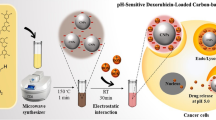
Abstract
In this work, we prepared a spherical CeO2/GO/c-MWCNT (Cerium oxide/Graphene oxide/Multiwalled Carbon nanotube) nanocomposite whose surface morphology looks porous and fibrous. The stacked layer of GO and c-MWCNT was stuffed with porous aminatedCeO2nanoparticles (NPs). Their physio-chemico aspects were probed using X-Ray Diffraction (XRD), Scanning Electron Microscopy (SEM) and Fourier Transform Infrared Spectroscopy (FTIR) analysis. In addition, proposed porous CeO2/GO/c-MWCNT hybrid nanosystem, cisplatin-loaded CeO2/GO/c-MWCNT hybrid system (CeO2/CDDP-GO/c-MWCNT) and cisplatin (CDDP) was subjected to HeLa cells to examine % cell inhibition under a suitable in-vitro microenvironment. % Cell Inhibition at 1000 µg/mL of CeO2/CDDP-GO/c-MWCNT system was found to be of 91.82% and for 7.8 µg/mL concentration, it was found to be 61.41% on HeLa cell lines. CeO2/GO/c-MWCNT system has achieved 99.96% encapsulation efficiency with cisplatin drug. Hence, Cytotoxicity results are more significant for CeO2/CDDP-GO/c-MWCNT when compared to CeO2/GO/c-MWCNT system. Further, the developed CeO2/CDDP-GO/c-MWCNT hybrid nanosystem was taken up for Acridine Orange/Ethidium Bromide (AO/EB) dual fluorescence staining study to record the morphology assisted variations in both live and dead cells using fluorescence spectroscopy. Later, a flow cytometry study was performed to monitor the cell cycle at which the maximum cells were dead. In the current work, at the first stage (R1) the CeO2/CDDP-GO/c-MWCNT system was able to kill 3,76,659 cells in a cell culture medium that has a total volume of 4,91,748 HeLa cells at a lower concentration of 15.6 µg/mL. Based on the observations from the present in-vitro investigations, the developed CeO2/CDDP-GO/c-MWCNT hybrid nanosystem can be used as therapeutic platform for cervical cancer.










Similar content being viewed by others
Data availability
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
References
Alili L, Sack M, Karakoti AS, Teuber S, Puschmann K, Hirst SM, Reilly CM, Zanger K, Stahl W, Das S, Seal S, Brenneisen P (2011) Combined cytotoxic and anti-invasive properties of redox-active nanoparticles in tumor - stroma interactions. Biomaterials 32:2918–2929
Asati A, Santra S, Kaittanis C, Nath S, Perez JM (2009) Oxidase-like activity of polymer-coated cerium oxide nanoparticles. Angew Chem Int Ed 48:2308–2312
Attari E, Nosrati H, Danafar H, KheiriManjili H (2019) Methotrexate anticancer drug delivery to breast cancer cell lines by iron oxide magnetic based nanocarriers. J Biomed Mater Res 107:2492–2500
Avgoustakis K, Beletsi A, Panagi Z, Klepetsanis P, Karydas AG, Ithakissios DS (2002) PLGA–mPEG nanoparticles of cisplatin: in vitro nanoparticle degradation, in vitro drug release and in vivo drug residence in blood properties. J Control Release 79(1–3):123–135
Baek S, Singh R, Khanal K, Patel D, Lee KD, Leong EJ, Chrzanowski KW, Kim W (2015) Smart multifunctional drug delivery towards anticancer therapy harmonized in mesoporous nanoparticles. Nanoscale 7:14191–14216
Celardo I, Pedersen JZ, Traversa E, Ghibelli L (2011) Pharmacological potential of cerium oxide nanoparticles. Nanoscale 3:1411–1420
Das J, Choi Y-J, WoongHan J, Reza AMM, Kim J-H (2017) Nanoceria-mediated delivery of doxorubicin enhances the antitumourefciency in ovarian cancer cells via apoptosis. Sci Rep 7:9513
dos Santos MC, Maynart MC, Aveiro LR, da Paz EC, dos Santos Pinheiro V (2017) Carbon-based materials: recent advances, challenges, and perspectives. Reference module in materials science and materials engineering. Elsevier. https://doi.org/10.1016/B978-0-12-803581-8.09262-6
Fallah-Shojaei A, Tabatabaeian K, Shirini F, Zoha Hejazi S (2014) Multi-walled carbon nanotube supported Fe3O4NPs: an efficient and reusable catalyst for the one-pot synthesis of 4H-pyran derivatives. RSC Adv 4:9509–9516
Fonseca C, Simoes S, Gaspar R (2002) paclitaxel-loaded PLGA nanoparticles: preparation, physicochemical characterization and in vitro anti-tumoral activity. J Control Release 83:273–286
Giri S, Karakoti A, Graham RP, Maguire JL, Reilly CM, Seal S, Rattan R, Shridhar V (2013) Nanoceria: a rare-earth nanoparticle as a novel antiangiogenic therapeutic agent in ovarian cancer. Plus one 8:e54578–e54583
He K, Chen N, Wang C, Wei L, Chen J (2018) Method for determining crystal grain size by X-Ray diffraction. Cryst Res Technol 53(2):1700157
Hijaz M, Das S, Mert I, Gupta A, Al-Wahab Z, Tebbe C, Dar S, Chhina J, Giri S, Munkarah A, Seal S, Rattan R (2016) Folic acid tagged nanoceria as a novel therapeutic agent in ovarian cancer. BMC Cancer 16:220
Huang Y, Tang Z, Zhang X, Haiyang Yu, Sun H, Pang X, Chen X (2013) pH-triggered charge-reversal polypeptide nanoparticles for cisplatin delivery: preparation and in vitro evaluation. Biomacromol 14:2023–2032
Huang J-Y, Chen M-H, Kuo W-T, Sun Y-J, Lin F-H (2015) The characterization and evaluation of cisplatin-loaded magnetite–hydroxyapatite nanoparticles (mHAp/CDDP) as dual treatment of hyperthermia and chemotherapy for lung cancer therapy. Ceram Int 41:2399–2410
Iwashita N (2016) X-ray powder diffraction. Materials science and engineering of carbon. Elsevier, pp 7–25
Jabr-Milane LS, Van Vlerken LE, Yadav S, Amiji MM (2008) Multi-functional nanocarriers to overcome tumor drug resistance. Cancer Treat Rev 34:592–602
Karakoti AS, Tsigkou O, Yue S, Lee PD, Stevens MM, Jones JR, Seal S (2010) Rare earth oxides as nanoadditives in 3-D nanocomposite scaffolds for bone regeneration. J Mater Chem 20:8912–8919
Karmakar A, Iancu C, Bartos DM, Mahmood MW, Ghosh A, Xu Y, Dervishi E, Collom SL, Khodakovskaya M, Mustafa T, Watanabe F, Biris AR, Zhang Y, Ali SF, Casciano D, Hassen S, Nima Z, Biris AS (2012) Raman spectroscopy as a detection and analysis tool for in vitro specific targeting of pancreatic cancer cells by EGF-conjugated, single-walled carbon nanotubes. J Appl Toxicol 32(5):365–375
Kumari A, Yadav SK, Yadav SC (2010) Biodegradable polymeric nanoparticles based drug delivery systems. Colloids Surf B: Biointerfaces 75:1–18
Lamba D (2014) Wide-angle X-ray diffraction pattern. In: Drioli E, Giorno L (eds) Encyclopedia of membranes. Springer, Berlin Heidelberg, pp 1–2
Li X, Sun L, Ge A, Guo Y (2011) Enhanced chemiluminescence detection of thrombin based on cerium oxide nanoparticles. Chem Commun 47:947–949
Li M, Shi P, Xu C, Ren J, Qu X (2013) Cerium oxide caged metal chelator: anti-aggregation and anti-oxidation integrated H2O2-responsive controlled drug release for potential Alzheimer’s disease treatment. Chem Sci 4:2536–2542
Lippert B (2006) Cisplatin: chemistry and biochemistry of a leading anticancer drug. Wiley. https://doi.org/10.1002/9783906390420
Mahmood M, Karmakar A, Fejleh A et al (2009) Synergistic enhancement of cancer therapy using a combination of carbon nanotubes and anti-tumor drug. Nanomedicine (Lond) 4(8):883–893
Mandoli C, Pagliari F, Pagliari S, Forte G, Di Nardo P, Licoccia S, Traversa E (2010) Stem cell aligned growth induced by CeO2 nanoparticles in PLGA Scaffolds with improved bioactivity for regenerative medicine. Adv Funct Mater 20:1617–1624
Masood F, Chen P, Yasin T, Fatima N, Hasan F, Hameed A (2013) Encapsulation of Ellipticine in poly-(3-hydroxybutyrate-co-3-hydroxyvalerate) based nanoparticles and its in-vitro application. Mater Sci Eng: C 33:1054–1060
Mura S, Nicolas J, Couvreur P (2013) Stimuli-responsive nanocarriers for drug delivery. Nat Mater 12:991–1003
Palanisamy K, Um JH, Jeong M, Yoon W-S (2016) Porous V2O5/RGO/CNT hierarchical architecture as a cathode material: emphasis on the contribution of surface lithium storage. Nat Sci Rep 6:31275
Popov AL, Popova NR, Tarakina NV, Ivanova OS, Ermakov AM, Ivanov VK, Sukhorukov GB (2018) Intracellular delivery of antioxidant CeO2 nanoparticles via polyelectrolyte microcapsules. ACS Biomater Sci Eng 4(7):2453–2462
Parhi P, Mohanty C, Sahoo SK (2012) ‘Nanotechnology-based combinational drug delivery: an emerging approach for cancer therapy. Drug Discov Today 17:1044–1052
Perez JM, Asati A, Nath S, Kaittanis C (2008) Synthesis of biocompatible dextran-coated nanoceria with pH-dependent antioxidant properties. Small 4:552–556
Petersen S, Fahr A, Bunjes H (2009) Flow cytometry as a new approach to investigate drug transfer between lipid particles. Mol Pharm 7:350–363
Rajendran R, Shrestha LK, Minami K, Subramanian M, Jayavel R, Ariga K (2014) Dimensionally integrated nanoarchitectonics for novel composite from 0D, 1D, and 2D nanomaterials: RGO/CNT/CeO2 ternary nanocomposite with electrochemical performance. J Mater Chem A 2:18480–18487
Ringe K, Walz C, Sabel B (2004) Nanoparticle drug delivery to the brain. In: Nalwa HS (ed) Encyclopedia of nanoscience and nanotechnology, vol 7. American Scientific Publishers, New York
Rosenberg B (1985) Fundamental studies with cisplatin. Cancer 55(10):2303–2306
Saranya J, Sreeja BS, Padmalaya G, Radha S, Manikandan T (2020) Ultrasonic assisted cerium oxide/graphene oxide hybrid: preparation, anti-proliferative, apoptotic induction and G2/M cell cycle arrest in HeLa cell lines. J Inorg Organomet Polym Mater 30:2666–2676
Schütt F, Postica V, Adelung R, Lupan O (2017a) Single and networked ZnO−CNT hybrid tetrapods for selective room-temperature high-performance ammonia sensors. ACS Appl Mater Interfaces 27:23107–23118
Schütt F, Signetti S, Krüger H, Röder S, Smazna D, Kaps S, Gorb SN, Mishra YK, Pugno NM, Adelung R (2017b) Hierarchical carbon nanotube tube networks: from strong foams to reinforced ceramics. Polym Hybrids Nat Commun 8:1215
Senapati S, Mahanta AK, Kumar S, Maiti P (2018) Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct Target Ther 3:7
Sharma G, Prema D, Venkataprasanna KS, Prakash J, Sahabuddin S, Devanand Venkatasubbu G (2020) Photo induced antibacterial activity of CeO2/GO against wound pathogens. Arab J Chem. https://doi.org/10.1016/j.arabjc.2020.09.004
Shen H, Ackerman AL, Cody V et al (2006) Enhanced and prolonged cross-presentation following endosomal escape of exogenous antigens encapsulated in biodegradable nanoparticles. Immunology 117(1):78–88
Tchounwou PB, Dasari S, Noubissi FK, Ray P, Kumar S (2021) Advances in our understanding of the molecular mechanisms of action of cisplatin in cancer therapy. J Exp Pharmacol 13:303–328
Wang Z, Ma R, Yan Li, Chen X, Zhu G (2015) Combined chemotherapy and photodynamic therapy using a nanohybrid based on layered double hydroxides to conquer cisplatin resistance. Chem Commun 51:11587–11590
Wei Y, Huang Q-A, Li M-G, Huang X-J, Fang B, Wang L (2011) CeO2 nanoparticles decorated multi-walled carbon nanotubes for electrochemical determination of guanine and adenine. ElectrochimicaActa 56:8571–8575
Wu X, Zhang Y, Lu Y, Pang S, Yang K, Tian Z, Pei Y, Qu Y, Wang F, Pei Z (2017) Synergistic and targeted drug delivery based on nano-CeO2 capped with galactose functionalized pillar[5]arene via host-guest interactions. J Mater Chem B 5:3483–3487
Xu P, Van Kirk EA, Murdoch WJ, Zhan Y, Isaak DD, Radosz M, Shen Y (2006) Anticancer efficacies of cisplatin-releasing pH-responsive nanoparticles. Biomacromolecules 7:829–835
Xu C, Lin Y, Wang J, Wu L, Wei W, Ren J, Qu X (2013) Nanoceria-triggered synergetic drug release based on CeO2-capped mesoporous silica host−guest interactions and switchable enzymatic activity and cellular effects of CeO2. Adv Healthcare Mater 2:1591–1599
Zhang J, Lan CQ, Post M, Simard B, Deslandes Y, HanHsieh T (2006) Design of nanoparticles as drug carriers for cancer therapy. Cancer Genom Proteom 3:147–158
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors gave their full contribution JS: Conceptualization, Methodology, Investigation, Writing – Original draft preparation. BSS: Data Curation, Validation & Formal Analysis. PS: Conceptualization, Methodology, Supervision.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study does not require ethical approval and experimentation works were carried out under in vitro conditions.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saranya, J., Sreeja, B.S. & Senthil Kumar, P. Microwave assisted cisplatin-loaded CeO2/GO/c-MWCNT hybrid as drug delivery system in cervical cancer therapy. Appl Nanosci 13, 4219–4233 (2023). https://doi.org/10.1007/s13204-023-02856-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-023-02856-9