
Oger C, Berthe T, Quillet L et al (2001) Estimation of the abundance of the cadmium resistance gene cadA in microbial communities in polluted estuary water. Res Microbiol 152:671–678. https://doi.org/10.1016/S0923-2508(01)01246-3
Ojuederie OB, Babalola OO (2017) Microbial and plant-assisted bioremediation of heavy metal polluted environments: a review. Int J Environ Res Public Health 14:2. https://doi.org/10.3390/ijerph14121504
Patten CL, Glick BR (2002) Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. Appl Environ Microbiol 68:3795–3801. https://doi.org/10.1128/AEM.68.8.3795-3801.2002
Pouyat RV, Szlavecz K, Yesilonis ID et al (2015) Multi-scale assessment of metal contamination in residential soil and soil fauna: a case study in the Baltimore-Washington metropolitan region, USA. Landsc Urban Plan 142:7–17. https://doi.org/10.1016/j.landurbplan.2015.05.001
Rajkumar M, Ae N, Prasad MNV, Freitas H (2010) Potential of siderophore-producing bacteria for improving heavy metal phytoextraction. Trends Biotechnol 28:142–149. https://doi.org/10.1016/j.tibtech.2009.12.002
Reddy S, Osborne JW (2020) Biodegradation and biosorption of reactive red 120 dye by immobilized Pseudomonas guariconensis. Kinet Toxicity Study. 2:1230–1241. https://doi.org/10.1002/wer.1319
Reeves RD, Baker AJM, Jaffré T et al (2018) A global database for plants that hyperaccumulate metal and metalloid trace elements. New Phytol 218:407–411. https://doi.org/10.1111/nph.14907
Rinklebe J, Antoniadis V, Shaheen SM et al (2019) Health risk assessment of potentially toxic elements in soils along the Central Elbe River, Germany. Environ Int 126:76–88. https://doi.org/10.1016/j.envint.2019.02.011
Saurav K, Kannabiran K (2011) Biosorption of Cd(II) and Pb(II) ions by aqueous solutions of novel alkalophillic Streptomyces VITSVK5 spp. biomass. J Ocean Univ China 10:61–66. https://doi.org/10.1007/s11802-011-1771-z
Schippers B, Bakker AW, Bakker PAHM (1987) Interactions of deleterious and beneficial rhizosphere microorganisms and the effect of crop** practices. Annu Rev Phytopathol 25:339–358. https://doi.org/10.1146/annurev.py.25.090187.002011
Schwyn B, Neilands JB (1987) Universal chemical assay for the detection and determination of siderophores. Anal Biochem 160:47–56. https://doi.org/10.1016/0003-2697(87)90612-9
Singh P, Chauhan PK, Upadhyay SK et al (2022) Mechanistic insights and potential use of siderophores producing microbes in rhizosphere for mitigation of stress in plants grown in degraded land. Front Microbiol 13:1–16. https://doi.org/10.3389/fmicb.2022.898979
Sinha A, Lulu S, Vino S et al (2018) Degradation of reactive green dye and textile effluent by Candida sp. VITJASS isolated from wetland paddy rhizosphere soil. J Environ Chem Eng 6:5150–5159. https://doi.org/10.1016/j.jece.2018.08.004
Song P, Gao J, Li X et al (2019) Phthalate induced oxidative stress and DNA damage in earthworms (Eisenia fetida). Environ Int 129:10–17. https://doi.org/10.1016/j.envint.2019.04.074
Sriram KP, Mangrolia U, Osborne WJ (2020) Isolation and characterization of culturable indigenous endophytic bacteria in the tender coconut. Food Biotechnol 34:228–242. https://doi.org/10.1080/08905436.2020.1789872
Tamilselvan N, Saurav K, Kannabiran K (2012) Biosorption of Cr (VI), Cr (III), Pb (II) and Cd (II) from aqueous solutions by Sargassum wightii and Caulerpa racemosa algal biomass. J Ocean Univ China 11:52–58. https://doi.org/10.1007/s11802-012-1843-8
Tiwari S, Hasan A, Pandey LM (2017) A novel bio-sorbent comprising encapsulated Agrobacterium fabrum (SLAJ731) and iron oxide nanoparticles for removal of crude oil co-contaminant, lead Pb(II). Elsevier, Amsterdam
Tran TA, Popova LP (2013) Functions and toxicity of cadmium in plants: recent advances and future prospects. Turk J Bot 37:1–13. https://doi.org/10.3906/bot-1112-16
Viji AS, Antony BT, Wagh MS, Osborne WJ (2021) Bioremoval of cadmium by co-cultivated bacterial strains, Bacillus paramycoides and Bacillus subtilis, in a pilot-scale phyto- and rhizoremediation approach. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-021-03540-7
Wagh MS, Osborne WJ, Sivarajan S (2022) Bacillus xiamenensis and earthworm Eisenia fetida in bio removal of lead, nickel and cadmium: a combined bioremediation approach. Appl Soil Ecol 176:104459. https://doi.org/10.1016/j.apsoil.2022.104459
Wagh MS, Sivarajan S, Osborne WJ (2023) A new paradigm in the bioremoval of lead, nickel, and cadmium using a cocktail of biosystems: a metagenomic approach. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-023-26705-y
Zhang FQ, Wang YS, Lou ZP, De DJ (2007) Effect of heavy metal stress on antioxidative enzymes and lipid peroxidation in leaves and roots of two mangrove plant seedlings (Kandelia candel and Bruguiera gymnorrhiza). Chemosphere 67:44–50. https://doi.org/10.1016/j.chemosphere.2006.10.007
Zhang YF, He LY, Chen ZJ et al (2011) Characterization of lead-resistant and ACC deaminase-producing endophytic bacteria and their potential in promoting lead accumulation of rape. J Hazard Mater 186:1720–1725. https://doi.org/10.1016/j.jhazmat.2010.12.069
Zhang N, Wang D, Liu Y et al (2014) Effects of different plant root exudates and their organic acid components on chemotaxis, biofilm formation and colonization by beneficial rhizosphere-associated bacterial strains. Plant Soil 374:689–700. https://doi.org/10.1007/s11104-013-1915-6
Zhang C, Bruins ME, Yang ZQ et al (2016) A new formula to calculate activity of superoxide dismutase in indirect assays. Anal Biochem 503:65–67. https://doi.org/10.1016/j.ab.2016.03.014
Zhao Y, Gu Y, Qin F et al (2017) Application of near-infrared spectroscopy to quantitatively determine relative content of Puccnia striiformis f. Sp. tritici DNA in wheat leaves in incubation period. J Spectrosc. https://doi.org/10.1155/2017/9740295
Acknowledgements
The authors are thankful to VIT-Vellore, for the assistance throughout the research. We extend our gratitude towards Technology Business Incubator (TBI) for AAS analysis, and SEM facility for the help during sample analysis. The authors would like to thank Dr. Prabavathy from MSSRF-Chennai for providing the reporter strains CVO26 and NTL4.
Funding
No funding was provided for the study.
Author information
Authors and Affiliations
Contributions
MSW: Conceptualization, Experimental designing and implementation, Manuscript writing; SS: Supervision, Validation; JWO: Work plan and Methods, Editing, Compiling data, Communication.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest regarding the manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wagh, M.S., Sivarajan, S. & Osborne, J.W. Deciphering the enhanced translocation of Pb, Ni and Cd from artificially polluted soil to Chrysopogon zizanioides augmented with Bacillus xiamenensis VITMSJ3. 3 Biotech 14, 180 (2024). https://doi.org/10.1007/s13205-024-04001-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-024-04001-x