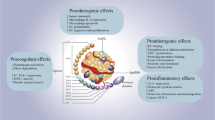
Abstract
LXRα agonists have attracted significant attention due to their potential biological activities on promoting cholesterol efflux. This study was designed to investigate whether setosphapyrone C and D have potential lipid-lowering capacity and the underlying mechanisms in vitro. Our data showed that setosphapyrone C and D had weak cytotoxicity compared to the liver X receptor α (LXRα) agonist T0901317. In RAW 264.7 macrophages, setosphapyrone C and D significantly enhanced [3H]-cholesterol efflux by ~ 21.3% and 32.4%, respectively; furthermore, setosphapyrone C and D enhanced the protein levels of ATP-binding cassette transporter (ABC) A1 and LXRα by 58% and 69%, and 60% and 70% (8 µM), respectively; however, they had no effect on the protein levels of ABCG1 and scavenger receptor B type 1; additionally, they had minor effect on the mRNA expression of lipogenic genes. Of note, setosphapyrone C and D significantly enhanced LXRα/ABCA1pathway in mice primary macrophages. In BRL cells, setosphapyrone C and D significantly improved the protein levels of ABCA1 and ABCG1; setosphapyrone D significantly enhanced the protein expression of low-density lipoprotein. Collectively, setosphapyrone C and D with weak cytotoxicity exhibited effective lipid-lowering effect via enhancing LXRα/ABC pathways. Setosphapyrones possess potential application for the treatment of hyperlipidemic diseases.




Similar content being viewed by others
References
Beltowski J (2008) Liver X receptors (LXR) as therapeutic targets in dyslipidemia. Cardiovasc Ther 26(4):297–316. doi:https://doi.org/10.1111/j.1755-5922.2008.00062.x
Breevoort SR, Angdisen J, Schulman IG (2014) Macrophage-independent regulation of reverse cholesterol transport by liver X receptors. Arterioscler Thromb Vasc Biol 34(8):1650–1660. doi:https://doi.org/10.1161/ATVBAHA.114.303383
Brewer HB Jr, Remaley AT, Neufeld EB, Basso F, Joyce C (2004) Regulation of plasma high-density lipoprotein levels by the ABCA1 transporter and the emerging role of high-density lipoprotein in the treatment of cardiovascular disease. Arterioscler Thromb Vasc Biol 24(10):1755–1760. doi:https://doi.org/10.1161/01.ATV.0000142804.27420.5b
Chiang JY (2009) Bile acids: regulation of synthesis. J Lipid Res 50(10):1955–1966. doi:https://doi.org/10.1194/jlr.R900010-JLR200
Chistiakov DA, Bobryshev YV, Orekhov AN (2016) Macrophage-mediated cholesterol handling in atherosclerosis. J Cell Mol Med 20(1):17–28. doi:https://doi.org/10.1111/jcmm.12689
Cui Y, Hou P, Li F, Liu Q, Qin S, Zhou G, Xu X, Si Y, Guo S (2017) Quercetin improves macrophage reverse cholesterol transport in apolipoprotein E-deficient mice fed a high-fat diet. Lipids Health Dis 16(1):9. doi:https://doi.org/10.1186/s12944-016-0393-2
Favari E, Chroni A, Tietge UJ, Zanotti I, Escolà-Gil JC, Bernini F (2015) Cholesterol efflux and reverse cholesterol transport. Handb Exp Pharmacol 224:181–206. https://doi.org/10.1007/978-3-319-09665-0_4
Florentin M, Liberopoulos EN, Mikhailidis DP, Elisaf MS (2008) Fibrate-associated adverse effects beyond muscle and liver toxicity. Curr Pharm Des 14(6):574–587. doi:https://doi.org/10.2174/138161208783885362
Guo S. Li L, Yin H (2018) Cholesterol homeostasis and liver X receptor (LXR) in atherosclerosis. Cardiovasc Hematol Disord Drug Targets 18(1):27–33. doi:https://doi.org/10.2174/1871529X18666180302113713
Henriksen T, Mahoney EM, Steinberg D (1981) Enhanced macrophage degradation of low density lipoprotein previously incubated with cultured endothelial cells: recognition by receptors for acetylated low density lipoproteins. Proc Natl Acad Sci U S A 78(10):6499–6503. doi:https://doi.org/10.1073/pnas.78.10.6499
Hou P, Hu S, Wang J, Yang Z, Yin J, Zhou G, Guo S (2019) Exogenous supplement of N-acetylneuraminic acid improves macrophage reverse cholesterol transport in apolipoprotein E-deficient mice. Lipids Health Dis 18(1):24. doi:https://doi.org/10.1186/s12944-019-0971-1
Hu S, Wang J, Li F, Hou P, Yin J, Yang Z, Yang X, Li T, **a B, Zhou G, Liu M, Song W, Guo S (2019) Structural characterisation and cholesterol efflux improving capacity of the novel polysaccharides from Cordyceps militaris. Int J Biol Macromol 131:264–272. doi:https://doi.org/10.1016/j.ijbiomac.2019.03.078
Li T, Hu SM, Pang XY, Wang JF, Yin JY, Li FH, Wang J, Yang XQ, **a B, Liu YH, Song WG, Guo SD (2020) J Cell Mol Med. doi:https://doi.org/10.1111/JCMM.15012
Linton MF, Tao H, Linton EF, Yancey PG (2017) SR-B1: a multifunctional receptor in cholesterol homeostasis and atherosclerosis. Trends Endocrinol Metab 28(6):461–472. doi:https://doi.org/10.1016/j.tem.2017.02.001
Ma AZ, Song ZY, Zhang Q (2014) Cholesterol efflux is LXRα isoform-dependent in human macrophages. BMC Cardiovas Disord 14:80. doi:https://doi.org/10.1186/1471-2261-14-80
Maguire EM, Pearce SWA, **ao Q (2019) Foam cell formation: a new target for fighting atherosclerosis and cardiovascular disease. Vascul Pharmacol 112:54–71. doi:https://doi.org/10.1016/j.vph.2018.08.002
Natuk RJ, Holowczak JA (1985) Vaccinia virus proteins on the plasma membrane of infected cells. III. Infection of peritoneal macrophages. Virology 147(2):354–372. doi:https://doi.org/10.1016/0042-6822(85)90138-2
Ouimet M, Barrett TJ, Fisher EA (2019) HDL and reverse cholesterol transport. Circ Res 124(10):1505–1518. doi:https://doi.org/10.1161/CIRCRESAHA.119.312617
Pang X, Lin X, Yang J, Zhou X, Yang B, Wang J, Liu Y (2018) Spiro-phthalides and isocoumarins isolated from the marine-sponge-derived fungus Setosphaeria sp. SCSIO41009. J Nat Prod 81(8):1860–1868. doi:https://doi.org/10.1021/acs.jnatprod.8b00345
Park JG, Oh GT (2019) Current pharmacotherapies for atherosclerotic cardiovascular disease. Arch Pharm Res 42(3):206–223. doi:https://doi.org/10.1007/s12272-019-01116-1
Rasheed A, Cummins CL (2018) Beyond the foam cell: the role of LXRs in preventing atherogenesis. Int J Mol Sci 19(8):E2307. https://doi.org/10.3390/ijms19082307
Ren K, Li H, Zhou HF, Liang Y, Tong M, Chen L, Zheng XL, Zhao GJ (2019) Mangiferin promotes macrophage cholesterol efflux and protects against atherosclerosis by augmenting the expression of ABCA1 and ABCG1. Aging 11(23):10992–11009. doi:https://doi.org/10.18632/aging.102498
Tian H, Liu Q, Qin S, Zong C, Zhang Y, Yao S, Yang N, Guan T, Guo S (2017) Synthesis and cardiovascular protective effects of quercetin 7-O-sialic acid. J Cell Mol Med 21(1):107–120. doi:https://doi.org/10.1111/jcmm.12943
Ulven SM, Dalen KT, Gustafsson JA, Nebb HI (2005) LXR is crucial in lipid metabolism. Prostaglandins Leukot Essent Fatty Acids 73(1):59–63. doi:https://doi.org/10.1016/j.plefa.2005.04.009
Varin A, Thomas C, Ishibashi M, Ménégaut L, Gautier T, Trousson A, Bergas V, de Barros JP, Narce M, Lobaccaro JM, Lagrost L, Masson D (2015) Liver X receptor activation promotes polyunsaturated fatty acid synthesis in macrophages: relevance in the context of atherosclerosis. Arteroscler Thromb Vasc Biol 35(6):1357–1365. doi:https://doi.org/10.1161/ATVBAHA.115.305539
Wang B, Tontonoz P (2018) Liver X receptors in lipid signaling and membrane homeostasis. Nat Rev Endocrinol 14(8):452–463. doi:https://doi.org/10.1038/s41574-018-0037-x
Ward NC, Watts GF, Eckel RH (2019) Statin Toxicity. Circ Res 124(2):328–350. doi:https://doi.org/10.1161/CIRCRESAHA.118.312782
Xu C, Li CY, Kong AN (2005) Induction of phase I, II and III drug metabolism/transport by xenobiotics. Arch Pharm Res 28(3):249–268. doi:https://doi.org/10.1007/bf02977789
Yang Z, Liu G, Wang Y, Yin J, Wang J, **a B, Li T, Yang X, Hou P, Hu S, Song W, Guo S (2019) Fucoidan A2 from the brown seaweed Ascophyllum nodosum lowers lipid by improving reverse cholesterol transport in C57BL/6J mice fed a high-fat diet. J Agric Food Chem 67(20):5782–5791. doi:https://doi.org/10.1021/acs.jafc.9b01321
Yin J, Yang X, **a B, Yang Z, Wang Z, Wang J, Li T, Lin P, Song X, Guo S (2019) The fucoidan from sea cucumber Apostichopus japonicus attenuates lipopolysaccharide-challenged liver injury in C57BL/6J mice. J Funct Foods 61:103493. doi:https://doi.org/10.1016/j.jff.2019.103493
Acknowledgements
This work was supported by Natural Science Foundation of China (81770463, 31300639), Shandong Provincial Natural Science Fund (ZR2013HQ014), Medicine & Health Scientific Technology Development Program of Shandong Province (2018WS064), and Study Abroad Fund of Weifang Medical University. We thank Guanghai Zhou at Shandong First Medical University & Shandong Academy of Medical Sciences for providing help in performing the isotope tracing assay.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declared there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, T., Yin, J., Ji, Y. et al. Setosphapyrone C and D accelerate macrophages cholesterol efflux by promoting LXRα/ABCA1 pathway. Arch. Pharm. Res. 43, 788–797 (2020). https://doi.org/10.1007/s12272-020-01255-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-020-01255-w