Abstract
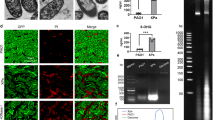
Streptococcus pneumoniae is a major respiratory pathogen that causes millions of deaths worldwide. Although subunit vaccines formulated with the capsular polysaccharides or their protein conjugates are currently-available, low-cost vaccines with wide serotype coverage still remain to be developed, especially for develo** countries. Recently, gamma- irradiation has been considered as an effective inactivation method to prepare S. pneumoniae vaccine candidate. In this study, we investigated the immunogenicity and protective immunity of gamma-irradiated S. pneumoniae (r-SP), by comparing with heat-inactivated S. pneumoniae (h-SP) and formalin-inactivated S. pneumoniae (f-SP), both of which were made by traditional inactivation methods. Intranasal immunization of C57BL/6 mice with r-SP in combination with cholera toxin as an adjuvant enhanced S. pneumoniaespecific antibodies on the airway mucosal surface and in sera more potently than that with h-SP or f-SP under the same conditions. In addition, sera from mice immunized with r-SP potently induced opsonophagocytic killing activity more effectively than those of h-SP or f-SP, implying that r-SP could induce protective antibodies. Above all, immunization with r-SP effectively protected mice against S. pneumoniae infection. Collectively, these results suggest that gamma- irradiation is an effective method for the development of a killed whole cell pneumococcal vaccine that elicits robust mucosal and systemic immune responses.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alderson, M.R. 2016. Status of research and development of pediatric vaccines for Streptococcus pneumoniae. Vaccine 34, 2959–2961.
Alloing, G., Martin, B., Granadel, C., and Claverys, J.P. 1998. Development of competence in Streptococcus pneumoniae: pheromone autoinduction and control of quorum sensing by the oligopeptide permease. Mol. Microbiol. 29, 75–83.
Babb, R., Chen, A., Hirst, T.R., Kara, E.E., McColl, S.R., Ogunniyi, A.D., Paton, J.C., and Alsharifi, M. 2016. Intranasal vaccination with ?-irradiated Streptococcus pneumoniae whole-cell vaccine provides serotype-independent protection mediated by B-cells and innate IL-17 responses. Clin. Sci. (Lond) 130, 697–710.
Chae, B.S. 2018. Pretreatment of low-dose and super-low-dose LPS on the production of in vitro LPS-induced inflammatory mediators. Toxicol. Res. 34, 65–73.
Chien, Y.W., Klugman, K.P., and Morens, D.M. 2010. Efficacy of whole-cell killed bacterial vaccines in preventing pneumonia and death during the 1918 influenza pandemic. J. Infect. Dis. 202, 1639–1648.
Clutterbuck, E.A., Lazarus, R., Yu, L.M., Bowman, J., Bateman, E.A.L., Diggle, L., Angus, B., Peto, T.E., Beverley, P.C., Mant, D., et al. 2012. Pneumococcal conjugate and plain polysaccharide vaccines have divergent effects on antigen-specific B cells. J. Infect. Dis. 205, 1408–1416.
Cunningham, A.F., Khan, M., Ball, J., Toellner, K.M., Serre, K., Mohr, E., and MacLennan, I.C. 2004. Responses to the soluble flagellar protein FliC are Th2, while those to FliC on Salmonella are Th1. Eur. J. Immunol. 34, 2986–2995.
Daniels, C.C., Rogers, P.D., and Shelton, C.M. 2016. A review of pneumococcal vaccines: current polysaccharide vaccine recommendations and future protein antigens. J. Pediatr. Pharmacol. Ther. 21, 27–35.
Datta, S.K., Okamoto, S., Hayashi, T., Shin, S.S., Mihajlov, I., Fermin, A., Guiney, D.G., Fierer, J., and Raz, E. 2006. Vaccination with irradiated Listeria induces protective T cell immunity. Immunity 25, 143–152.
Dillard, J.P., Vandersea, M.W., and Yother, J. 1995. Characterization of the cassette containing genes for type-3 capsular polysaccharide biosynthesis in Streptococcus pneumoniae. J. Exp. Med. 181, 973–983.
Fan, Y.C., Chiu, H.C., Chen, L.K., Chang, G.J., and Chiou, S.S. 2015. Formalin inactivation of Japanese encephalitis virus vaccine alters the antigenicity and immunogenicity of a neutralization epitope in envelope protein domain III. PLoS Negl. Trop. Dis. 9, e0004167.
Flynn, S., Toellner, K.M., Raykundalia, C., Goodall, M., and Lane, P. 1998. CD4 T cell cytokine differentiation: the B cell activation molecule, OX40 ligand, instructs CD4 T cells to express interleukin 4 and upregulates expression of the chemokine receptor, Blr-1. J. Exp. Med. 188, 297–304.
Flamaing, J., De Backer, W., Van Laethem, Y., Heijmans, S., and Mignon, A. 2015. Pneumococcal lower respiratory tract infections in adults: an observational case-control study in primary care in Belgium. BMC Fam. Pract. 16, 66.
Geno, K.A., Gilbert, G.L., Song, J.Y., Skovsted, I.C., Klugman, K.P., Jones, C., Konradsen, H.B., and Nahm, M.H. 2015. Pneumococcal capsules and their types: past, present, and future. Clin. Microbiol. Rev. 28, 871–899.
Henriques-Normark, B. and Tuomanen, E.I. 2013. The pneumococcus: epidemiology, microbiology, and pathogenesis. Cold Spring Harb. Perspect. Med. 3, a010215.
Hicks, L.A., Harrison, L.H., Flannery, B., Hadler, J.L., Schaffner, W., Craig, A.S., Jackson, D., Thomas, A., Beall, B., Lynfield, R., et al. 2007. Incidence of pneumococcal disease due to non-pneumococcal conjugate vaccine (PCV7) serotypes in the United States during the era of widespread PCV7 vaccination, 1998–2004. J. Infect. Dis. 196, 1346–1354.
Holmgren, J. and Czerkinsky, C. 2005. Mucosal immunity and vaccines. Nat. Med. 11, S45–S53.
Inzana, T.J. and Champion, A. 2007. Use of an inhibition enzymelinked immunosorbent assay for quantification of capsular polysaccharide or proteins in vaccines. Clin. Vaccine Immunol. 14, 323–327.
Kang, S.S., Yang, J.S., Kim, K.W., Yun, C.H, Holmgren, J., Czerkinsky, C., and Han, S.H. 2013. Anti-bacterial and anti-toxic immunity induced by a killed whole-cell-cholera toxin B subunit cholera vaccine is essential for protection against lethal bacterial infection in mouse pulmonary cholera model. Mucosal Immunol. 6, 826–837.
Keller, L.E., Robinson, D.A., and McDaniel, L.S. 2016. Nonencapsulated Streptococcus pneumoniae: emergence and pathogenesis. MBio 7, e01792.
Magi, M., Garcia, L., Vandenbranden, M., Palmantier, R., and Jacquet, A. 2004. Heat denaturation affects the ProDer p 1 IgE reactivity and downregulates the development of the specific allergic response. J. Allergy Clin. Immunol. 114, 545–552.
Metz, B., Kersten, G.F.A., Hoogerhout, P., Brugghe, H.F., Timmermans, H.A.M., de Jong, A., Meiring, H., ten Hove, J., Hennink, W.E., Crommelin, D.J.A., et al. 2004. Identification of formaldehyde-induced modifications in proteins: reactions with model peptides. J. Biol. Chem. 279, 6235–6243.
Mohammadzadeh, M., Pourakbari, B., Mahmoudi, S., Keshtkar, A., Habibi-Anbouhi, M., and Mamishi, S. 2018. Efficacy of whole-cell pneumococcal vaccine in mice: a systematic review and metaanalysis. Microb. Pathog. 122, 122–129.
Mook-Kanamori, B.B., Geldhoff, M., van der Poll, T., and van de Beek, D. 2011. Pathogenesis and pathophysiology of pneumococcal meningitis. Clin. Microbiol. Rev. 24, 557–591.
Moffitt, K.L. and Malley, R. 2011. Next generation pneumococcal vaccines. Curr. Opin. Immunol. 23, 407–413.
O’Brien, K.L., Wolfson, L.J., Watt, J.P., Henkle, E., Deloria-Knoll, M., McCall, N., Lee, E., Mulholland, K., Levine, O.S., Cherian, T., et al. 2009. Burden of disease caused by Streptococcus pneumoniae in children younger than 5 years: global estimates. Lancet 374, 893–902.
Odutola, A., Ota, M.O.C., Antonio, M., Ogundare, E.O., Saidu, Y., Foster-Nyarko, E., Owiafe, P.K., Ceesay, F., Worwui, A., Idoko, O.T., et al. 2017. Efficacy of a novel, protein-based pneumococcal vaccine against nasopharyngeal carriage of Streptococcus pneumoniae in infants: a phase 2, randomized, controlled, observer- blind study. Vaccine 35, 2531–2542.
Parameswarappa, S.G., Reppe, K., Geissner, A., Ménová, P., Govindan, S., Calow, A.D.J., Wahlbrink, A., Weishaupt, M.W., Monnanda, B.P., Bell, R.L., et al. 2016. A semi-synthetic oligosaccharide conjugate vaccine candidate confers protection against Streptococcus pneumoniae serotype 3 infection. Cell Chem. Biol. 23, 1407–1416.
Pollard, A.J., Perrett, K.P., and Beverley, P.C. 2009. Maintaining protection against invasive bacteria with protein-polysaccharide conjugate vaccines. Nat. Rev. Immunol. 9, 213–220.
Rodriguez, A., Tjarnlund, A., Ivanji, J., Singh, M., Garcia, I., Williams, A., Marsh, P.D., Troye-Blomberg, M., and Fernandez, C. 2005. Role of IgA in the defense against respiratory infections IgA deficient mice exhibited increased susceptibility to intranasal infection with Mycobacterium bovis BCG. Vaccine 23, 2565–2572.
Romero-Steiner, S., Frasch, C.E., Carlone, G., Fleck, R.A., Goldblatt, D., and Nahm, M.H. 2006. Use of opsonophagocytosis for serological evaluation of pneumococcal vaccines. Clin. Vaccine Immunol. 13, 165–169.
Rosch, J.W., Iverson, A.R., Humann, J., Mann, B., Gao, G., Vogel, P., Mina, M., Murrah, K.A., Perez, A.C., Edward Swords, W., et al. 2014. A live-attenuated pneumococcal vaccine elicits CD4+ Tcell dependent class switching and provides serotype independent protection against acute otitis media. EMBO Mol. Med. 6, 141–154.
Sanakkayala, N., Sokolovska, A., Gulani, J., Hogenesch, H., Sriranganathan, N., Boyle, S.M., Schurig, G.G., and Vemulapalli, R. 2005. Induction of antigen-specific Th1-type immune responses by gamma-irradiated recombinant Brucella abortus RB51. Clin. Diagn. Lab. Immunol. 12, 1429–1436.
Seo, H.S. 2015. Application of radiation technology in vaccines development. Clin. Exp. Vaccine Res. 4, 145–158.
Seo, H.S., **ong, Y.Q., Mitchell, J., Seepersaud, R., Bayer, A.S., and Sullam, P.M. 2010. Bacteriophage lysin mediates the binding of Streptococcus mitis to human platelets through interaction with fibrinogen. PLoS Pathog. 6, e1001047.
Tian, H., Weber, S., Thorkildson, P., Kozel, T.R., and Pirofski, L.A. 2009. Efficacy of opsonic and nonopsonic serotype 3 pneumococcal capsular polysaccharide-specific monoclonal antibodies against intranasal challenge with Streptococcus pneumoniae in mice. Infect. Immun. 77, 1502–1513.
Urban, J.F.Jr., Noben-Trauth, N., Donaldson, D.D., Madden, K.B., Morris, S.C., Collins, M., and Finkelman, F.D. 1998. IL-13, IL- 4Ra, and Stat6 are required for the expulsion of the gastrointestinal nematode parasite Nippostrongylus brasiliensis. Immunity 8, 255–264.
van der Poll, T. and Opal, S.M. 2009. Pathogenesis, treatment, and prevention of pneumococcal pneumonia. Lancet 374, 1543–1556.
Vissers, M., Wijmenga-Monsuur, A.J., Knol, M.J., Badoux, P., van Houten, M.A., van der Ende, A., Sanders, E.A.M., and Rots, N.Y. 2018. Increased carriage of non-vaccine serotypes with low invasive disease potential four years after switching to the 10-valent pneumococcal conjugate vaccine in the Netherlands. PLoS One 13, e0194823.
Xu, X., Meng, J., Wang, Y., Zheng, J., Wu, K., Zhang, X., Yin, Y., and Zhang, Q. 2014. Serotype-independent protection against pneumococcal infections elicited by intranasal immunization with ethanol-killed pneumococcal strain, SPY1. J. Microbiol. 52, 315–323.
Yang, J.S., Kim, H.J., Kang, S.S., Kim, K.W., Kim, D.W., Yun, C.H, Park, S.J, Seo, H.S., Finlay, B.B., and Han, S.H. 2015. TLR2, but not TLR4, plays a predominant role in the immune responses to cholera vaccines. J. Leukoc. Biol. 98, 661–669.
Zaman, M., Chandrudu, S., and Toth, I. 2013. Strategies for intranasal delivery of vaccines. Drug Deliv. Transl. Res. 3, 100–109.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jwa, M.Y., Jeong, S., Ko, E.B. et al. Gamma-irradiation of Streptococcus pneumoniae for the use as an immunogenic whole cell vaccine. J Microbiol. 56, 579–585 (2018). https://doi.org/10.1007/s12275-018-8347-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-018-8347-1