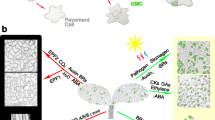
Abstract
Stomata are small pores on the surface of plant leaves that play a critical role in regulating gas exchange and water loss. The development of stomata is a complex process regulated by a network of signaling pathways and regulatory factors. In this review, we discuss the major signaling pathways and regulatory factors involved in stomatal development, including the roles of the EPF ligands family, the TMM and ERECTA receptor complex, the MAPK cascade, and various bHLH transcription factors such as SPCH. The interconnections between these pathways and regulatory factors are also summarized, revealing a complex network of interactions that integrate signals from multiple sources. Although significant progress has been made in understanding stomatal development, there are still knowledge gaps. Future studies should focus on elucidating the precise processes of the interaction between regulatory factors and signaling pathways, investigating the function of epigenetic regulation in stomatal development, and understanding the impact of environmental cues on it. This review provides insights into the complex intricate network that underlies stomatal development.


Similar content being viewed by others
Data Availability
Not applicable.
References
Abrash EB, Bergmann DC (2010) Regional specification of stomatal production by the putative ligand CHALLAH. Development 137(3):447–455. https://doi.org/10.1242/dev.040931. Epub 2010 Jan 7
Bemis SM, Lee JS, Shpak ED, Torii KU (2013) Regulation of floral patterning and organ identity by Arabidopsis ERECTA-family receptor kinase genes. J Exp Bot 64(17):5323–5333. https://doi.org/10.1093/jxb/ert270. Epub 2013 Sep 4
Bhave NS, Veley KM, Nadeau JA, Lucas JR, Bhave SL, Sack FD (2009) TOO MANY MOUTHS promotes cell fate progression in stomatal development of Arabidopsis stems. Planta 229:357–367. https://doi.org/10.1007/s00425-008-0835-9. Epub 2008 Nov 1
Buckley TN (2019) How do stomata respond to water status? New Phytol 224(1):21–36. https://doi.org/10.1111/nph.15899. Epub 2019 Jun 11
Chater CC, Oliver J, Casson S, Gray JE (2014) Putting the brakes on: abscisic acid as a central environmental regulator of stomatal development. New Phytol 202(2):376–391. https://doi.org/10.1111/nph.12713. Epub 2014 Mar 10
Chen L (2023) Emerging roles of protein phosphorylation in regulation of stomatal development. J Plant Physiol 280:153882. https://doi.org/10.1016/j.jplph.2022.153882. Epub 2022 Nov 26
Chen L, Wu Z, Hou S (2020) SPEECHLESS speaks loudly in stomatal development. Front Plant Sci 11:114. https://doi.org/10.3389/fpls.2020.00114. eCollection 2020
Chen L, Cochran AM, Waite JM, Shirasu K, Bemis SM, Torii KU (2023) Direct attenuation of Arabidopsis ERECTA signalling by a pair of U-box E3 ligases. Nat Plants 9(1):112–127. https://doi.org/10.1038/s41477-022-01303-x. Epub 2022 Dec 20
Dubey R, Pandey BK, Sawant SV, Shirke PA (2023) Drought stress inhibits stomatal development to improve water use efficiency in cotton. Acta Physiol Plant 45(2):30. https://doi.org/10.1007/s11738-022-03511-6
Endo H, Torii KU (2019) Stomatal development and perspectives toward agricultural improvement. Cold Spring Harb Perspect Biol 11(5):a034660. https://doi.org/10.1101/cshperspect.a034660
Engineer CB, Ghassemian M, Anderson JC, Peck SC, Hu H, Schroeder JI (2015) Carbonic anhydrases, EPF2 and a novel protease mediate CO2 control of stomatal development. Nature 526(7573):458. https://doi.org/10.1038/nature13452. Epub 2014 Jul 6
Fu ZW, Wang YL, Lu YT, Yuan TT (2016) Nitric oxide is involved in stomatal development by modulating the expression of stomatal regulator genes in Arabidopsis. Plant Sci 252:282–289. https://doi.org/10.1016/j.plantsci.2016.08.005. Epub 2016 Aug 10
Gray JE, Hetherington AM (2004) Plant Development: YODA the Stomatal switch. Curr Biol 14(12):R488–R490. https://doi.org/10.1016/j.cub.2004.06.019
Guo XY, Wang Y, Zhao PX, Xu P, Yu GH, Zhang LY, **ong Y, **ang CB (2019) AtEDT1/HDG11 regulates stomatal density and water-use efficiency via ERECTA and E2Fa. New Phytol 223(3):1478–1488. https://doi.org/10.1111/nph.15861. Epub 2019 May 15
Guo X, Ding X, Dong J (2022) Dichotomy of the BSL phosphatase signaling spatially regulates MAPK components in stomatal fate determination. Nat Commun 13(1):2438. https://doi.org/10.1038/s41467-022-30254-2
Guo T, Lu ZQ, **ong Y, Shan JX, Ye WW, Dong NQ, Kan Y, Yang YB, Zhao HY, Yu HX, Guo SQ, Lei JJ, Liao B, Chai J, Lin HX (2023) Optimization of rice panicle architecture by specifically suppressing ligand-receptor pairs. Nat Commun 14(1):1640. https://doi.org/10.1038/s41467-023-37326-x
Han S-K, Torii KU (2019) Linking cell cycle to stomatal differentiation. Curr Opin Plant Biol 51:66–73. https://doi.org/10.1016/j.pbi.2019.03.010. Epub 2019 May 7
Han S-K, Qi X, Sugihara K, Dang JH, Endo TA, Miller KL, Kim E-D, Miura T, Torii KU (2018) MUTE directly orchestrates cell-state switch and the single symmetric division to create stomata. Dev cell 45(3):303–315. https://doi.org/10.1016/j.devcel.2018.04.010
Hao Y, Zong X, Ren P, Qian Y, Fu A (2021) Basic Helix-Loop-Helix (bHLH) transcription factors regulate a wide range of functions in Arabidopsis. Int J Mol Sci 22(13):7152. https://doi.org/10.3390/ijms22137152
Herrmann A, Torii KU (2021) Shouting out loud: signaling modules in the regulation of stomatal development. Plant Physiol 185(3):765–780. https://doi.org/10.1093/plphys/kiaa061
Hofmann NR (2008) They all scream for ICE1/SCRM2: core regulatory units in stomatal development. Plant Cell 20(7):1732–1732. https://doi.org/10.1105/tpc.108.200711. Epub 2008 Jul 22
Hunt L, Gray JE (2009) The signaling peptide EPF2 controls asymmetric cell divisions during Stomatal Development. Curr Biol 19(10):864–869. https://doi.org/10.1016/j.cub.2009.03.069. Epub 2009 Apr 23
Hunt L, Bailey KJ, Gray JE (2010) The signalling peptide EPFL9 is a positive regulator of stomatal development. New Phytol 609–614. https://doi.org/10.1111/j.1469-8137.2010.03200.x. Epub 2010 Feb 10
Jewaria PK, Hara T, Tanaka H, Kondo T, Betsuyaku S, Sawa S, Sakagami Y, Aimoto S, Kakimoto T (2013) Differential effects of the peptides Stomagen, EPF1 and EPF2 on activation of MAP kinase MPK6 and the SPCH protein level. Plant Cell Physiol 54(8):1253–1262. https://doi.org/10.1093/pcp/pct076. Epub 2013 May 17
Jiao Q, Chen T, Niu G, Zhang H, Zhou C, Hong Z (2020) N-glycosylation is involved in stomatal development by modulating the release of active abscisic acid and auxin in Arabidopsis. J Exp Bot 71(19):5865–5879. https://doi.org/10.1093/jxb/eraa321
Jiao Z, Han S, Li Z, Huang M, Niu M-X, Yu X, Liu C, Wang H-L, Yin W, **a X (2022) PdEPFL6 reduces stomatal density to improve drought tolerance in poplar. Ind Crops Prod 182:114873. https://doi.org/10.1016/j.indcrop.2022.114873
Katsir L, Davies KA, Bergmann DC, Laux T (2011) Peptide signaling in plant development. Curr Biol 21(9): R356-R364. https://doi.org/10.1016/j.cub.2011.03.012
Kenta H, Ryoko K, Torii KU, Bergmann DC, Tatsuo KJG (2007) The secretory peptide gene EPF1 enforces the stomatal one-cell-spacing rule. Genes Dev 21(14):1720–1725. https://doi.org/10.1101/gad.1550707
Kollist H, Nuhkat M, Roelfsema MRG (2014) Closing gaps: linking elements that control stomatal movement. New Phytol 203(1):44–62. https://doi.org/10.1111/nph.12832. Epub 2014 May 6
Lampard GR, MacAlister CA, Bergmann DC (2008) Arabidopsis stomatal initiation is controlled by MAPK-mediated regulation of the bHLH SPEECHLESS. Science 322(5904):1113–1116. https://doi.org/10.1126/science.1162263
Lampard GR, Lukowitz W, Ellis BE, Bergmann DC (2009) Novel and expanded roles for MAPK signaling in Arabidopsis Stomatal Cell Fate revealed by cell type–specific manipulations. Plant Cell 21(11):3506–3517. https://doi.org/10.1105/tpc.109.070110. Epub 2009 Nov 6
Lau OS, Lau, Bergmann DC (2012) Stomatal development: a plant’s perspective on cell polarity, cell fate transitions and intercellular communication. Development 139(20):3683–3692. https://doi.org/10.1242/dev.080523
Lau OS, Davies KA, Chang J, Adrian J, Rowe MH, Ballenger CE, Bergmann DC (2014) Direct roles of SPEECHLESS in the specification of stomatal self-renewing cells. Science 345(6204):1605–1609. https://doi.org/10.1126/science.1256888. Epub 2014 Sep 4
Lau OS, Song Z, Zhou Z, Davies KA, Chang J, Yang X, Wang S, Lucyshyn D, Tay IHZ, Wigge PA, Bergmann DC (2018) Direct Control of SPEECHLESS by PIF4 in the high-temperature response of Stomatal Development. Curr Biol 28(8):1273–1280e1273. https://doi.org/10.1016/j.cub.2018.02.054. Epub 2018 Apr 5
Lawson T, Matthews J (2020) Guard cell metabolism and stomatal function. Annu Rev Plant Biol 71:273–302. https://doi.org/10.1146/annurev-arplant-050718-100251. Epub 2020 Mar 10
Lee JS, Kuroha T, Hnilova M, Khatayevich D, Kanaoka MM, McAbee JM, Sarikaya M, Tamerler C, Torii KU (2012) Direct interaction of ligand–receptor pairs specifying stomatal patterning. Genes Dev 26(2):126–136. https://doi.org/10.1101/gad.179895.111. Epub 2012 Jan 12
Lee E, Lucas JR, Goodrich J, Sack FD (2014a) Arabidopsis guard cell integrity involves the epigenetic stabilization of the FLP and FAMA transcription factor genes. Plant J 78(4):566–577. https://doi.org/10.1111/tpj.12516. Epub 2014 Apr 23
Lee E, Lucas JR, Sack FD (2014b) Deep functional redundancy between FAMA and FOUR LIPS in stomatal development. Plant J 78(4):555–565. https://doi.org/10.1111/tpj.12489. Epub 2014 Apr 23
Lee JH, Jung JH, Park CM (2017) Light inhibits COP1-Mediated degradation of ICE transcription factors to Induce Stomatal Development in Arabidopsis. Plant Cell 29(11):2817–2830. https://doi.org/10.1105/tpc.17.00371. Epub 2017 Oct 25
Li F-C, Wang J, Wu M-M, Fan C-M, Li X, He J-M (2017) Mitogen-activated protein kinase phosphatases affect UV-B-induced stomatal closure via controlling NO in guard cells. Plant Physio 173(1):760–770. https://doi.org/10.1104/pp.16.01656. Epub 2016 Nov 11
Li H, Yang Y, Wang H, Liu S, Jia F, Su Y, Li S, He F, Feng C, Niu M (2021) The receptor-like kinase ERECTA confers improved water use efficiency and drought tolerance to poplar via modulating stomatal density. Int J Mol Sci 22(14):7245. https://doi.org/10.3390/ijms22147245
Li M, Lv M, Wang X, Cai Z, Yao H, Zhang D, Li H, Zhu M, Du W, Wang R (2023a) The EPFL–ERf–SERK signaling controls integument development in Arabidopsis. New Phytol 238(1):186–201. https://doi.org/10.1111/nph.18701. Epub 2023 Jan 17
Li P, Chen L, Gu X, Zhao M, Wang W, Hou S (2023b) FOUR LIPS plays a role in meristemoid-to-GMC fate transition during stomatal development in Arabidopsis. Plant J 114(2):424–436. https://doi.org/10.1111/tpj.16146. Epub 2023 Mar 8
Lin G, Zhang L, Han Z, Yang X, Liu W, Li E, Chang J, Qi Y, Shpak ED, Chai J (2017) A receptor-like protein acts as a specificity switch for the regulation of stomatal development. Genes Dev 31(9). https://doi.org/10.1101/gad.297580.117. Epub 2017 May 23
McKown KH, Anleu Gil MX, Mair A, Xu SL, Raissig MT, Bergmann DC (2022) Expanded roles and divergent regulation of FAMA in Brachypodium and Arabidopsis stomatal development. Plant Cell 35(2):756–775. https://doi.org/10.1093/plcell/koac341
Meng L-S, Yao S-Q (2015) Transcription co-activator Arabidopsis ANGUSTIFOLIA3 (AN3) regulates water-use efficiency and drought tolerance by modulating stomatal density and improving root architecture by the transrepression of YODA (YDA). Plant Biotechnol J 13(7):893–902. https://doi.org/10.1111/pbi.12324. Epub 2015 Jan 20
Nunes TD, Zhang D, Raissig MT (2020) Form, development and function of grass stomata. Plant J 101(4):780–799. https://doi.org/10.1111/tpj.14552. Epub 2019 Nov 7
Ohashi ito K, Bergmann DC (2006) Arabidopsis FAMA controls the final proliferation/differentiation switch during stomatal development. Plant Cell 18(10):2493–2505. https://doi.org/10.1105/tpc.106.046136
Okuda S (2021) Molecular mechanisms of plant peptide binding to receptors. Peptides 144:170614. https://doi.org/10.1016/j.peptides.2021.170614. Epub 2021 Jul 29
Pillitteri LJ, Torii KU (2007) Breaking the silence: three bHLH proteins direct cell-fate decisions during stomatal development. BioEssays 29(9):861–870. https://doi.org/10.1002/bies.20625
Pillitteri LJ, Torii KU (2012) Mechanisms of stomatal development. Annu Rev Plant Biol 63:591–614. https://doi.org/10.1146/annurev-arplant-042811-105451. Epub 2012 Jan 30
Qi X, Han SK, Dang JH, Garrick JM, Ito M, Hofstetter AK, Torii KU (2017) Autocrine regulation of stomatal differentiation potential by EPF1 and ERECTA-LIKE1 ligand-receptor signaling. eLife 6:e24102. https://doi.org/10.7554/eLife.24102
Qi X, Yoshinari A, Bai P, Maes M, Zeng SM, Torii KU (2020) The manifold actions of signaling peptides on subcellular dynamics of a receptor specify stomatal cell fate. eLife 9:e58097. https://doi.org/10.7554/eLife.58097
Raissig MT, Matos JL, Anleu Gil MX, Kornfeld A, Bettadapur A, Abrash E, Allison HR, Badgley G, Vogel JP, Berry JA, Bergmann DC (2017) Mobile MUTE specifies subsidiary cells to build physiologically improved grass stomata. Science 355(6330):1215–1218. https://doi.org/10.1126/science.aal3254Epub 2017 Mar 16
Raschke K (1975) Stomatal action. Annu Rev Plant Physiol 26(1):309–340. https://doi.org/10.1146/annurev.pp.26.060175.001521
Rodriguez-Dominguez CM, Brodribb TJ (2020) Declining root water transport drives stomatal closure in olive under moderate water stress. New Phytol 225(1):126–134. https://doi.org/10.1111/nph.16177. Epub 2019 Sep 21
Samakovli D, Tichá T, Vavrdová T, Ovečka M, Luptovčiak I, Zapletalová V, Kuchařová A, Křenek P, Krasylenko Y, Margaritopoulou T (2020) YODA-HSP90 module regulates phosphorylation-dependent inactivation of SPEECHLESS to control stomatal development under acute heat stress in Arabidopsis. Mol Plant 13(4):612–633. https://doi.org/10.1016/j.molp.2020.01.001. Epub 2020 Jan 11
Seo H, Sepuru KM, Putarjunan A, Aguirre L, Burrows BA, Torii KU (2022) Intragenic suppressors unravel the role of the SCREAM ACT-like domain for bHLH partner selectivity in stomatal development. Proc Natl Acad Sci USA 119(9):e2117774119. https://doi.org/10.1073/pnas.2117774119
Shirakawa M, Ueda H, Nagano AJ, Shimada T, Kohchi T, Hara-Nishimura I (2014) FAMA is an essential component for the differentiation of two distinct cell types, myrosin cells and guard cells, in Arabidopsis. Plant Cell 26(10):4039–4052. https://doi.org/10.1105/tpc.114.129874. Epub 2014 Oct 10
Shpak ED, Jessica Messmer M, Lynn Jo P, Torii KU (2005) Stomatal patterning and differentiation by synergistic interactions of receptor kinases. Science 309(5732):290–293. https://doi.org/10.1126/science.1109710
Smit ME, Vatén A, Mair A, Northover CA, Bergmann DC (2023) Extensive embryonic patterning without cellular differentiation primes the plant epidermis for efficient post-embryonic stomatal activities. Dev Cell 58(6):506–521. https://doi.org/10.1016/j.devcel.2023.02.014. Epub 2023 Mar 16 e505
Sugano SS, Tomoo S, Yu I, Katsuya O, Atsushi T, Masashi M, Ikuko HNJN (2009) Stomagen positively regulates stomatal density in Arabidopsis. Nature 463(7278):241. https://doi.org/10.1038/nature08682. Epub 2009 Dec 9
Tatsuhiko K, Ryoko K, Aya M, Mayumi H, Touko NM, Satoko M, Yuichi M, Kazuhiro I, Yuki T, Shinobu TJP (2010) Stomatal density is controlled by a mesophyll-derived signaling molecule. Plant Cell Physiol 51(1):1–8. https://doi.org/10.1093/pcp/pcp180. Epub 2009 Dec 9
Torii KU (2021) Stomatal development in the context of epidermal tissues. Ann Bot 128(2):137–148. https://doi.org/10.1093/aob/mcab052
Uchida N, Lee JS, Horst RJ, Lai H-H, Kajita R, Kakimoto T, Tasaka M, Torii KU (2012) Regulation of inflorescence architecture by intertissue layer ligand–receptor communication between endodermis and phloem. Proc Natl Acad Sci USA 109(16):6337–6342. https://doi.org/10.1073/pnas.1117537109
Verma D, Bhagat PK, Sinha AK (2020) MKK3-MPK6-MYC2 module positively regulates ABA biosynthesis and signalling in Arabidopsis. J Plant Biochem Biotechnol 29(4):785–795. https://doi.org/10.1007/s13562-020-00621-5
Wakeel A, Wang L, Xu M (2021) SPEECHLESS and MUTE mediate feedback Regulation of Signal Transduction during stomatal development. Plants 10(3):432. https://doi.org/10.3390/plants10030432
Wallner E-S, Dolan L, Bergmann DC (2023) Arabidopsis stomatal lineage cells establish bipolarity and segregate differential signaling capacity to regulate stem cell potential. Dev Cell 58(18):1643–1656. https://doi.org/10.1016/j.devcel.2023.07.024. Epub 2023 Aug 21
Wang H, Ngwenyama N, Liu Y, Walker JC, Zhang S (2007) Stomatal development and patterning are regulated by environmentally responsive mitogen-activated protein kinases in Arabidopsis. Plant Cell 19(1):63–73. https://doi.org/10.1105/tpc.106.048298. Epub 2007 Jan 26
Wang Y, Xue X, Zhu J-K, Dong J (2016) Demethylation of ERECTA receptor genes by IBM1 histone demethylase affects stomatal development. Development 143(23):4452–4461. https://doi.org/10.1242/dev.129932. Epub 2016 Oct 3
Wang S, Zhou Z, Rahiman R, Lee GSY, Yeo YK, Yang X, Lau OS (2021) Light regulates stomatal development by modulating paracrine signaling from inner tissues. Nat Commun 12(1):3403. https://doi.org/10.1038/s41467-021-23728-2
Wu X, Cai C, Zhang X, Wu S, Wang R, Li N, Li Y, Sun Y, ang W (2022) ERECTA regulates seed size independently of its intracellular domain via MAPK-DA1-UBP15 signaling. Plant Cell 34(10):3773–3789. https://doi.org/10.1093/plcell/koac194
**e Z, Li D, Wang L, Sack FD, Grotewold E (2010) Role of the stomatal development regulators FLP/MYB88 in abiotic stress responses. Plant J 64(5):731–739. https://doi.org/10.1111/j.1365-313X.2010.04364.x. Epub 2010 Oct 13
Xu J, Zhang S (2015) Mitogen-activated protein kinase cascades in signaling plant growth and development. Trends Plant Sci 20(1):56–64. https://doi.org/10.1016/j.tplants.2014.10.001
Xue X, Bian C, Guo X, Di R, Dong J (2020) The MAPK substrate MASS proteins regulate stomatal development in Arabidopsis. PLoS Genet 16(4):e1008706. https://doi.org/10.1371/journal.pgen.1008706. eCollection 2020 Apr
Yamamuro C, Miki D, Zheng Z, Ma J, Wang J, Yang Z, Dong J, Zhu J-K (2014) Overproduction of stomatal lineage cells in Arabidopsis mutants defective in active DNA demethylation. Nat Commun 5(1):4062. https://doi.org/10.1038/ncomms5062
Yang K-Z, Jiang M, Wang M, Xue S, Zhu L-L, Wang H-Z, Zou J-J, Lee E-K, Sack F, Le J (2015) Phosphorylation of serine 186 of bHLH transcription factor SPEECHLESS promotes stomatal development in Arabidopsis. Mol Plant 8(5):783–795. https://doi.org/10.1016/j.molp.2014.12.014. Epub 2014 Dec 30
Yang S-L, Tran N, Tsai M-Y, Ho C-MK (2021) Misregulation of MYB16 expression causes stomatal cluster formation by disrupting polarity during asymmetric cell divisions. Plant Cell 34(1):455–476. https://doi.org/10.1093/plcell/koab260
Yang K-Z, Zuo C-R, Leng Y-J, Yue J-L, Liu H-C, Fan Z-B, Xue X-Y, Dong J, Chen L-Q, Le J (2022a) The functional specificity of ERECTA-family receptors in Arabidopsis stomatal development is ensured by molecular chaperones in the endoplasmic reticulum. Development 149(17):dev200892. https://doi.org/10.1242/dev.200892. Epub 2022 Sep 12
Yang X, Gavya SL, Zhou Z, Urano D, Lau OS (2022b) Abscisic acid regulates stomatal production by imprinting a SnRK2 kinase–mediated phosphocode on the master regulator SPEECHLESS. Sci Adv 8(40): eadd2063. 2063. Epub 2022 Oct 7 https://doi.org/10.1126/sciadv.add
Zhang M, Zhang S (2022) Mitogen-activated protein kinase cascades in plant signaling. J Integr Plant Biol 64(2):301–341. https://doi.org/10.1111/jipb.13215
Zhang Y, Guo X, Dong J (2016) Phosphorylation of the polarity protein BASL differentiates asymmetric cell fate through MAPKs and SPCH. Curr Biol 26(21):2957–2965. https://doi.org/10.1016/j.cub.2016.08.066. Epub 2016 Oct 13
Zhu J, Park J-H, Lee S, Lee JH, Hwang D, Kwak JM, Kim YJ (2020) Regulation of stomatal development by stomatal lineage miRNAs. Proc Natl Acad Sci USA 117(11):6237–6245. https://doi.org/10.1073/pnas.1919722117
Zoulias N, Harrison EL, Casson SA, Gray JE (2018) Molecular control of stomatal development. Biochem J 475(2):441–454. https://doi.org/10.1042/BCJ20170413
Zoulias N, Rowe J, Thomson EE, Dabrowska M, Sutherland H, Degen GE, Johnson MP, Sedelnikova SE, Hulmes GE, Hettema EH (2021) Inhibition of Arabidopsis stomatal development by plastoquinone oxidation. Curr Biol 31(24):5622–5632 e5627. https://doi.org/10.1016/j.cub.2021.10.018
Acknowledgements
This study was supported by the National Natural Science Foundation of China (32000197, 32200429), Yangzhou “Lv Yang **feng Project” excellent doctoral candidates (YZLYJFJH2021YXBS161). The authors wish to thank the two anonymous reviewers for their careful reading and their many valuable and insightful comments which helped improve the quality of the manuscript.
Author information
Authors and Affiliations
Contributions
Qingsong Jiao, Investigation, writing– original draft. Xueyun Hu, Conceptualization, supervision, review and editing. Hongbao Bai and Ahmad Zada manuscript review. All authors contributed to revising the manuscript. All authors have approved the submitted version. None of the authors have a conflict of interest to disclose.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jiao, Q., Bai, H., Zada, A. et al. Intrinsic Signaling Pathways and key Regulatory Factors of Stomatal Development. J. Plant Biol. 67, 185–194 (2024). https://doi.org/10.1007/s12374-024-09427-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12374-024-09427-7