Abstract
Clear cell renal cell carcinoma (ccRCC) is one of the most aggressive urological malignancies and a highly immunogenic cancer. Yet, its pathogenesis is still not fully understood. This study analyzed the role of the miR-320 family in ccRCC using bioinformatics algorithms and a series of in vitro experiments. miR-4429 was found to be significantly down-regulated in ccRCC tissues and cell lines, while overexpression of miR-4429 significantly inhibited renal cancer cell proliferation, migration, and invasion in vitro. In addition, the UALCAN database, immunohistochemistry, and protein blotting results showed that CD274 expression was up-regulated in ccRCC tissues and correlated with higher histologic grading. Dual luciferase assay indicated that CD274 was a direct target of miR-4429. Overexpression of miR-4429 in 786-O, Caki-2 cells significantly inhibited CD274 expression. KEGG results indicated that the potential target function of miR-4429 was associated with the PI3K/AKT signaling pathway, and protein blotting verified the results. In summary, this data shows that miR-4429 targets CD274 and inhibits ccRCC proliferation, migration, and invasion by regulating PI3K/AKT signaling, thus potentially providing a promising therapeutic target and prognostic biomarker for renal cell carcinoma patients.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Renal cell carcinoma (RCC) ranks as the third most prevalent urinary tract neoplasm, constituting about 90% of renal malignancies and 4% of all malignant tumours in the adult population [1]. Clear cell RCC (ccRCC), the most common and aggressive RCC subtype, represents 70–75% of RCC cases [2]. Owing to its elusive early clinical manifestations, more than 30% of ccRCC patients manifest metastases at their initial diagnosis, thereby forgoing surgical options. Additionally, roughly 40% of patients who underwent primary ccRCC resection still experienced cancer recurrence and metastasis, resulting in an overall 5-year survival rate less than 10% [3, 4]. Consequently, exploring the molecular underpinnings of ccRCC development and identifying potential targets for ccRCC assume paramount importance in enhancing ccRCC treatment.
Micro RNAs (miRNAs) are a class of small non-coding RNAs, consisting of approximately 22 nucleotides, that are indispensable factors in the regulation of tumorigenesis and metastasis [5]. miRNAs function as either tumour suppressors or oncomiRNAs by binding to the 3′-untranslated region (3′-UTR) of target genes, thereby downregulating the transcription of associated genes [6]. Extensive reports indicate that miR-320 family members, including miR-320a, miR-320b, miR-320c, miR-320d, and miR-4429 are downregulated in various malignancies and have been linked to tumour aggressiveness and an unfavourable prognosis [7,8,9,10,2.9 Cell migration and invasion assays We employed Transwell chambers (Corning Costar Corp., Cambridge, MA, USA) with 8 μm pores, placed in 24-well culture plates. For the invasion assay, matrix gel (BD Bioscience, USA) stored at − 20 °C was allowed to dissolve overnight at 4 °C. It was subsequently diluted in a 1:8 ratio with FBS-free cell-affiliated medium, and 100 µL of the gel mixture was used to coat the upper chamber evenly, followed by an incubation period at 37 °C for 2 h. 786-O and Caki-2 cells, transfected for 48 h, were diluted with RPMI 1640 and McCoy's 5A medium without FBS, to a concentration of 5 × 105 cells per 200 µL, and seeded into the upper chamber. The lower chamber was supplemented with RPMI 1640 and McCoy's 5A medium containing 10% FBS. After 48 h of incubation, cells were fixed with 4% PFA for 15 min at room temperature and then stained with 1% crystal violet for 20 min. Subsequently, five different field of view statistics were taken at random. To prepare cell lysates, cells were collected at 70–80% confluence 48 h post-transfection. Protein concentrations were determined, and a loading buffer was prepared according to the Solabank BCA Protein Concentration Assay Kit (Bei**g, China). The prepared loading buffer was applied to individual lanes on a 4–15% gradient polyacrylamide gel (Mini-PROTEAN TGX™, Bio-Rad). Blotting was executed using a gel transfer system (Invitrogen) to transfer proteins onto PVDF membranes, which were then immersed in TBST (TBS containing 5% skim milk powder and 0.1% Tween) for 2 h. Following this, the blotted membranes were incubated with primary antibodies at a concentration of 1:1000 overnight at 4 °C. After washing the membrane three times, it was incubated with rabbit-specific secondary antibody (1:3000) for 2 h at room temperature. To develop the blotted membrane, the membrane was washed thrice with the ELC kit (New Cell & Molecular Biotech Co., Ltd. SuZhou, China), and the grayscale values of the target bands were assessed utilising Image J. GAPDH expression level was employed to normalise the target proteins. All antibodies were sourced from Abcam, including CD274 (ab205921), PI3K (ab302958), p-PI3K (#4228), p-AKT (ab8805), AKT (ab8805), and GAPDH (ab8245). We amplified the 3′-UTR of CD274, which carried the putative binding site for miR-4429, and then cloned it into the pmirGLO vector. To eliminate complementarity with miR-4429, the miR-4429 complementary site within the CD274 3′-UTR, with the sequence 5′-CAGCUUU-3′, was individually mutated. 293T cells were seeded into 96-well plates at a density of 4000 cells per well and transfected with luciferase reporter vectors containing either the wild-type (WT) or mutated (MUT) CD274 3′-UTR (constructed by Ribobio), along with miR-4429 mimics or a negative control (NC). After 48 h of transfection, cells were harvested, and the Dual-Luciferase® Reporter Assay System was utilised to measure luciferase activity. Normalised luciferase activity was determined as the ratio of luciferase activity to Renilla luciferase activity. For immunohistochemistry, the hydrated sections were treated with an antigen repair solution (Shanghai Shumba Biotech; Shanghai, China). Subsequently, the slides were soaked in 3% H2O2 for 20 min and then incubated with Primary antibody (CD274, 1:150) was incubated at 4 °C for 12 h. The following day, the slides were rinsed and incubated with rabbit secondary antibody at 15 °C for 90 min and then transferred to 37 °C for 30 min. Separate staining with 3,3′-diaminobenzidine (DAB) and hematoxylin was conducted. The slides were then assessed and captured using an Olympus BX53 fluorescence microscope (Tokyo, Japan). We conducted statistical analyses utilising SPSS 19.0 (IBM SPSS, Chicago, IL) and GraphPad Prism 6.0 (IBM, Armonk, NY, USA). Paired and unpaired samples were analyzed for statistical significance using t-tests. For samples with more than two groups, analysis of variance (ANOVA) was used. Correlations between gene expression levels were analyzed using Spearman's correlation analysis. All experiments were repeated at least three times. Statistical significance was set at P values < 0.05.2.10 Western blot
2.11 Dual-luciferase reporter assay
2.12 Immunohistochemical staining
2.13 Statistical analysis
3 Results
3.1 MiR-4429 is significantly down-regulated in ccRCC tissues
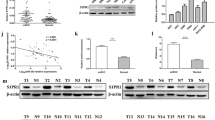
To investigate the potential association of miR-320 family members with the progression of ccRCC, we conducted a comprehensive analysis of their expression patterns utilising the TCGA database and bioinformatics algorithms. The analysis was based on three dimensions, including differential expression, prognosis, and metastasis (relative to normal tissue) in ccRCC. In terms of prognostic stratification, we segregated patients into two groups: those with less than one year of survival and those with over five years of survival. In the context of metastasis, ccRCC patients were categorised into metastatic (M1) and non-metastatic (M0) groups.
Differential expression analysis revealed up-regulation of miR-320 family members, including miR-320a, miR-320b, miR-320c, and miR-320d, as depicted in Fig. 1a. However, when considering prognosis (Fig. 1b), there was no significant differences. In contrast, miR-320b exhibited significant variation in the context of the metastatic group, as shown in Fig. 1c.
MiR-4429 was significantly downregulated in ccRCC tissues a–c MiR-320a, miR-320b, miR-320c, and miR-320d expression were up-regulated in the a differentially expressed group compared to normal tissues. miR-320 family members were not significantly different in the b prognosis group, and only miR-320b was significantly different in the c metastasis group, d The expression levels of miR-320 family members were detected in the tissues of 30 ccRCC patients, and miR-4429 expression was significantly down-regulated. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001
To further validate the expression patterns of miR-320 family members in ccRCC patients, we conducted RT-qPCR to assess their levels in 30 ccRCC tumour samples and compared with control tissues. The outcomes revealed a noteworthy down-regulation of miR-4429 in ccRCC tissues. However, the expression levels of miR-320a, miR-320b, miR-320c, and miR-320d did not exhibit statistically significant differences, as illustrated in Fig. 1d. This finding suggests that miR-4429, a member of the miR-320 family, may serve as an independent biomarker in the context of ccRCC.
3.2 Overexpression of miR-4429 inhibits proliferation, migration, and invasion of 786-O and Caki-2 cells in vitro
In order to elucidate the involvement of miR-320 family members in the proliferation and metastasis of ccRCC, we employed miR-4429, a representative of this family, and utilised RT-qPCR to assess its expression levels in various cell lines including HK-2, ACHN, 786-O, Caki-1, and Caki-2. Subsequently, we transfected 786-O and Caki-2 cells with mimic, inhibitor, and negative control to gauge transfection efficiency. The biological function of miR-4429 was rigorously examined through a battery of assays, including cell viability assay performed with CCK-8, clone formation assay, wound healing assay, and Transwell assay. Predictive target analysis of miR-4429 was conducted using miRDB and TargetScanv7.1, and all putative targets underwent functional enrichment analysis employing the KEGG database.
Our findings unveil a substantial downregulation of miR-4429 in 786-O and Caki-2 cell lines (Fig. 2a). Furthermore, the expression level of miR-4429 experienced a significant upregulation following transfection with miR-4429-mimic (Fig. 2b). Importantly, this overexpression of miR-4429 exerted a notable inhibitory effect on cell proliferation (Fig. 2c, d), migration, and invasion (Fig. 2e–g). These compelling results posit miR-4429 as a promising candidate in the role of a tumour suppressor in ccRCC.
MiR-4429 overexpression inhibited proliferation, migration, and invasion of 786-O and Caki-2 cells in vitro a miR-4429 mRNA levels were significantly down-regulated in renal cancer cell lines compared to HK-2 in renal tubular epithelial cells. b Transfection of miR-4429-mimic in 786-O and Caki-2 cells significantly up-regulated miR-4429 mRNA levels. c–f Overexpression of miR-4429 significantly inhibited cell proliferation rate (c), clone formation (d), wound healing (e), migration (f), and invasion (g). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001
3.3 CD274, a potential target of miR-4429, has elevated expression in ccRCC and positively correlates with histologic grading
As widely acknowledged, immune cell infiltration within the tumor microenvironment (TME) can profoundly influence the effectiveness of immunotherapy and the subsequent prognosis of ccRCC patients. Extensive research has demonstrated that CD274 (PD-L1) is prominently overexpressed on the surface of malignant tumour cells [12, 29]. In our research, we definitively identified CD274 as a direct target of miR-4429 and established a negative correlation between miR-4429 and CD274 mRNA expression levels. Our in vitro experiments demonstrated that miR-4429 overexpression significantly curtails the proliferation, migration, and invasion of ccRCC cells. Additionally, our analysis of the UALCAN database and clinical specimens unveiled the upregulation of CD274 in ccRCC and its positive correlation with the Fuhrman grade and the infiltration of PD-1, Immune infiltrating cells. Hence, the combinatory evaluation of miR-4429 and CD274 bears great potential for assessing the clinical prognosis of ccRCC patients, suggesting a plausible regulatory mechanism for CF274 as an oncogene in ccRCC.
While multiple studies have illuminated the association between abnormal microRNA expression and various intrinsic tumour processes, showcasing the substantial impact of microRNA up- or down-regulation in suppressing tumour cell proliferation, migration, in vitro invasiveness, and subcutaneous tumour growth, the clinical application of miRNA regulation and its targeted therapies remains constrained by limitations. Significantly, ccRCC as an immune-responsive tumour, is likely intertwined with immune system orchestration throughout its development and progression. Hence, modulating the immune response emerges as a promising avenue for ccRCC therapy. Consequently, delving into the potential of the miR-4429/CD274 axis as a target for immunomodulation and immunotherapy in ccRCC is merited. However, further investigations are warranted to ascertain whether CD274 fosters ccRCC progression by sustaining the activation of the PI3K/AKT signalling pathway.
In summary, our study has elucidated that miR-4429 overexpression yields a significant impediment to ccRCC progress, chiefly mediated through the PI3K/AKT signalling pathway. We have pinpointed CD274 as a direct and functional target of miR-4429, with its expression levels correlating with higher histological grading in ccRCC. Therefore, the newfound miR-4429/CD274 axis holds promise as a therapeutic target and prognostic biomarker for ccRCC patients.
Data availability
Data is provided within the manuscript or supplementary information files.
References
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71(1):7–33.
Shuch B, Amin A, Armstrong AJ, Eble JN, Ficarra V, Lopez-Beltran A, Martignoni G, Rini BI, Kutikov A. Understanding pathologic variants of renal cell carcinoma: distilling therapeutic opportunities from biologic complexity. Eur Urol. 2015;67(1):85–97.
Koul H, Huh JS, Rove KO, Crompton L, Koul S, Meacham RB, Kim FJ. Molecular aspects of renal cell carcinoma: a review. Am J Cancer Res. 2011;1(2):240–54.
Sanchez-Gastaldo A, Kempf E, Gonzalez Del Alba A, Duran I. Systemic treatment of renal cell cancer: a comprehensive review. Cancer Treat Rev. 2017;60:77–89.
Jafri MA, Al-Qahtani MH, Shay JW. Role of miRNAs in human cancer metastasis: implications for therapeutic intervention. Semin Cancer Biol. 2017;44:117–31.
Budakoti M, Panwar AS, Molpa D, Singh RK, Busselberg D, Mishra AP, Coutinho HDM, Nigam M. Micro-RNA: the darkhorse of cancer. Cell Signal. 2021;83: 109995.
Wu YY, Chen YL, Jao YC, Hsieh IS, Chang KC, Hong TM. miR-320 regulates tumor angiogenesis driven by vascular endothelial cells in oral cancer by silencing neuropilin 1. Angiogenesis. 2014;17(1):247–60.
Schepeler T, Reinert JT, Ostenfeld MS, Christensen LL, Silahtaroglu AN, Dyrskjot L, Wiuf C, Sorensen FJ, Kruhoffer M, Laurberg S, Kauppinen S, Orntoft TF, Andersen CL. Diagnostic and prognostic microRNAs in stage II colon cancer. Cancer Res. 2008;68(15):6416–24.
Gattolliat CH, Uguen A, Pesson M, Trillet K, Simon B, Doucet L, Robaszkiewicz M, Corcos L. MicroRNA and targeted mRNA expression profiling analysis in human colorectal adenomas and adenocarcinomas. Eur J Cancer. 2015;51(3):409–20.
Ak G, Tomaszek SC, Kosari F, Metintas M, Jett JR, Metintas S, Yildirim H, Dundar E, Dong J, Aubry MC, Wigle DA, Thomas CF Jr. MicroRNA and mRNA features of malignant pleural mesothelioma and benign asbestos-related pleural effusion. Biomed Res Int. 2015;2015: 635748.
Zhang T, Zou P, Wang T, **ang J, Cheng J, Chen D, Zhou J. Down-regulation of miR-320 associated with cancer progression and cell apoptosis via targeting Mcl-1 in cervical cancer. Tumour Biol. 2016;37(7):8931–40.
Liu J, Chen Z, Li Y, Zhao W, Wu J, Zhang Z. PD-1/PD-L1 checkpoint inhibitors in tumor immunotherapy. Front Pharmacol. 2021;12: 731798.
Yu W, Hua Y, Qiu H, Hao J, Zou K, Li Z, Hu S, Guo P, Chen M, Sui S, **ong Y, Li F, Lu J, Guo W, Luo G, Deng W. PD-L1 promotes tumor growth and progression by activating WIP and beta-catenin signaling pathways and predicts poor prognosis in lung cancer. Cell Death Dis. 2020;11(7):506.
Gao Y, Yang J, Cai Y, Fu S, Zhang N, Fu X, Li L. IFN-gamma-mediated inhibition of lung cancer correlates with PD-L1 expression and is regulated by PI3K-AKT signaling. Int J Cancer. 2018;143(4):931–43.
Wang F, Yang L, **ao M, Zhang Z, Shen J, Anuchapreeda S, Tima S, Chiampanichayakul S, **ao Z. PD-L1 regulates cell proliferation and apoptosis in acute myeloid leukemia by activating PI3K-AKT signaling pathway. Sci Rep. 2022;12(1):11444.
Choueiri TK, Halabi S, Sanford BL, Hahn O, Michaelson MD, Walsh MK, Feldman DR, Olencki T, Picus J, Small EJ, Dakhil S, George DJ, Morris MJ. Cabozantinib versus sunitinib as initial targeted therapy for patients with metastatic renal cell carcinoma of poor or intermediate risk: the alliance A031203 CABOSUN trial. J Clin Oncol. 2017;35(6):591–7.
Motzer RJ, Barrios CH, Kim TM, Falcon S, Cosgriff T, Harker WG, Srimuninnimit V, Pittman K, Sabbatini R, Rha SY, Flaig TW, Page R, Bavbek S, Beck JT, Patel P, Cheung FY, Yadav S, Schiff EM, Wang X, Niolat J, Sellami D, Anak O, Knox JJ. Phase II randomized trial comparing sequential first-line everolimus and second-line sunitinib versus first-line sunitinib and second-line everolimus in patients with metastatic renal cell carcinoma. J Clin Oncol. 2014;32(25):2765–72.
Falagario UG, Veccia A, Cormio L, Simeone C, Carbonara U, Crocerossa F, Antonelli A, Porpiglia F, Carrieri G, Autorino R. Nomogram predicting 30-day mortality after nephrectomy in the contemporary era: results from the SEER database. Int J Urol. 2021;28(3):309–14.
Moch H, Cubilla AL, Humphrey PA, Reuter VE, Ulbright TM. The 2016 WHO classification of tumours of the urinary system and male genital organs-part A: renal penile, and testicular tumours. Eur Urol. 2016;70(1):93–105.
Iorio MV, Croce CM. MicroRNA dysregulation in cancer: diagnostics, monitoring and therapeutics. A comprehensive review. EMBO Mol Med. 2012;4(3):143–59.
Sun H, Fan G, Deng C, Wu L. miR-4429 sensitized cervical cancer cells to irradiation by targeting RAD51. J Cell Physiol. 2020;235(1):185–93.
Zhu YM, Chen P, Shi L, Zhu T, Chen X. MiR-4429 suppresses the malignant development of ovarian cancer by targeting YOD1. Eur Rev Med Pharmacol Sci. 2020;24(17):8722–30.
Zhou S, Qian K, Yu S, Zhao Y, Shen Q, Li Y. MiR-4429 alleviates malignant behaviors of lung adenocarcioma through Wnt/beta-catenin pathway. Cancer Biother Radiopharm. 2021. https://doi.org/10.1089/cbr.2021.0154.
Pan H, Hong Y, Yu B, Li L, Zhang X. miR-4429 inhibits tumor progression and epithelial-mesenchymal transition via targeting CDK6 in clear cell renal cell carcinoma. Cancer Biother Radiopharm. 2019;34(5):334–41.
Li HB, You QS, Xu LX, Sun LX, Abdul Majid AS, **a XB, Ji D. Long non-coding RNA-MALAT1 mediates retinal ganglion cell apoptosis through the PI3K/Akt signaling pathway in rats with glaucoma. Cell Physiol Biochem. 2017;43(5):2117–32.
Lipson EJ, Sharfman WH, Drake CG, Wollner I, Taube JM, Anders RA, Xu H, Yao S, Pons A, Chen L, Pardoll DM, Brahmer JR, Topalian SL. Durable cancer regression off-treatment and effective reinduction therapy with an anti-PD-1 antibody. Clin Cancer Res. 2013;19(2):462–8.
Ueda K, Suekane S, Kurose H, Chikui K, Nakiri M, Nishihara K, Matsuo M, Kawahara A, Yano H, Igawa T. Prognostic value of PD-1 and PD-L1 expression in patients with metastatic clear cell renal cell carcinoma. Urol Oncol. 2018;36(11):499e9–16.
Leite KR, Reis ST, Junior JP, Zerati M, de Gomes O, Camara-Lopes LH, Srougi M. PD-L1 expression in renal cell carcinoma clear cell type is related to unfavorable prognosis. Diagn Pathol. 2015;10:189.
Stenzel PJ, Schindeldecker M, Tagscherer KE, Foersch S, Herpel E, Hohenfellner M, Hatiboglu G, Alt J, Thomas C, Haferkamp A, Roth W, Macher-Goep**er S. Prognostic and predictive value of tumor-infiltrating leukocytes and of immune checkpoint molecules PD1 and PDL1 in clear cell renal cell carcinoma. Transl Oncol. 2020;13(2):336–45.
Funding
This research was funded by the Department of Science and Technology of Guizhou Province, China (No. Qiankehe Support [2021] General 085) and National Natural Science Foundation of China (No. 82160145).
Author information
Authors and Affiliations
Contributions
GYH and YKW wrote the paper and performed the experiments. SYH and YH analyzed the statistics. GYH and CCG conceptualized and engineered the experiments. All authors read and approved the final manuscript for publication.
Corresponding authors
Ethics declarations
Institutional review board
This research was executed in conformity with the guiding principles of the Declaration of Helsinki and was authorized by the Ethics Committee of Guizhou Provincial People’s Hospital.
Informed consent
Informed consent was obtained from all subjects involved in the study.
Competing interests
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hong, G., Wu, Y., Huang, S. et al. miR-4429 inhibits ccRCC proliferation, migration, and invasion by directly targeting CD274. Discov Onc 15, 190 (2024). https://doi.org/10.1007/s12672-024-01055-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12672-024-01055-4