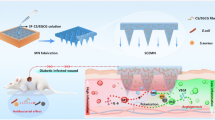
Abstract
Collagen is the most prevalent scaffold material for in vitro skin models. The major limitation of collagen scaffold is its mechanical weakness, resulting in severe contraction during differentiation. Here, we presented a slime-webbed scaffold composed of perpendicularly stacked fibers with large pores. This slime-webbed scaffold did not contract while improving molecular transport and achieving comparable cell viability. Fibroblasts were seeded into the slime-webbed scaffold to mimic the dermal layer. In the epidermal layer, which was on top of this scaffold, keratinocytes expressed the differentiation biomarkers, keratin-5 and involucrin. Our slime-webbed scaffold-based human skin models overcome the critical limitations of collagen scaffold, suggesting a promising alternative skin model for consistent testing of drugs or cosmetic products.
Similar content being viewed by others
References
Matsusaki, M., K. Fujimoto, Y. Shirakata, S. Hirakawa, K. Hashimoto, and M. Akashi (2015) Development of full-thickness human skin equivalents with blood and lymph-like capillary networks by cell coating technology. J. Biomed. Mater. Res. A 103: 3386–3396.
Serpell, J. (1996) In the Company of Animals: A Study of Human-Animal Relationships. Cambridge University Press, Cambridge, UK.
Watson, M. E., Jr., M. N. Neely, and M. G. Caparon (2016) Animal models of Streptococcus pyogenes infection. pp. 497–521. In: J. J. Ferretti, D. L. Stevens, and V. A. Fischetti (eds.). Streptococcus pyogenes: Basic Biology to Clinical Manifestations. University of Oklahoma Health Sciences Center, Oklahoma City, OK, USA.
Schmook, F. P., J. G. Meingassner, and A. Billich (2001) Comparison of human skin or epidermis models with human and animal skin in in-vitro percutaneous absorption. Int. J. Pharm. 215: 51–56.
Seok, J., H. S. Warren, A. G. Cuenca, M. N. Mindrinos, H. V. Baker, W. Xu, D. R. Richards, G. P. McDonald-Smith, H. Gao, L. Hennessy, C. C. Finnerty, C. M. López, S. Honari, E. E. Moore, J. P. Minei, J. Cuschieri, P. E. Bankey, J. L. Johnson, J. Sperry, A. B. Nathens, T. R. Billiar, M. A. West, M. G. Jeschke, M. B. Klein, R. L. Gamelli, N. S. Gibran, B. H. Brownstein, C. Miller-Graziano, S. E. Calvano, P. H. Mason, J. P. Cobb, L. G. Rahme, S. F. Lowry, R. V. Maier, L. L. Moldawer, D. N. Herndon, R. W. Davis, W. **ao, R. G. Tompkins, and Inflammation and Host Response to Injury, Large Scale Collaborative Research Program (2013) Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc. Natl. Acad. Sci. U. S. A. 110: 3507–3512.
Warren, H. S., C. Fitting, E. Hoff, M. Adib-Conquy, L. Beasley-Topliffe, B. Tesini, X. Liang, C. Valentine, J. Hellman, D. Hayden, and J. M. Cavaillon (2010) Resilience to bacterial infection: difference between species could be due to proteins in serum. J. Infect. Dis. 201: 223–232.
Olson, H., G. Betton, J. Stritar, and D. Robinson (1998) The predictivity of the toxicity of pharmaceuticals in humans from animal data—an interim assessment. Toxicol. Lett. 102–103: 535–538.
Kola, I. and J. Landis (2004) Can the pharmaceutical industry reduce attrition rates? Nat. Rev. Drug Discov. 3: 711–716.
Bell, E., H. P. Ehrlich, D. J. Buttle, and T. Nakatsuji (1981) Living tissue formed in vitro and accepted as skin-equivalent tissue of full thickness. Science 211: 1052–1054.
Adler, S., D. Basketter, S. Creton, O. Pelkonen, J. van Benthem, V. Zuang, K. E. Andersen, A. Angers-Loustau, A. Aptula, A. Bal-Price, E. Benfenati, U. Bernauer, J. Bessems, F. Y. Bois, A. Boobis, E. Brandon, S. Bremer, T. Broschard, S. Casati, S. Coecke, R. Corvi, M. Cronin, G. Daston, W. Dekant, S. Felter, E. Grignard, U. Gundert-Remy, T. Heinonen, I. Kimber, J. Kleinjans, H. Komulainen, R. Kreiling, J. Kreysa, S. B. Leite, G. Loizou, G. Maxwell, P. Mazzatorta, S. Munn, S. Pfuhler, P. Phrakonkham, A. Piersma, A. Poth, P. Prieto, G. Repetto, V. Rogiers, G. Schoeters, M. Schwarz, R. Serafimova, H. Tähti, E. Testai, J. Delft, H. Loveren, M. Vinken, A. Worth, and J. M. Zaldivar (2011) Alternative (non-animal) methods for cosmetics testing: current status and future prospects-2010. Arch. Toxicol. 85: 367–485.
Vinci, M., S. Gowan, F. Boxall, L. Patterson, M. Zimmermann, W. Court, C. Lomas, M. Mendiola, D. Hardisson, and S. A. Eccles (2012) Advances in establishment and analysis of three-dimensional tumor spheroid-based functional assays for target validation and drug evaluation. BMC Biol. 10: 29.
Ravi, M., V. Paramesh, S. R. Kaviya, E. Anuradha, and F. D. Solomon (2015) 3D cell culture systems: advantages and applications. J. Cell. Physiol. 230: 16–26.
Rhee, S. (2009) Fibroblasts in three dimensional matrices: cell migration and matrix remodeling. Exp. Mol. Med. 41: 858–865.
Chau, D. Y., C. Johnson, S. MacNeil, J. W. Haycock, and A. M. Ghaemmaghami (2013) The development of a 3D immunocompetent model of human skin. Biofabrication 5: 035011.
Wenger, M. P., L. Bozec, M. A. Horton, and P. Mesquida (2007) Mechanical properties of collagen fibrils. Biophys. J. 93: 1255–1263.
Sutterby, E., P. Thurgood, S. Baratchi, K. Khoshmanesh, and E. Pirogova (2020) Microfluidic skin-on-a-chip models: toward biomimetic artificial skin. Small 16: e2002515.
Grinnell, F. (2000) Fibroblast-collagen-matrix contraction: growth-factor signalling and mechanical loading. Trends Cell Biol. 10: 362–365.
Han, Y. L., P. Ronceray, G. Xu, A. Malandrino, R. D. Kamm, M. Lenz, C. P. Broedersz, and M. Guo (2018) Cell contraction induces long-ranged stress stiffening in the extracellular matrix. Proc. Natl. Acad. Sci. U. S. A. 115: 4075–4080.
Ramtani, S. (2004) Mechanical modelling of cell/ECM and cell/cell interactions during the contraction of a fibroblast-populated collagen microsphere: theory and model simulation. J. Biomech. 37: 1709–1718.
Grinnell, F., C. H. Ho, Y. C. Lin, and G. Skuta (1999) Differences in the regulation of fibroblast contraction of floating versus stressed collagen matrices. J. Biol. Chem. 274: 918–923.
Ackermann, K., S. L. Borgia, H. C. Korting, K. R. Mewes, and M. Schäfer-Korting (2010) The Phenion full-thickness skin model for percutaneous absorption testing. Skin Pharmacol. Physiol. 23: 105–112.
Charulatha, V. and A. Rajaram (2003) Influence of different crosslinking treatments on the physical properties of collagen membranes. Biomaterials 24: 759–767.
Jeong, S., J. Kim, H. M. Jeon, K. Kim, and G. Y. Sung (2021) Development of an aged full-thickness skin model using flexible skin-on-a-chip subjected to mechanical stimulus reflecting the circadian rhythm. Int. J. Mol. Sci. 22: 12788.
Zhao, X., Q. Lang, L. Yildirimer, Z. Y. Lin, W. Cui, N. Annabi, K. W. Ng, M. R. Dokmeci, A. M. Ghaemmaghami, and A. Khademhosseini (2016) Photocrosslinkable gelatin hydrogel for epidermal tissue engineering. Adv. Healthc. Mater. 5: 108–118.
Kwak, B. S., W. Choi, J. W. Jeon, J. I. Won, G. Y. Sung, B. Kim, and J. H. Sung (2018) In vitro 3D skin model using gelatin methacrylate hydrogel. J. Ind. Eng. Chem. 66: 254–261.
Sriram, G., M. Alberti, Y. Dancik, B. Wu, R. Wu, Z. Feng, S. Ramasamy, P. L. Bigliardi, M. Bigliardi-Qi, and Z. Wang (2018) Full-thickness human skin-on-chip with enhanced epidermal morphogenesis and barrier function. Mater. Today (Kidlington) 21: 326–340.
Jusoh, N., J. Ko, and N. L. Jeon (2019) Microfluidics-based skin irritation test using in vitro 3D angiogenesis platform. APL Bioeng. 3: 036101.
Rhee, J. K. (2019) Fiber manufacturing apparatus. US Patent 11,091,853.
Avendano, A., J. J. Chang, M. G. Cortes-Medina, A. J. Seibel, B. R. Admasu, C. M. Boutelle, A. R. Bushman, A. A. Garg, C. M. DeShetler, S. L. Cole, and J. W. Song (2020) Integrated biophysical characterization of fibrillar collagen-based hydrogels. ACS Biomater. Sci. Eng. 6: 1408–1417.
Lee, S., S. P. **, Y. K. Kim, G. Y. Sung, J. H. Chung, and J. H. Sung (2017) Construction of 3D multicellular microfluidic chip for an in vitro skin model. Biomed. Microdevices 19: 22.
Lee, W., V. Lee, S. Polio, P. Keegan, J. H. Lee, K. Fischer, J. K. Park, and S. S. Yoo (2010) On-demand three-dimensional freeform fabrication of multi-layered hydrogel scaffold with fluidic channels. Biotechnol. Bioeng. 105: 1178–1186.
Song, H. J., H. Y. Lim, W. Chun, K. C. Choi, J. H. Sung, and G. Y. Sung (2017) Fabrication of a pumpless, microfluidic skin chip from different collagen sources. J. Ind. Eng. Chem. 56: 375–381.
Carlson, M. W., A. Alt-Holland, C. Egles, and J. A. Garlick (2008) Three-dimensional tissue models of normal and diseased skin. Curr. Protoc. Cell Biol. Chapter 19: Unit 19.9.
D’Orazio, J., S. Jarrett, A. Amaro-Ortiz, and T. Scott (2013) UV radiation and the skin. Int. J. Mol. Sci. 14: 12222–12248.
Reijnders, C. M., A. van Lier, S. Roffel, D. Kramer, R. J. Scheper, and S. Gibbs (2015) Development of a full-thickness human skin equivalent in vitro model derived from TERT-immortalized keratinocytes and fibroblasts. Tissue Eng. Part A 21: 2448–2459.
Acknowledgements
This work was supported in part by the Technology development Program of MSS (S3251721, S3316767) by Korea Technology and Information Promotion Agency, Basic Research Lab (2022R1A4A2000748) and Bio & Medical Technology Development Program (2022M3A9B6018217) by National Research Foundation of Korea, Technology Innovation Program (20008414) and the Alchemist Project of the Korea Evaluation Institute of Industrial Technology (KEIT 20018560, NTIS 1415180625) by the Ministry of Trade, Industry & Energy (MOTIE), Hongik University Research Fund. It was also supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) by the Ministry of Education, Science and Technology (NRF-2021R1F1A1056188), the High Value-added Food Technology Development Program, the Ministry of Agriculture, Food and Rural Affairs (MAFRA) (321021031HD030), RP-Grant 2020 and Research Grant 2020–2021 of Ewha Womans University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no conflict of interest.
Neither ethical approval nor informed consent was required for this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material (ESM)
Rights and permissions
About this article
Cite this article
Kim, J.J., Lee, N.K., Ryu, D.E. et al. Highly Porous and Rigid, Full-thickness Human Skin Model from the Slime-webbed Fiber Scaffold. Biotechnol Bioproc E 28, 246–254 (2023). https://doi.org/10.1007/s12257-022-0341-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-022-0341-0