Abstract
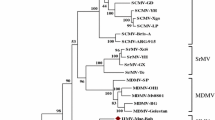
To date, the complete genome of two wisteria vein mosaic virus (WVMV) has been sequenced worldwide. Here, the genomic sequence of WVMV isolated from Wisteria sinensis in Iran was determined for the first time, using deep RNA sequencing and RT-PCR followed by Sanger sequencing. The sequence was 9694 nucleotides in length; excluding the 3’-poly(A) tail and contained a single open reading frame of 9279 nucleotides encoding a large polyprotein of 3092 amino acids and predicted molecular weight of 35,368 KDa. The genome contained nine putative proteolytic cleavage sites and motifs conserved in homologous proteins of other potyviruses. Sequence analysis suggested that WVMV-Ir sequence shared 76.37–86.01% nucleotide (nt) identity and 82.45–91.91% amino acid (aa) identity with two other isolates (Bei**g and JEBU-p) available in the GenBank, the highest with the Chinese isolate Bei**g (86.01% nt identity, 91.91% aa identity). Sequence identities over most of the genome were within the range 80–86% and 85–95% at the nt and aa levels, respectively; however, high variability was observed in the 5’-UTR (51.62%), P1 (62.03% nt identity, 50.78% aa identity) and P3 (79.82%nt identity, 78.67% aa identity) regions, suggesting that Ir, Bei**g, and JEBU-p are three different strains. These variabilities may be due to different mutation phenomena of a common ancestor virus or mutations caused by different selection pressures in different agro-ecological regions. The results of the phylogenetic analysis indicated that WVMV was most closely related to soybean mosaic virus and watermelon mosaic virus and less closely related to the zantedeschia mild mosaic virus and dasheen mosaic virus. In the greenhouse, WVMV-Ir caused severe symptoms in Phaseolus vulgaris, Vicia faba, W. sinensis, Chenopodium quinoa, C. amaranticolor, and Nicotiana benthamiana.


Similar content being viewed by others
References
Adams MJ, Antoniw JF, Beaudoin F (2005a) Overview and analysis of the polyprotein cleavage sites in the family Potyviridae. Mol Plant Pathol 6:471–487
Adams MJ, Antoniw JF, Fauquet CM (2005b) Molecular criteria for genus and species discrimination within the family Potyviridae. Arch Virol 150:459–479
Adams MJ, Zerbini FM, French R, Rabenstein F, Stenger DC (2012) Valkonen JPT. In: King AMQ, Adams MJ, Carstens EB (eds) Virus taxonomy: 9th report of the International Committee on the Taxonomy of Viruses. Elsevier Academic Press, San Diego, pp 1069–1089
Al Jaberi M, Zakiaghl M, Mehrvar M (2018) First report of Wisteria vein mosaic virus on Wisteria sinensis in Iran. New Dis Rep 38:18
Bos L (1996) Wisteria vein mosaic potyvirus. In: Brunt AA, Crabtree K, Dallwitz MJ, Gibbs AJ, Watson L (eds) Viruses of plants: descriptions and lists from the VIDE Database. CAB International, Wallingford, pp 1407–1409
Bos L (1970) The identification of three new viruses isolated from Wisteria and Pisum in The Netherlands and the problem of variation with the potato virus Y group. Netherlands J Plant Pathol. https://doi.org/10.1007/BF01976763
Chung BY-W, Miller WA, Atkins JF, Firth AE (2008) An overlap** essential gene in the Potyviridae. Proc Natl Acad Sci USA 105:5897–5902
Clover GRG, Tang Z, Smales TE, Pearson MN (2003) Taxonomy of Wisteria vein mosaic virus and extensions to its host range and geographical distribution. Plant Pathol 52:92–96
Clover GRG, Denton JO, Denton GJ (2015) First report of Wisteria vein mosaic virus on Wisteria spp. in the United Kingdom. New Dis Rep. https://doi.org/10.5197/j.2044-0588.2015.031.001
Dougherty WG, Semler BL (1993) Expression of virus-encoded proteinases: functional and structural similarities with cellular enzymes. Microbiol Rev 57:781–822
Ferrer-Orta C, Ferrero D, Verdaguer N (2015) RNA-dependent RNA polymerases of picornaviruses: from the structure to regulatory mechanisms. Viruses 7:4438–4460
Gallie DR, Walbot V (1992) Identification of the motifs within the Tobacco mosaic virus 5’-leader responsible for enhancing translation. Nucleic Acids Res 20:4631–4638
Gao F, Zou W, **e L, Zhan J (2017) Adaptive evolution and demographic history contribute to the divergent population genetic structure of Potato virus Y between China and Japan. Evol Appl 10:379–390
Garcı´a-Arenal F, Fraile A, Malpica JM (2001) Variability and genetic structure of plant virus populations. Annu Rev Phytopathol 39:157–186
Gibbs A, Ohshima K (2010) Potyviruses and the digital revolution. Annu Rev Phytopathol 48:205–223
Grylls NE, Butler FC (1959) Subterranean clover stunt, a virus disease of pasture legumes. Aust J Agric Res 10:145–159
Ha C, Coombs S, Revill PA, Harding RM, Vu M, Dale JL (2008) Design and application of two novel degenerate primer pairs for the detection and complete genomic characterization of potyviruses. Arch Virol 153:25–36
Jiang YF, Chen XL, Lin H, Wang F, Chen F (2011) Floral Scent in Wisteria: chemical composition, emission pattern, and regulation. J Am Soc Hortic Sci 136:307–314
Kadare´ G, Haenni A-L (1997) Virus-encoded RNA helicases. J Virol 71:2583–2590
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549
L´opez-Moya JJ, Wang RY, Pirone TP (1999) Context of the coat protein DAG motif affects potyvirus transmissibility by aphids. J Gen Virol 80:3281–3288
Li Y, Deng C, Qiao Y, Zhao X, Zhou Q (2017) Characterization of a new badnavirus from Wisteria sinensis. Arch Virol 162(7):2125–2129
Liang WX, Song LM, Li Y, Tian GZ, Li HF, Fan ZF (2004) First report of Wisteria vein mosaic virus in China. Plant Pathol 53:516
Liang WX, Song LM, Tian GZ, Li HF, Fan ZF (2006) The genomic sequence of Wisteria vein mosaic virus and its similarities with other potyviruses. Arch Virol 151:2311–2319
Marie-Jeanne V, Ioos R, Peyre J, Alliot B, Signoret P (2000) Differentiation of Poaceae potyviruses by reverse transcription-polymerase chain reaction and restriction analysis. J Phytopathol 148:141–151
Martin DP, Murrell B, Golden M, Khoosal A, Muhire B (2015) RDP4: detection and analysis of recombination patterns in virus genomes. Virus Evol. https://doi.org/10.1093/ve/vev003
Milojević K, Radović N, Stanković I, Vučurović A, Nikolić D, Bulajić A, Krstić B (2016) First report of Cucumber mosaic virus infecting Wisteria sinensis in Serbia. Plant Dis 100:1799
Moradi Z, Mehrvar M (2019) Genetic variability and molecular evolution of bean common mosaic virus populations in Iran: comparison with the populations in the world. Eur J Plant Pathol 154:673–690
Moradi Z, Mehrvar M, Nazifi E, Zakiaghl M (2017a) Iranian johnsongrass mosaic virus: the complete genome sequence, molecular and biological characterization, and comparison of coat protein gene sequences. Virus Genes 53:77–88
Moradi Z, Nazifi E, Mehrvar M (2017b) Molecular characterization of two Sugarcane streak mosaic virus isolates from Iran with emphasis on its population structure. Acta Virol 61:428–437
Naidu RA, Karthikeyan G (2008) First report of Wisteria vein mosaic virus in Wisteria sinensis in the United States of America. Plant Health Progress. https://doi.org/10.1094/PHP-2008-0818-01-BR
Nigam D, LaTourrette K, Souza PFN, Garcia-Ruiz H (2019) Genome-wide variation in potyviruses. Front Plant Sci 10:1439
Plisson C, Drucker M, Blanc S, German-Retana S, Le Gall O, Thomas D, Bron P (2003) Structural characterization of HC-Pro, a plant virus multifunctional protein. J Biol Chem 278:23753–23761
Revers F, Le Gall O, Candresse T, Maule AJ (1999) New advances in understanding the molecular biology of plant: potyvirus interactions. Mol Plant-Microbe Interact 12:367–376
Rodamilans B, Valli A, Mingot A, San Leon D, Baulcombe D, Lopez-Moya JJ, Garcia JA (2015) RNA polymerase slippage as a mechanism for the production of frameshift gene products in plant viruses of the Potyviridae family. J Virol 89:6965–6967
Rozas J, Ferrer-Mata A, Sanchez-DelBarrio JC, Guirao-Rico S, Librado P, Ramos-Onsins SE, Sanchez-Gracia A (2017) DnaSP 6: DNA sequence polymorphism analysis of large datasets. Mol Biol Evol 34:3299–3302
Turpen TJ (1989) Molecular cloning of a potato virus Y genome: nucleotide sequence homology in non-coding regions of potyviruses. J Gen Virol 70:1951–1960
Urcuqui-Inchima S, Haenni A, Bernardi F (2001) Potyvirus proteins: a wealth of functions. Virus Res 74:157–175
Valouzi H, Hashemi SS, Wylie SJ, Ahadiyat A, Golnaraghi A (2020) Wisteria vein mosaic virus detected for the first time in Iran from an unknown host by analysis of aphid vectors. Plant Pathol J 36:87–97
Worrall EA, Hayward AC, Fletcher SJ, Mitter N (2019) Molecular characterization and analysis of conserved potyviral motifs in Bean common mosaic virus (BCMV) for RNAi-mediated protection. Arch Virol 164:181–194
Author information
Authors and Affiliations
Contributions
MM conceived and designed the experiments. MSAJ did the experimental works. ZM and HRAI helped to conduct some laboratory experiments. ZM prepared the manuscript, performed data analysis, and interpretation. ZM, MM, MZ and MSAJ contributed to the interpretation and critical revision of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Al-Jaberi, M.S., Moradi, Z., Mehrvar, M. et al. Whole genome characterization of wisteria vein mosaic virus from Iran and its relationship to other members of bean common mosaic virus group. 3 Biotech 11, 407 (2021). https://doi.org/10.1007/s13205-021-02957-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02957-8