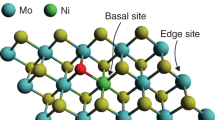
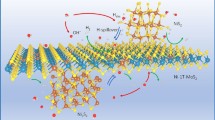
Abstract
Develo** the electrocatalyst with low cost, high efficiency, and environmentally friendly for hydrogen evolution reaction (HER) is important for the goal of high conversion of sustainable energy resources. Herein, a transition metal electrocatalyst named as Mn0.09-MoS2 is designed via a facile route. The successful substituting of Mn allows partial Mo in the original sulfide replaced by Mn and forms Mn-S bond, leading the lattice mismatch of original MoS2 and large quantities of assisted sulfur vacancies. The high S vacancy structure has the advantages of both morphology and intrinsic chemical properties such as rich vacancy assistance, abundant active sites, and fast electron transfer speed, promoting Mn0.09-MoS2/NF with excellent HER performance. The density functional theory calculations reveal the intrinsic activation reason that the energy gap of Mn0.09-MoS2 is narrower and the bandgap appears close to the Fermi level, illustrating that Mn substituting can effectively lower the energy barrier for electron transition and promote the electronic conductivity. Benefiting to these advantages, the designed Mn0.09-MoS2/NF only requires an overpotential of 141 mV to reach 10 mA cm−2 current density with an excellent electrochemical active surface area (1980 cm2) and pretty fast turnover frequency (0.299 s−1) in 1 M KOH. This exploration provides a feasible strategy for the controllable preparation and application of transition metal–based electrocatalyst.






Similar content being viewed by others
References
H. Du, R.M. Kong, X. Guo, F. Qu, J. Li, Recent progress in transition metal phosphides with enhanced electrocatalysis for hydrogen evolution. Nanoscale 10, 21617–21624 (2018)
S.H. Oh, R. Black, E. Pomerantseva, J.H. Lee, L.F. Nazar, Synthesis of a metallic mesoporous pyrochlore as a catalyst for lithium-O2 batteries. Nat Chem 4, 1004–1010 (2012)
Y. Yan, B.Y. **a, B. Zhao, X. Wang, A review on noble-metal-free bifunctional `heterogeneous catalysts for overall electrochemical water splitting. J. Mater. Chem. A. 4, 17587–17603 (2016)
G. Darabdhara, P.K. Boruah, P. Borthakur, N. Hussain, M.R. Das, T. Ahamad, S.M. Alshehri, V. Malgras, K.C. Wu, Y. Yamauchi, Correction: reduced graphene oxide nanosheets decorated with Au-Pd bimetallic alloy nanoparticles towards efficient photocatalytic degradation of phenolic compounds in water. Nanoscale 8, 19174–19175 (2016)
W. Zhu, X. Yue, W. Zhang, S. Yu, Y. Zhang, J. Wang, J. Wang, Nickel sulfide microsphere film on Ni foam as an efficient bifunctional electrocatalyst for overall water splitting. Chem Commun (Camb) 52, 1486–1489 (2016)
S. Peng, L. Li, J. Zhang, T.L. Tan, T. Zhang, D. Ji, X. Han, F. Cheng, S. Ramakrishna, Engineering Co9S8/WS2 array films as bifunctional electrocatalysts for efficient water splitting. J. Mater. Chem. A. 5, 23361–23368 (2017)
Q. Liu, Q. Fang, W. Chu, Y. Wan, X. Li, W. Xu, M. Habib, S. Tao, Y. Zhou, D. Liu, T. **ang, A. Khalil, X. Wu, M. Chhowalla, P.M. Ajayan, L. Song, Electron-substituted 1T-MoS2 via interface engineering for enhanced electrocatalytic hydrogen evolution. Chem. Mater. 29, 4738–4744 (2017)
G. Ye, Y. Gong, J. Lin, B. Li, Y. He, S.T. Pantelides, W. Zhou, R. Vajtai, P.M. Ajayan, Defects engineered monolayer MoS2 for improved Hydrogen evolution reaction. Nano Lett. 16, 1097–1103 (2016)
Y. Huang, Y. Sun, X. Zheng, T. Aoki, B. Pattengale, J. Huang, X. He, W. Bian, S. Younan, N. Williams, J. Hu, J. Ge, N. Pu, X. Yan, X. Pan, L. Zhang, Y. Wei, J. Gu, Atomically engineering activation sites onto metallic 1T-MoS2 catalysts for enhanced electrochemical hydrogen evolution. Nat Commun 10, 982 (2019)
Q. Wu, A. Dong, C. Yang, L. Ye, L. Zhao, Q. Jiang, Metal-organic framework derived Co3O4@Mo-Co3S4-Ni3S2 heterostructure supported on Ni foam for overall water splitting. Chem. Eng. J. 413, 127482 (2021)
L. Zeng, Z. Liu, K. Sun, Y. Chen, J. Zhao, Y. Chen, Y. Pan, Y. Lu, Y. Liu, C. Liu, Multiple modulations of pyrite nickel sulfides via metal heteroatom substituting engineering for boosting alkaline and neutral hydrogen evolution. J. Mater. Chem. A. 7, 25628–25640 (2019)
Y. Li, B. Jia, B. Chen, Q. Liu, M. Cai, Z. Xue, Y. Fan, H.P. Wang, C.Y. Su, G. Li, MoF-derived Mn substituted porous CoP nanosheets as efficient and stable bifunctional electrocatalysts for water splitting. Dalton Trans 47, 14679–14685 (2018)
J.K. Nørskov, T. Bligaard, A. Logadottir, J.R. Kitchin, J.G. Chen, S. Pandelov, U. Stimming, Trends in the exchange current for Hydrogen evolution. J. Electrochem. Soc. 152, 23–26 (2005)
H. Zhong, Z. Bai, B. Zou, Tuning the luminescence properties of colloidal I-III-VI semiconductor nanocrystals for optoelectronics and biotechnology applications. J Phys Chem Lett 3, 3167–3175 (2012)
L. Zhi**, X. BeiBei, H. Min, Y. Linghui, W. Zongpeng, H. Yucong, S. Shijie, Gu. Zhang Qinghua, Lin and Zhong Wenwu., Realizing negatively charged metal atoms through controllable d-electron transfer in ternary Ir1−xRhxSb intermetallic alloy for hydrogen evolution reaction. Adv. Energy Mater. 13, 2200855 (2022)
W. Zongpeng, S. Shijie, L. Zhi**, T. Weiying, Z. QingHua, Gu. Meng Fanqi, Lin and Zhong Wenwu., Regulating the local spin state and band structure in Ni3S2 nanosheet for improved oxygen evolution activity. Adv. Funct. Mater. 32, 2112832 (2022)
S. Shijie, W. Zongpeng, L. Zhi**, S. Kai, Z. Qinghua, Gu. Meng Fanqi, Lin and Zhong Wenwu., Crystalline-amorphous interfaces coupling of CoSe2/CoP with optimized d-band center and boosted electrocatalytic hydrogen evolution. Adv. Mater 34, 2110631 (2022)
H. Li, Q. Zhang, C.C.R. Yap, B.K. Tay, T.H.T. Edwin, A. Olivier, D. Baillargeat, From bulk to monolayer MoS2: evolution of raman scattering. Adv. Funct. Mater. 22, 1385–1390 (2012)
C. Chen, Z. Feng, Y. Feng, Y. Yue, C. Qin, D. Zhang, W. Feng, Large-Scale synthesis of a uniform film of bilayer MoS2 on graphene for 2D heterostructure phototransistors. ACS Appl Mater Interfaces 8, 19004–19011 (2016)
J.A. Dawson, I. Tanaka, Oxygen vacancy formation and reduction properties of beta-MnO2 grain boundaries and the potential for high electrochemical performance. ACS Appl. Mater. 6, 17776–17784 (2014)
W. Song, W. Lai, Z. Chen, J. Cao, H. Wang, Y. Lian, W. Yang, X. Jiang, Fabrication of 3D porous hierarchical NiMoS flowerlike architectures for hydrodesulfurization applications. ACS Appl. Nano Mater. 1, 442–454 (2017)
S. Eijsbouts, L. Vandenoetelaar, R. Vanpuijenbroek, MoS morphology and promoter segregation in commercial type 2 NiMo/AlO and CoMo/AlO hydroprocessing catalysts. J Catal 229, 352–364 (2005)
Y. Guo, J. Tang, H. Qian, Z. Wang, Y. Yamauchi, One-Pot synthesis of zeolitic imidazolate framework 67-derived hollow Co3S4@MoS2 heterostructures as efficient bifunctional catalysts. Chem. Mater. 29, 5566–5573 (2017)
R. Tong, Y. Qu, Q. Zhu, X. Wang, Y. Lu, S. Wang, H. Pan, Combined experimental and theoretical assessment of WXy (X = C, N, S, P) for hydrogen evolution reaction. ACS Appl. Energy Mater. 3, 1082–1088 (2019)
M.A. Lukowski, A.S. Daniel, F. Meng, A. Forticaux, L. Li, S. **, Enhanced hydrogen evolution catalysis from chemically exfoliated metallic MoS2 nanosheets. J. Am. Chem. Soc. 135, 10274–10277 (2013)
R. Jiang, B. Deng, L. Pi, L. Hu, D. Chen, Y. Dou, X. Mao, D. Wang, Molten electrolyte-modulated electrosynthesis of multi-anion Mo-based lamellar nanohybrids derived from natural minerals for boosting hydrogen evolution. ACS Appl. Mater. 12, 57870–57880 (2020)
Y. Huang, L. Sun, Z. Yu, R. Jiang, J. Huang, Y. Hou, F. Yang, B. Zhang, R. Zhang, Y. Zhang, Adjustable anchoring of Ni/Co cations by oxygen-containing functional groups on functionalized graphite paper and accelerated mass/electron transfer for overall water splitting. Catal. 10, 2627–2643 (2020)
L. Wen, J. Yu, C. **ng, D. Liu, X. Lyu, W. Cai, X. Li, Flexible vanadium-substituted Ni2P nanosheet arrays grown on carbon cloth for an efficient hydrogen evolution reaction. Nanoscale 11, 4198–4203 (2019)
Y. Gong, Y. Lin, Z. Yang, F. Jiao, J. Li, W. Wang, High-performance bifunctional flower-like Mn-substituted Cu7.2S4@NiS2@NiS/NF catalyst for overall water splitting, Appl. Surf. Sci. 476, 840–849 (2019)
Y. Zhang, B. Cui, O. Derr, Z. Yao, Z. Qin, X. Deng, J. Li, H. Lin, Hierarchical cobalt-based hydroxide microspheres for water oxidation. Nanoscale 6, 3376–3383 (2014)
M.J. Szary, Al substituted MoS2 for adsorption-based water collection. Appl. Surf. Sci. 529, 147083 (2020)
C. Gu, G. Zhou, J. Yang, H. Pang, M. Zhang, Q. Zhao, X. Gu, S. Tian, J. Zhang, L. Xu, Y. Tang, NiS/MoS2 Mott-Schottky heterojunction-induced local charge redistribution for high-efficiency urea-assisted energy-saving hydrogen production, Chem. Eng. J. 443 (2022)
L. Liu, S.J. Tan, T. Horikawa, D.D. Do, D. Nicholson, J. Liu, Water adsorption on carbon - a review. Adv Colloid Interface Sci 250, 64–78 (2017)
J. Zhu, L. Hu, P. Zhao, L.Y.S. Lee, K.Y. Wong, Recent advances in electrocatalytic hydrogen evolution using nanoparticles. Chem Rev 120, 851–918 (2020)
J.Y. Zhang, H. Wang, Y. Tian, Y. Yan, Q. Xue, T. He, H. Liu, C. Wang, Y. Chen, B.Y. **a, Anodic hydrazine oxidation assists energy-efficient hydrogen evolution over a bifunctional cobalt perselenide nanosheet electrode. Angew. Chem. Int. Ed. 57, 7649–7653 (2018)
Funding
This work was supported by the Foundation of the Undergraduate Innovation and Entrepreneurship Training Program of Guangxi University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cao, Z., Song, H., Liu, C. et al. Vacancy-Assisted Fast Electron Transport Non-noble Metal Electrocatalyst Mn0.09-MoS2 for Hydrogen Evolution Reaction. Electrocatalysis 13, 807–817 (2022). https://doi.org/10.1007/s12678-022-00765-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12678-022-00765-z