Abstract
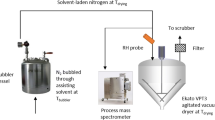
In this study, a drying method using ethanol pretreatment was developed that can effectively remove residual solvent from crystalline vancomycin. Microwave-assisted drying with ethanol pretreatment was sufficient to remove residual acetone and ethanol concentrations below the ICH limits (5,000 ppm for acetone and 5,000 ppm for ethanol). At all the microwave power (100, 200, 300 W), a large amount of the residual acetone was initially removed during the drying, and the drying efficiency increased when increasing the microwave power. In addition, the drying kinetics and thermodynamics were examined. The experimental data were fitted to the Newton, Page, Modified Page, Henderson and Pabis, and Geometric models. Comparison of results revealed that the Page model was the most appropriate. The activation energy of microwave-assisted drying was found to be 10.8792 kJ/mol. The Standard Gibbs free energy change was determined to be negative, while the standard enthalpy change and entropy change were positive. These results revealed the endothermic, irreversible and spontaneous nature of drying.
Similar content being viewed by others
References
Yan, H., D. Qi, X. Cheng, Z. Song, W. Li, and B. He (1998) Antibiotic activities and affinities for bacterial cell wall analogue of N-demethylvancomycin and its derivatives. J. Antibiot. 51: 750–756.
Griffith, R. S. (1984) Vancomycin use-an historical review. J. Antimicrob. Chemother. 14: 1–5.
Kim, S. J. and J. H. Kim (2015) Investigation on the role of ion exchange resin in the crystallization process for the purification of vancomycin. Korean J. Chem. Eng. 32: 465–470.
Nambiar, S., R. D. Madurawe, S. M. Zuk, S. R. Khan, C. D. Ellison, P. J. Faustino, D. J. Mans, M. L. Trehy, M. E. Hadwiger, M. T. Boyne, K. Biswas, and E. M. Cox (2012) Product quality of parenteral vancomycin products in the United States. Antimicrob. Agents Chemother. 56: 2819–2823.
Lee, J. Y., K. H. Lee, H. J. Chae, and J. H. Kim (2010) Systematic evaluation and optimization of crystallization conditions for vancomycin purification. Korean J. Chem. Eng. 27: 1538–1546.
Javadzadeh, Y., A. Mohammadi, N. S. Khoei, and A. Nokhodchi (2009) Improvement of physicomechanical properties of carbamazepine by recrystallization at different pH values. Acta Pharm. 59: 188–197.
Kim, S. I., K. E. Jeong, S. Y. Jeong, and C. U. Kim (2008) A study on separation of naphthalene from naphthalene and 2-methylnaphthalene mixture by melt and solution crystallization. J. Korean Oil Chemists’ Soc. 25: 232–239.
Kim, Y. N., J. Y. Lee, and J. H. Kim (2011) Improvement of a crystallization process for the purification of vancomycin. Process Biochem. 46: 2068–2073.
Kwak, E. A., S. J. Kim, and J. H. Kim (2012) Effect of ion exchange resin on increased surface area crystallization process for purification of vancomycin. Korean J. Chem. Eng. 29: 1487–1492.
Kim, S. J. and J. H. Kim (2014) The reduction of crystal formation time of vancomycin using silica gel. Korean J. Microbiol. Biotechnol. 42: 232–237.
Kim, S. J. and J. H. Kim (2014) Effect of ionic liquid on increased surface area crystallization process for vancomycin. Korean J. Microbiol. Biotechnol. 42: 297–301.
Ha, G. S. and J. H. Kim (2015) Effect of an ionic liquid on vancomycin crystallization. Korean J. Chem. Eng. 32: 576–582.
Ha, G. S. and J. H. Kim (2017) Kinetic and thermodynamic characteristics of crystallization of vancomycin. Korean J. Chem. Eng. 34: 2451–2458.
Food and Drug Administration (1997) International Conference on Harmonisation; Guidance on impurities: residual solvents. Fed. Regist. 62: 67377–67388.
Lee, J. W., Y. T. Jung, J. W. Suh, and K. S. Lee (2005) Process of purifying vancomycin hydrochloride. US Patent 7,018,814 B2.
Teo, C., X. Sun, and X. Y. Wang (2016) Method and system for producing high purity vancomycin hydrochloride. US Patent 9,428,291B2.
Lee, C. G. and J. H. Kim (2015) Improved drying method for removal of residual solvents from paclitaxel by pre-treatment with ethanol and water. Process Biochem. 50: 1031–1036.
Lee, J. Y. and J. H. Kim (2013) Removal of residual methylene chloride from homoharringtonine by pre-treatment with ethanol. Process Biochem. 48: 1809–1813.
Kang, H. J. and J. H. Kim (2020) Removal of residual toluene and methyl tertiary butyl ether from amorphous paclitaxel by simple rotary evaporation with alcohol pretreatment. Biotechnol. Bioprocess Eng. 25: 86–93.
Kang, H. J. and J. H. Kim (2019) Removal of residual chloroform from amorphous paclitaxel pretreated by alcohol. Korean J. Chem. Eng. 36: 1965–1970.
Jang, W. S. and J. H. Kim (2019) Characteristics and mechanism of microwave-assisted drying of amorphous paclitaxel for removal of residual solvent. Biotechnol. Bioprocess Eng. 24: 529–535.
Kang, S. S. and J. H. Kim (2020) Kinetics, mechanism, and thermodynamics studies of vacuum drying of biomass from Taxus chinensis cell cultures. Biotechnol. Bioprocess Eng. 25: 336–343.
Kim, H. S. and J. H. Kim (2017) Kinetics and thermodynamics of microwave-assisted drying of paclitaxel for removal of residual methylene chloride. Process Biochem. 56: 163–170.
Cho, D. N. and J. H. Kim (2020) Isotherm, kinetic and thermodynamic characteristics for adsorption of acenaphthene onto sylopute. Korean Chem. Eng. Res. 58: 127–134.
Moosavi, H., M. Forghani, and E. Managhebi (2013) Effect of different air-drying time on the microleakage of single-step self-etch adhesives. Restor. Dent. Endod. 38: 73–78.
Lee, J. Y. and J. H. Kim (2011) Effect of water content of organic solvent on microwave-assisted extraction efficiency of paclitaxel from plant cell culture. Korean J. Chem. Eng. 28: 1561–1565.
Gupta, S., S. Rafiq, M. Kundu, and P. Sen (2012) Origin of strong synergism in weakly perturbed binary solvent system: A case study of primary alcohols and chlorinated methanes. J. Phys. Chem. B. 116: 1345–1355.
Kamath, G., G. Georgiev, and J. J. Potoff (2005) Molecular modeling of phase behavior and microstructure of acetone-chloroform-methanol binary mixtures. J. Phys. Chem. B. 109: 19463–19473.
Nam, H. W. and J. H. Kim (2020) Characteristics of microwave-assisted drying of plant cells of Taxus chinensis for moisture removal. Appl. Chem. Eng. 31: 208–214.
Lee, J. Y. and J. H. Kim (2013) Microwave-assisted drying of paclitaxel for removal of residual solvents. Process Biochem. 48: 545–550.
Park, S. H. and J. H. Kim (2020) Equilibrium, isotherm, kinetic and thermodynamic studies for adsorption of 7-Epi-10-deacetylpaclitaxel from Taxus chinensis on Sylopute. Korean Chem. Eng. Res. 58: 113–121.
Acknowledgements
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Korean Ministry of Education, Science and Technology (Grant Number: 2018R1D1A3A 03000683).
The authors declare no conflict of interest.
Neither ethical approval nor informed consent was required for this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yoon, TH., Kim, JH. Development of Drying Process for Removal of Residual Solvent from Crystalline Vancomycin and Kinetic and Thermodynamic Analysis Thereof. Biotechnol Bioproc E 25, 777–786 (2020). https://doi.org/10.1007/s12257-020-0290-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-020-0290-4