Abstract
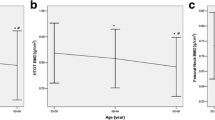
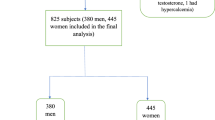
Osteoporosis a major public health problem of the elderly, is associated with substantial morbidity and socio economic burden. The aim of the study was to screen women with low bone mass using the indigenously developed Osteocalcin (OC) ELISA kit and compare it with commercial ELISA kit and evaluate. The diagnostic potential of the assay was assessed in 359 samples from neighboring tertiary care hospitals over a period of 2 years. OC levels were estimated by the developed indigenous assay in samples, correlated with the Bone Mineral Density (BMD) measurements and compared by a commercial ELISA kit. On the basis of T-scores the women were stratified into Normal and case groups as Osteopenia and Osteoporosis. The serum biochemical parameters calcium and phosphorus were estimated on an auto-analyzer. To compare two different assays Bland–Altman plot and Deming linear regression analysis was performed. The prevalence of Osteopenia was high (56%) and Osteoporosis (13%) in the healthy Indian women aged 21–65 years with significant differences in OC levels in normal and women with low bone mass. Good correlation (p < 0.0001) in the OC levels by the two assays was observed. Cut off limits established earlier with indigenous assay (11.9 ng/mL and 14.9 ng/mL) for Osteopenia and Osteoporosis were similar to those with the commercial kit (13.2 ng/mL and 16.8 ng/mL) respectively. The diagnostic sensitivity, specificity and accuracy of the OC prototype was > 85%. The cost effective OC prototype can be used in screening and management of Indian women with low bone mass.




Similar content being viewed by others
References
Khadilkar A, Mandlik R. Epidemiology and treatment of osteoporosis in women: an Indian perspective. Int J Womens Health. 2015;7:841–50.
Mithal A, Kaur P. Osteoporosis in Asia: a call to action. Curr Osteoporos Rep. 2012;10:245–7.
Acharya S, Srivastava A, Sen I. Osteoporosis in Indian women aged 40–60 years. Arch Osteoporos. 2010;5:83–9.
Seeman E, Delmas PD. Bone quality the material and structural basis of bone strength and fragility. N Engl J Med. 2006;354:2250–61.
Kadam N, Chiplonkar S, Khadilkar A, Divate U, Khadilkar V. Low bone mass in urban Indian women above 40 years of age: prevalence and risk factors. Gynecol Endocrinol. 2010;26(12):909–17.
Mithal A, Bansal B, Kyer CS, Ebeling P. The Asia-Pacific regional audit-epidemiology, costs, and burden of osteoporosis in India 2013: a report of international osteoporosis foundation. Indian J Endocrinol Metab. 2014;18(4):449.
Desai MP, Khatkhatay MI, Bhanu Prakash KV, Savardekar LS, Shah RS, Ansari Z. Hormonal profiles and biochemical indices of bone turnover in Indian women. Osteoporos Int. 2007;18(7):923–9.
Eastell R, Hannon RA. Biomarkers of bone health and osteoporosis risk. Proc Nutr Soc. 2008;67:157–62.
Jagtap VR, Ganu JV, Nagane NS. BMD and serum intact osteocalcin in postmenopausal osteoporosis women. Ind J Clin Biochem. 2011;26(1):70–3.
Kalaiselvi VS, Prabhu K, Mani R, Vathsala V. The association of serum Osteocalcin with the bone mineral density in postmenopausal women. J Clin Diagn Res. 2013;7(5):814–6.
Sushir S, Dharmendra K, Atil L. Serum osteocalcin as a diagnostic biomarker for primary osteoporosis in women. J Clin Diagn Res. 2015;9(8):RC04–7.
Bhadricha H, Khatkhatay MI, Desai M. Development of an Indigenous ELISA for human intact Osteocalcin and its utility in diagnosis and management of osteoporosis. Clinica Chimica Acta. 2019;489:117–23.
Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;1(8476):307–10.
Passing H, Bablok W. A new procedure for testing the equality of measurements from two methods. J Clin Chem Clin Biochem. 1983;21:709–20.
Giavarina D. Understanding Bland-Altman analysis. Biochemia Medica. 2015;25(2):141–51.
Benu D, Animesh D, Amitabha C, Madhumita B, Jashodip B. Prevalence and risk factors of osteopenia and osteoporosis in Indian women. J Dent Med Sci. 2016;15(2):15–8.
Babu AS, Ikbal FM, Noone MS, Joseph AN, Samuel P. Osteoporosis and osteopenia in India: a few more observations. Indian J Med Sci. 2009;63:76.
Civitelli R, Armamento-Villareal R, Napoli N. Bone turnover markers: understanding their value in clinical trials and clinical practice. Osteoporos Int. 2009;20:843–51.
Agrawal S, Jain A, Mahajan D, Raghunandan C. Correlation of bone mineral density with biochemical markers in post- menopausal women. Indian J Clin Biochem. 2009;24(3):262–5.
Jaffrey B, et al. Measurement procedure, comparison and bias estimation using patient samples; approved guideline—Third Edition. Clinical and Laboratory Standards Institute (CLSI): CLSI document EP09-A3 Wayne; 2013. PA: 201.
Dundas CM, Demonte D, Park S. Streptavidin-biotin technology: improvements and innovations in chemical and biological applications. Appl Microbiol Biotechnol. 2013;97(21):9343–53.
Bossuyt PM, Reitsma JB, Bruns DE, Gatsonis CA, Glasziou PP, Irwig LM, et al. The STARD statement for reporting studies of diagnostic accuracy: explanation and elaboration. Clin Chem. 2003;49:7–18.
Acknowledgements
We thank Director, Dr. Smita Mahale for her support and encouragement during the entire course of the study and Clinical Collaborators Dr. Anushree Patil, Dr. Suchitra Surve, Dr. Tushar Bandgar from KEM Hospital, Dr. Renuka Munshi from BYL Nair Hospital for providing the valuable samples and Dr. S. Gaitonde from Global Hospital for the BMD measurements for validation of the Osteocalcin prototype.
Funding
This work was supported by the Department of Science and Technology (DST), Govt. of India (IDP/MED/20/2015) and Indian Council of Medical Research Grant (ITR/Ad hoc/2/2017-18)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Sonam Hatkar, Seema Kadam, M Ikram Khatkhatay and Meena Desai declare that they have no conflict of interest.
Human and Animal Rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional ethics committee, NIRRH ethics committee for clinical studies (292/2016) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hatkar, S.S., Kadam, S.S., Khatkhatay, M.I. et al. Screening and Assessment of Bone Health in Indian Women Using an Indigenous ELISA of Human Osteocalcin a Bone Turnover Marker. Ind J Clin Biochem 35, 436–441 (2020). https://doi.org/10.1007/s12291-019-00841-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12291-019-00841-0