Abstract
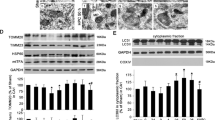
Oxidative stress and mitochondrial dysfunction are involved in cerebral ischemia/reperfusion injury-induced neuronal apoptosis. Mitophagy is the main method to eliminate dysfunctional mitochondria. Apelin-36, a type of neuropeptide, has been reported to exert protective effects in cerebral I/R (I/R) injury, but its precise mechanisms remain to be elucidated. To study the effects of Apelin-36 on oxidative stress and mitochondrial dysfunction in cerebral I/R injury, the oxygen-glucose deprivation/reperfusion (OGD/R) model with 6 h of ischemia and 6 h of reperfusion was established in HT22 cells. Results demonstrated that Apelin-36 protected against OGD/R injury by improving cell viability, decreasing the apoptotic cells ratio and increasing the ratio of Bcl-2/Bax. In addition, Apelin-36 treatment inhibited oxidative stress by downregulating the level of reactive oxygen species (ROS) and malondialdehyde (MDA) as well as the expression of inducible nitric oxide synthase (iNOS). And Apelin-36 also activated the level of superoxide dismutase (SOD) and glutathione (GSH). Mitochondrial apoptosis was also alleviated with Apelin-36 treatment detected by the mitochondrial membrane potential (MMP) and the expression of Cytochrome c (Cyt c), Cleaved caspase-9, and Cleaved caspase-3. Furthermore, the SIRT1-mediated PINK1/Parkin-dependent mitophagy was activated by Apelin-36 treatment with the downregulation of p62 and upregulation of LC3B-II and Beclin1. Both EX527 and Cyclosporine A (CsA), which are inhibitors of SIRT1 and mitophagy, markedly alleviated the inhibition of oxidative stress and mitochondrial dysfunction caused by Apelin-36. These findings suggest that SIRT1-mediated PINK1/Parkin-dependent mitophagy is involved in the neuroprotective effects of Apelin-36 on OGD/R-induced oxidative stress and mitochondrial dysfunction.








Similar content being viewed by others
Data Availability
All data generated and analyzed in this study are included in this article.
References
Ahmad S, Khan SA, Kindelin A, Mohseni T, Bhatia K, Hoda MN, Ducruet AF (2019) Acetyl-11-keto-β-boswellic acid (AKBA) attenuates oxidative stress, inflammation, complement activation and cell death in brain endothelial cells following OGD/reperfusion. Neuro Mol Med 21:505–516. https://doi.org/10.1007/s12017-019-08569-z
Annesley SJ, Fisher PR (2019) Mitochondria in health and disease. Cells 8. https://doi.org/10.3390/cells8070680
Baek SH, Noh AR, Kim KA, Akram M, Shin YJ, Kim ES, Yu SW, Majid A, Bae ON (2014) Modulation of mitochondrial function and autophagy mediates carnosine neuroprotection against ischemic brain damage. Stroke 45:2438–2443. https://doi.org/10.1161/strokeaha.114.005183
Benjamin EJ et al (2019) Heart disease and stroke statistics-2019 update: a report from the American Heart Association. Circulation 139:e56–e528. https://doi.org/10.1161/CIR.0000000000000659
Britz G, Ferrell A (2013) Developments on the horizon in the treatment of neurovascular problems. Surg Neurol Int 4(Suppl 1):S31. https://doi.org/10.4103/2152-7806.109194
Chen T, Zhu J, Wang YH, Hang CH (2019) ROS-mediated mitochondrial dysfunction and ER stress contribute to compression-induced neuronal injury. Neuroscience 416:268–280. https://doi.org/10.1016/j.neuroscience.2019.08.007
Cheng B, Chen J, Bai B, **n Q (2012) Neuroprotection of apelin and its signaling pathway. Peptides 37:171–173. https://doi.org/10.1016/j.peptides.2012.07.012
Dan Dunn J, Alvarez LAJ, Zhang X, Soldati T (2015) Reactive oxygen species and mitochondria: a nexus of cellular homeostasis. Redox Biol 6:472–485. https://doi.org/10.1016/j.redox.2015.09.005
Diebold L, Chandel NS (2016) Mitochondrial ROS regulation of proliferating cells. Free Radic Biol Med 100:86–93. https://doi.org/10.1016/j.freeradbiomed.2016.04.198
Elfawy HA, Das B (2019) Crosstalk between mitochondrial dysfunction, oxidative stress, and age related neurodegenerative disease: etiologies and therapeutic strategies. Life Sci 218:165–184. https://doi.org/10.1016/j.lfs.2018.12.029
Gong Z, Pan J, Shen Q, Li M, Peng Y (2018) Mitochondrial dysfunction induces NLRP3 inflammasome activation during cerebral ischemia/reperfusion injury. J Neuroinflammation 15(1):1–7. https://doi.org/10.1186/s12974-018-1282-6
Guan R, Zou W, Dai X, Yu X, Liu H, Chen Q, Teng W (2018) Mitophagy, a potential therapeutic target for stroke. J Biomed Sci 25:87. https://doi.org/10.1186/s12929-018-0487-4
Guardia-Laguarta C, Liu Y, Lauritzen KH, Erdjument-Bromage H, Martin B, Swayne TC, Jiang X, Przedborski S (2019) PINK1 content in mitochondria is regulated by ER-associated degradation. J Neurosci 39:7074–7085. https://doi.org/10.1523/JNEUROSCI.1691-18.2019
Halestrap AP, Doran E, Gillespie JP, O’Toole A (2000) Mitochondria and cell death. Biochem Soc Trans 28:170–177. https://doi.org/10.1042/bst0280170
Hannibal L (2016) Nitric oxide homeostasis in neurodegenerative diseases. Curr Alzheimer Res 13:135–149. https://doi.org/10.2174/1567205012666150921101250
Jiang X, Wang X (2004) CytochromeC-mediated apoptosis. Annu Rev Biochem 73:87–106. https://doi.org/10.1146/annurev.biochem.73.011303.073706
Koyano F, Okatsu K, Kosako H, Tamura Y, Go E, Kimura M, Kimura Y, Tsuchiya H, Yoshihara H, Hirokawa T, Endo T, Fon EA, Trempe JF, Saeki Y, Tanaka K, Matsuda N (2014) Ubiquitin is phosphorylated by PINK1 to activate parkin. Nature 510:162–166. https://doi.org/10.1038/nature13392
Li L, Tan J, Miao Y, Lei P, Zhang Q (2015) ROS and autophagy: interactions and molecular regulatory mechanisms. Cell Mol Neurobiol 35:615–621. https://doi.org/10.1007/s10571-015-0166-x
Li Q, Zhang T, Wang J, Zhang Z, Zhai Y, Yang GY, Sun X (2014) Rapamycin attenuates mitochondrial dysfunction via activation of mitophagy in experimental ischemic stroke. Biochem Biophys Res Commun 444:182–188. https://doi.org/10.1016/j.bbrc.2014.01.032
Liu T, Yang Q, Zhang X, Qin R, Shan W, Zhang H, Chen X (2020) Quercetin alleviates kidney fibrosis by reducing renal tubular epithelial cell senescence through the SIRT1/PINK1/mitophagy axis. Life Sci 257:118116. https://doi.org/10.1016/j.lfs.2020.118116
Lv SY, Chen WD, Wang YD (2020) The Apelin/APJ system in psychosis and neuropathy. Front Pharmacol 11:320. https://doi.org/10.3389/fphar.2020.00320
Meng X, Tan J, Li M, Song S, Miao Y, Zhang Q (2016) Sirt1: role under the condition of ischemia/hypoxia. Cell Mol Neurobiol 37:17–28. https://doi.org/10.1007/s10571-016-0355-2
Narne P, Pandey V, Phanithi PB (2017) Interplay between mitochondrial metabolism and oxidative stress in ischemic stroke: an epigenetic connection. Mol Cell Neurosci 82:176–194. https://doi.org/10.1016/j.mcn.2017.05.008
Newsholme P, Cruzat VF, Keane KN, Carlessi R, de Bittencourt PIH (2016) Molecular mechanisms of ROS production and oxidative stress in diabetes. Biochem J 473:4527–4550. https://doi.org/10.1042/bcj20160503c
O’Dowd BF, Heiber M, Chan A, Heng HHQ, Tsui LC, Kennedy JL, Shi X, Petronis A, George SR, Nguyen T (1993) A human gene that shows identity with the gene encoding the angiotensin receptor is located on chromosome 11. Gene 136:355–360. https://doi.org/10.1016/0378-1119(93)90495-o
Perelman A, Wachtel C, Cohen M, Haupt S, Shapiro H, Tzur A (2012) JC-1: alternative excitation wavelengths facilitate mitochondrial membrane potential cytometry. Cell Death Dis 3:e430–e430. https://doi.org/10.1038/cddis.2012.171
Qiu J, Wang X, Wu F, Wan L, Cheng B, Wu Y, Bai B (2017) Low dose of Apelin-36 attenuates ER stress-associated apoptosis in rats with ischemic stroke. Front Neurol 8:556. https://doi.org/10.3389/fneur.2017.00556
Rabinstein AA (2017) Treatment of acute ischemic stroke. CONTINUUM: Lifelong Learning in Neurology 23:62–81. https://doi.org/10.1212/con.0000000000000420
Rastogi RP, Singh SP, Häder DP, Sinha RP (2010) Detection of reactive oxygen species (ROS) by the oxidant-sensing probe 2′,7′-dichlorodihydrofluorescein diacetate in the cyanobacterium Anabaena variabilis PCC 7937. Biochem Biophys Res Commun 397:603–607. https://doi.org/10.1016/j.bbrc.2010.06.006
Rodrigo R, Fernandez-Gajardo R, Gutierrez R, Matamala J, Carrasco R, Miranda-Merchak A, Feuerhake W (2013) Oxidative stress and pathophysiology of ischemic stroke: novel therapeutic opportunities. CNS Neurol Disord Drug Targets 12:698–714. https://doi.org/10.2174/1871527311312050015
Shah FA, Kury LA, Li T, Zeb A, Koh PO, Liu F, Zhou Q, Hussain I, Khan AU, Jiang Y, Li S (2019) Polydatin attenuates neuronal loss via reducing neuroinflammation and oxidative stress in rat MCAO models. Front Pharmacol 10:663. https://doi.org/10.3389/fphar.2019.00663
Sinha N, Dabla P (2015) Oxidative stress and antioxidants in hypertension—a current review. Curr Hypertens Rev 11:132–142. https://doi.org/10.2174/1573402111666150529130922
Tang BL (2016) Sirt1 and the mitochondria. Mol Cells 39:87–95. https://doi.org/10.14348/molcells.2016.2318
Tang YC, Tian HX, Yi T, Chen HB (2016) The critical roles of mitophagy in cerebral ischemia. Protein Cell 7:699–713. https://doi.org/10.1007/s13238-016-0307-0
Tian Y, Chen R, Jiang Y, Bai B, Yang T, Liu H (2020) The protective effects and mechanisms of Apelin/APJ system on ischemic stroke: a promising therapeutic target. Front Neurol 11:75. https://doi.org/10.3389/fneur.2020.00075
Tolkovsky AM (2009) Mitophagy. Biochim Biophys Acta 1793:1508–1515. https://doi.org/10.1016/j.bbamcr.2009.03.002
Wang H, Chen S, Zhang Y, Xu H, Sun H (2019) Electroacupuncture ameliorates neuronal injury by Pink1/Parkin-mediated mitophagy clearance in cerebral ischemia-reperfusion. Nitric Oxide 91:23–34. https://doi.org/10.1016/j.niox.2019.07.004
Wu F, Qiu J, Fan Y, Zhang Q, Cheng B, Wu Y, Bai B (2018a) Apelin-13 attenuates ER stress-mediated neuronal apoptosis by activating Galphai/Galphaq-CK2 signaling in ischemic stroke. Exp Neurol 302:136–144. https://doi.org/10.1016/j.expneurol.2018.01.006
Wu X, Li X, Liu Y, Yuan N, Li C, Kang Z, Zhang X, **a Y, Hao Y, Tan Y (2018b) Hydrogen exerts neuroprotective effects on OGD/R damaged neurons in rat hippocampal by protecting mitochondrial function via regulating mitophagy mediated by PINK1/Parkin signaling pathway. Brain Res 1698:89–98. https://doi.org/10.1016/j.brainres.2018.06.028
Wu Y, Chen M, Jiang J (2019) Mitochondrial dysfunction in neurodegenerative diseases and drug targets via apoptotic signaling. Mitochondrion 49:35–45. https://doi.org/10.1016/j.mito.2019.07.003
**n Q, Cheng B, Pan Y, Liu H, Yang C, Chen J, Bai B (2015) Neuroprotective effects of apelin-13 on experimental ischemic stroke through suppression of inflammation. Peptides 63:55–62. https://doi.org/10.1016/j.peptides.2014.09.016
Yang JL, Mukda S, Chen SD (2018) Diverse roles of mitochondria in ischemic stroke. Redox Biol 16:263–275. https://doi.org/10.1016/j.redox.2018.03.002
Yao H, Zhang W, Wu H, Yang M, Wei P, Ma H, Duan J, Zhang C, Li Y (2018a) Sikokianin A from Wikstroemia indica protects PC12 cells against OGD/R-induced injury via inhibiting oxidative stress and activating Nrf2. Nat Prod Res 33:3450–3453. https://doi.org/10.1080/14786419.2018.1480019
Yao ZQ, Zhang X, Zhen Y, He XY, Zhao S, Li XF, Yang B, Gao F, Guo FY, Fu L, Liu XZ, Duan CZ (2018b) A novel small-molecule activator of Sirtuin-1 induces autophagic cell death/mitophagy as a potential therapeutic strategy in glioblastoma. Cell Death Dis 9(7):1–4. https://doi.org/10.1038/s41419-018-0799-z
Yi S, Zheng B, Zhu Y, Cai Y, Sun H, Zhou J (2020) Melatonin ameliorates excessive PINK1/Parkin-mediated mitophagy by enhancing SIRT1 expression in granulosa cells of PCOS. Am J Physiol Endocrinol Metab 319:E91–E101. https://doi.org/10.1152/ajpendo.00006.2020
Yu J, Wang WN, Matei N, Li X, Pang JW, Mo J, Chen SP, Tang JP, Yan M, Zhang JH (2020) Ezetimibe attenuates oxidative stress and neuroinflammation via the AMPK/Nrf2/TXNIP pathway after MCAO in rats. Oxidative Med Cell Longev 2020:1–14. https://doi.org/10.1155/2020/4717258
Zhang X, Yan H, Yuan Y, Gao J, Shen Z, Cheng Y, Shen Y, Wang RR, Wang X, Hu WW, Wang G, Chen Z (2014) Cerebral ischemia-reperfusion-induced autophagy protects against neuronal injury by mitochondrial clearance. Autophagy 9:1321–1333. https://doi.org/10.4161/auto.25132
Zhao N, **a J, Xu B (2020) Physical exercise may exert its therapeutic influence on Alzheimer’s disease through the reversal of mitochondrial dysfunction via SIRT1-FOXO1/3-PINK1-Parkin–mediated mitophagy. J Sport Health Sci. https://doi.org/10.1016/j.jshs.2020.08.009
Zhu J, Dou S, Wang C, Jiang Y, Wang C, Cheng B (2019a) Apelin-36 mitigates MPTP/MPP+-induced neurotoxicity: involvement of α-synuclein and endoplasmic reticulum stress. Brain Res 1721:146334. https://doi.org/10.1016/j.brainres.2019.146334
Zhu J, Dou S, Jiang Y, Bai B, Chen J, Wang C, Cheng B (2019b) Apelin-36 exerts the cytoprotective effect against MPP+-induced cytotoxicity in SH-SY5Y cells through PI3K/Akt/mTOR autophagy pathway. Life Sci 224:95–108. https://doi.org/10.1016/j.lfs.2019.03.047
Zhu J, Gao W, Shan X, Wang C, Wang H, Shao Z, Dou S, Jiang Y, Wang C, Cheng B (2020) Apelin-36 mediates neuroprotective effects by regulating oxidative stress, autophagy and apoptosis in MPTP-induced Parkinson’s disease model mice. Brain Res 1726:146493. https://doi.org/10.1016/j.brainres.2019.146493
Funding
The study was supported by the National Natural Science Foundation of China, Nos. 81870948 (to BB), 81671276 (to BHC), 81501018 (to CMW); the Natural Science Foundation of Shandong Province of China, No. ZR2014HL040 (to BHC); and Program Supporting Foundation for Teachers’ Research of **ing Medical University of China, No. JYFC2018KJ003 (to SSD). The funding sources had no role in study conception and design, data analysis or interpretation, paper writing, or deciding to submit this paper for publication.
Author information
Authors and Affiliations
Contributions
Study design: Baohua Cheng, Bo Bai; experimental implementation: Ziqi Shao, Shanshan Dou, Junge Zhu; data analysis: Ziqi Shao, Huiqing Wang, Dandan Xu, Chunmei Wang; manuscript writing: Ziqi Shao. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
The study was approved by the Ethical Committee of **ing Medical University, China (approval No. 2018-JS-001) in February 2018.
Conflict of Interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shao, Z., Dou, S., Zhu, J. et al. Apelin-36 Protects HT22 Cells Against Oxygen-Glucose Deprivation/Reperfusion-Induced Oxidative Stress and Mitochondrial Dysfunction by Promoting SIRT1-Mediated PINK1/Parkin-Dependent Mitophagy. Neurotox Res 39, 740–753 (2021). https://doi.org/10.1007/s12640-021-00338-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-021-00338-w