Abstract
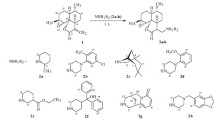
Artemongolins A–K (1–11), which are undescribed sesquiterpenoid dimers, were obtained from Artemisia mongolica and characterized through comprehensive spectral data, including HRESIMS, IR, 1D and 2D NMR, and ECD calculations. The absolute configurations of compounds 1, 4, and 7 were undoubtedly determined by a single-crystal X-ray crystallography. Artemongolins A–K (1–11) featured a rare 5/7/5/5/5/10 hexacyclic system composed of a germacrene and a guaianolide by a fused 2-oxaspiro[4,4]nonane-1-one ring system. Antihepatoma evaluation against three human hepatoma cell lines demonstrated that the most active compounds 5 and 6 displayed inhibitory activity with IC50 values of 88.6 and 57.0 (HepG2), 59.1 and 26.4 (Huh7), and 67.5 and 32.5 (SK-Hep-1) µM, respectively.






Similar content being viewed by others
References
Appendino G, Taglialatela-Scafati O, Romano A, Pollastro F, Avonto C, Rubiolo P (2009) Genepolide, a sesterpene γ-lactone with a novel carbon skeleton from Mountain wormwood (Artemisia umbelliformis). J Nat Prod 72(3):340–344. https://doi.org/10.1021/np800468m
Dai Q, Zhang C, Yuan Z, Sun Q, Jiang Y (2020) Current discovery strategies for hepatocellular carcinoma therapeutics. Expert Opin Drug Discov 15(2):243–258. https://doi.org/10.1080/17460441.2020.1696769
Dawkins J, Webster RM (2019) The hepatocellular carcinoma market. Nat Rev Drug Discov 18(1):13–14. https://doi.org/10.1038/nrd.2018.146
Dong W, Li TZ, Huang XY, He XF, Geng CA, Zhang XM, Chen JJ (2022) Artemzhongdianolides A1–A21, antihepatic fibrosis guaiane-type sesquiterpenoid dimers from Artemisia zhongdianensis. Bioorg Chem 128:106056. https://doi.org/10.1016/j.bioorg.2022.106056
Dong W, Ma WJ, Ma YB, Li FJ, Li TZ, Wang YC, He XF, Geng CA, Zhang XM, Chen JJ (2023a) Guaiane-type sesquiterpenoid dimers from Artemisia zhongdianensis and antihepatoma carcinoma activity via the p38MAPK pathway. Chin J Chem. https://doi.org/10.1002/cjoc.202300166
Dong W, Yang KX, He XF, Li TZ, Chen JJ (2023b) New eudesmanolides from Artemisia verlotorum and their potential targets of hepatocellular carcinoma by network pharmacology. Fitoterapia 167:105491. https://doi.org/10.1016/j.fitote.2023.105491
Dusmatova DE, Bobakulov KM, Mukhamatkhanova RF, Turgunov KK, Terenteva EO, Tsay EA, Sham’yanov ID, Tashkhodzhaev B, Azimova SS, Abdullaev ND (2021) Isolation of cytotoxic sesquiterpene lactones from the Tanacetopsis karataviensis (Kovalevsk.) Kovalevsk. Nat Prod Res 35(12):1939–1948. https://doi.org/10.1080/14786419.2019.1647423
Gao Z, Ma WJ, Li TZ, Ma YB, Hu J, Huang XY, Geng CA, He XF, Zhang XM, Chen JJ (2022) Artemidubolides A–T, cytotoxic unreported guaiane-type sesquiterpenoid dimers against three hepatoma cell lines from Artemisia dubia. Phytochemistry 202:113299. https://doi.org/10.1016/j.phytochem.2022.113299
Geng CA, Huang XY, Ma YB, Hou B, Li TZ, Zhang XM, Chen JJ (2017) (+/-)-Uncarilins A and B, dimeric isoechinulin-type alkaloids from Uncaria rhynchophylla. J Nat Prod 80(4):959–964. https://doi.org/10.1021/acs.jnatprod.6b00938
He XF, Li QH, Li TZ, Ma YB, Dong W, Yang KX, Geng CA, Zhang HW, Wang Y, Chen JJ (2023a) Artemeriopolides A–D, two types of sesquiterpenoid dimers with rare carbon skeletons from Artemisia eriopoda and their antihepatoma cytotoxicity. Org Chem Front 10:2635–2641. https://doi.org/10.1039/D3QO00223C
He XF, Ma WJ, Hu J, Li TZ, Geng CA, Ma YB, Wang MF, Yang KX, Zhang XM, Chen JJ (2023b) Diverse structures and antihepatoma effect of sesquiterpenoid dimers from Artemisia eriopoda by AKT/STAT signaling pathway. Signal Transduct Tar 8:64. https://doi.org/10.1038/s41392-022-01267-6
Hu JF, Zhu QX, Bai SP, Jia ZJ (1996) New eudesmane sesquiterpene and other constituents from Artemisia mongolica. Planta Med 62(5):477–478. https://doi.org/10.1055/s-2006-957946
Hu JF, He WY, Kong M, Jia ZJ, Feng XZ (2006) Mongolenin: a new triterpene from Artemisia mongolica. Nat Prod Lett 14(3):211–215. https://doi.org/10.1080/10575630008041233
Li S, Liu R, Pan Q, Wang G, Cheng D, Yang J, Chen H, Xu G (2020) De novo lipogenesis is elicited dramatically in human hepatocellular carcinoma especially in hepatitis C virus-induced hepatocellular carcinoma. Med Comm 1(2):178–187. https://doi.org/10.1002/mco2.15
Li TZ, Yang XT, Wang JP, Geng CA, Ma YB, Su LH, Zhang XM, Chen JJ (2021) Biomimetic synthesis of lavandiolides H, I, and K and artematrolide F via Diels–Alder reaction. Org Lett 23(21):8380–8384. https://doi.org/10.1021/acs.orglett.1c03120
Mata R, Delgado G, De Vivar AR (1985) Sesquiterpene lactones of Artemisia klotzchiana. Phytochemistry 24(7):1515–1519. https://doi.org/10.1016/S0031-9422(00)81057-2
Oikawa H, Tokiwano T (2004) Enzymatic catalysis of the Diels–Alder reaction in the biosynthesis of natural products. Nat Prod Rep 21(3):321–352. https://doi.org/10.1039/b305068h
Shang C, Huang XY, Wang Y, Dong W, He XF, Li TZ, Chen JJ (2023) Artemongolides A–F, undescribed sesquiterpenoid dimers from Artemisia mongolica and their antihepatic fibrosis activities. Org Biomol Chem 21:823–831. https://doi.org/10.1039/d2ob02182j
Shao ZG, Li LZ, Zheng YZ, Gong Q, Ke CQ, Yao S, Zhang HY, Tang CP, Ye Y (2022) Anti-inflammatory sesquiterpenoid dimers from Artemisia atrovirens. Fitoterapia 159:105199. https://doi.org/10.1016/j.fitote.2022.105199
Shen C, Huang XY, Geng CA, Li TZ, Wang JP, Tang S, Su LH, Gao Z, Zhang XM, Chen JJ (2020) Cytotoxic sesquiterpenoids against hepatic stellate cell line LX2 from Artemisia lavandulaefolia. Bioorg Chem 103:104107. https://doi.org/10.1016/j.bioorg.2020.104107
Su LH, Geng CA, Li TZ, Ma YB, Huang XY, Zhang XM, Chen JJ (2020) Artatrovirenols A and B: two cagelike sesquiterpenoids from Artemisia atrovirens. J Org Chem 85:13466–13471. https://doi.org/10.1021/acs.joc.0c01491
Su LH, Li TZ, Ma YB, Geng CA, Huang XY, Zhang X, Gao Z, Chen JJ (2021) Artematrovirenolides A–D and artematrolides S–Z, sesquiterpenoid dimers with cytotoxicity against three hepatoma cell lines from Artemisia atrovirens. Chin J Chem 40(1):104–114. https://doi.org/10.1002/cjoc.202100528
Su LH, Zhang XT, Ma YB, Geng CA, Huang XY, Hu J, Li TZ, Tang S, Shen C, Gao Z, Zhang XM, Chen JJ (2021) New guaiane-type sesquiterpenoid dimers from Artemisia atrovirens and their antihepatoma activity. Acta Pharm Sin B 11(6):1648–1666. https://doi.org/10.1016/j.apsb.2020.12.006
Su LH, Li TZ, Geng CA, Ma YB, Huang XY, Wang JP, Zhang XM, Chen JJ (2021) Trimeric and dimeric sesquiterpenoids from Artemisia atrovirens and their cytotoxicities. Org Chem Front 8(6):1249–1256. https://doi.org/10.1039/d0qo01615b
Su LH, Ma WJ, Ma YB, Li TZ, Geng CA, Dong W, He XF, Chen JJ (2023a) Artemiprinolides A–M, thirteen undescribed sesquiterpenoid dimers from Artemisia princeps and their antihepatoma activity. Phytochemistry 211:113714. https://doi.org/10.1016/j.phytochem.2023.113714
Su LH, Ma WJ, Ma YB, Li TZ, Geng CA, Dong W, He XF, Zhang XM, Chen JJ (2023b) Artemiprincepsolides A–F, novel germacrane-guaiane and eudesmane-guaiane sesquiterpenoid dimers from Artemisia princeps and their antihepatoma activity. Chin J Chem. https://doi.org/10.1002/cjoc.202300242
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249. https://doi.org/10.3322/caac.21660
Tang S, Ma YB, Geng CA, Shen C, Li TZ, Zhang XM, Su LH, Gao Z, Hu J, Chen JJ (2020a) Artemyrianins A–G from Artemisia myriantha and their cytotoxicity against HepG2 cells. Nat Prod Bioprospect 10:251–260. https://doi.org/10.1007/s13659-020-00255-z
Tang S, Zhang XT, Ma YB, Huang XY, Geng CA, Li TZ, Zhang XM, Shen C, Su LH, Gao Z, Chen JJ (2020b) Artemyrianolides A–S, cytotoxic sesquiterpenoids from Artemisia myriantha. J Nat Prod 83(9):2618–2630. https://doi.org/10.1021/acs.jnatprod.0c00396
Wang JP, Li TZ, Huang XY, Geng CA, Shen C, Sun JJ, Xue D, Chen JJ (2021) Synthesis and anti-fibrotic effects of santamarin derivatives as cytotoxic agents against hepatic stellate cell line LX2. Bioorg Med Chem Lett 41:127994. https://doi.org/10.1016/j.bmcl.2021.127994
Wang S, Sun J, Zeng KW, Chen X, Zhou W, Zhang C, ** H, Jiang Y, Tu PF (2014) Sesquiterpenes from Artemisia argyi: absolute configurations and biological activitie. Eur J Org Chem 2014(5):973–983. https://doi.org/10.1002/ejoc.201301445
Wang Q, Zhang T, Ke CQ, Tang C, Yao S, Lin L, Ye Y (2020) Sesquiterpene lactone dimers from Artemisia lavandulifolia inhibit interleukin-1β production in macrophages through activating autophagy. Bioorg Chem 105:104451. https://doi.org/10.1016/j.bioorg.2020.104451
Xue GM, Han C, Chen C, Li LN, Wang XB, Yang MH, Gu YC, Luo JG, Kong LY (2017) Artemisians A–D, diseco-guaianolide involved heterodimeric [4 + 2] adducts from Artemisia argyi. Org Lett 19(19):5410–5413. https://doi.org/10.1021/acs.orglett.7b02681
Xue GM, Zhu DR, Zhu TY, Wang XB, Luo JG, Kong LY (2019a) Lactone ring-opening seco-guaianolide involved heterodimers linked via an ester bond from Artemisia argyi with NO inhibitory activity. Fitoterapia 132:94–100. https://doi.org/10.1016/j.fitote.2018.12.004
Xue GM, Zhu DR, Han C, Wang XB, Luo JG, Kong LY (2019b) Artemisianins A–D, new stereoisomers of seco-guaianolide involved heterodimeric [4 + 2] adducts from Artemisia argyi induce apoptosis via enhancement of endoplasmic reticulum stress. Bioorg Chem 84:295–301. https://doi.org/10.1016/j.bioorg.2018.11.013
Yang JD, Roberts LR (2010) Hepatocellular carcinoma: a global view. Nat Rev Gastroenterol Hepatol 7(8):448–458. https://doi.org/10.1038/nrgastro.2010.100
Zhang XT, Hu J, Su LH, Geng CA, Chen JJ (2021) Artematrolide a inhibited cervical cancer cell proliferation via ROS/ERK/mTOR pathway and metabolic shift. Phytomedicine 91:153707. https://doi.org/10.1016/j.phymed.2021.153707
Zhou XD, Chai XY, Zeng KW, Zhao MB, Jiang Y, Tu PF (2015) Artesin A, a new cage-shaped dimeric guaianolide from Artemisia sieversiana. Tetrahedron Lett 56(9):1141–1143. https://doi.org/10.1016/j.tetlet.2015.01.081
Zhu Z, Turak A, Aisa HA (2022) Sesquiterpene lactones from Artemisia mongolica. Phytochemistry 199:113158. https://doi.org/10.1016/j.phytochem.2022.113158
Acknowledgements
This work was financially supported by the Key Program of the National Natural Science Foundation of China (22137008), the **ngdian Yingcai Project (YNWR-KJLJ-2019-002), the Youth Innovation Promotion Association, CAS (2020386), the Reserve Talents of Young and Middle-aged Academic and Technical Leaders in Yunnan Province (202105AC160021).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors do not have any conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shang, C., Ma, YB., Wang, Y. et al. Artemongolins A–K, undescribed germacrane-guaiane sesquiterpenoid dimers from Artemisia mongolica and their antihepatoma activities. Arch. Pharm. Res. 46, 782–794 (2023). https://doi.org/10.1007/s12272-023-01466-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-023-01466-x