Abstract
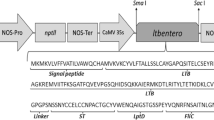
Porphyromonas gingivalis induces destruction of periodontal tissues that surround and support the teeth, contributing to the development of periodontitis, which eventually results in tooth loss in adults. A 53-kDa protein of P. gingivalis is a major subunit variant protein of minor fimbriae (Mfa1), and is reported to be highly immunogenic and considered as a potential vaccine candidate. The gene encoding 53-kDa protein, was divided into three segments, and each DNA segment was fused to the gene coding for cholera toxin B subunit (CTB) to improve gut mucosal immune responses. The ctb-mfal fusion genes were expressed in the leaf tissues of Nicotiana benthamiana using agroinfiltration. Immunoblot analysis revealed that CTB-Mfa1 fusion proteins were produced in the agroinfiltrated leaves. The interaction of the plant-produced CTB-Mfa1 fusion proteins with GM1-ganglioside, which acts as the binding site for native CTB, was confirmed by GM1-ELISA. Mice immunized orally with the agroinfiltrated leaf powder containing the CTB-Mfa1 fusion proteins elicited serum IgG and fecal IgA antibodies to CTB and Mfa1. These results suggest that CTB-Mfa1 fusion proteins produced in plants can be used as an oral vaccine to control P. gingivalis infection-associated periodontitis.
Similar content being viewed by others
References
Loesche, W. J. and N. S. Grossman (2001) Periodontal disease as a specific, albeit chronic, infection: diagnosis and treatment. Clin. Microbiol. Rev. 14: 727–752.
Kim, J. and S. Amar (2006) Periodontal disease and systemic conditions: a bidirectional relationship. Odontology. 94: 10–21.
Lamont, R. J. and H. F. Jenkinson (1998) Life below the gum line: pathogenic mechanisms of Porphyromonas gingivalis. Microbiol. Mol. Biol. Rev. 62: 1244–1263.
Lamont, R. J. and H. F. Jenkinson (2000) Subgingival colonization by Porphyromonas gingivalis. Oral Microbiol. Immunol. 15: 341–349.
Holt, S. C., L. Kesavalu, S. Walker, and C. A. Genco (1999) Virulence factors of Porphyromonas gingivalis. Periodontol. 2000. 20: 168–238.
O-Brien-Simpson, N. M., P. D. Veith, S. G. Dashper, and E. C. Reynolds (2003) Porphyromonas gingivalis gingipains: the molecular teeth of a microbial vampire. Curr. Protein. Pept. Sci. 4: 409–426.
Enersen, M., K. Nakano, and A. Amano (2013) Porphyromonas gingivalis fimbriae. J. Oral Microbiol. 5: 20265.
Kokeguchi, S., K. Kato, H. Kurihara, F. Nishimura, and Y. Murayama (1990) Purification and characterization of two major outer membrane proteins from Porphyromonas gingivalis. Dent. Jpn. (Tokyo) 27: 29–34.
Kurihara, H., F. Nishimura, T. Nakamura, M. Nakagawa, I. Tanimoto, Y. Nomura, S. Kokeguchi, K. Kato, and Y. Murayama (1991) Humoral immune response to an antigen from Porphyromonas gingivalis 381 in periodontal disease. Infect. Immun. 59: 2758–2762.
Guo, S., K. Takahashi, S. Kokeguchi, S. Takashiba, D. F. Kinane, and Y. Murayama (2000) Antibody responses against Porphyromonas gingivalis infection in patients with early-onset periodontitis. J. Clin. Periodontol. 27: 769–777.
Arai, M., N. Hamada, and T. Umemoto (2000) Purification and characterization of a novel secondary fimbrial protein from Porphyromonas gingivalis strain 381. FEMS Microbiol. Lett. 193: 75–81.
Nagano, K., Y. Hasegawa, Y. Yoshida, and F. Yoshimura (2015) A major fimbrilin variant of Mfa1 fimbriae in Porphyromonas gingivalis. J. Dent. Res. 94: 1143–1148.
Takaiwa, F., Y. Wakasa, H. Tagagi, and T. Hioi (2015) Rice seed for delivery of vaccines to gut mucosal immune tissues. Plant Biotechnol. J. 13: 1041–1055.
Sack, M., T. Rademacher, H. Spiegel, A. Boes, S. Hellwig, J. Drossard, E. Stoger, and R. Fischer (2015) From gene to harvest: insights into upstream process development for the GMP production of a monoclonal antibody in transgenic tobacco plants. Plant Biotechnol. J. 13: 1094–1105.
Karg, S. R. and P. T. Kallio (2009) The production of biopharmaceuticals in plant systems. Biotechnol. Adv. 27: 879–894.
Chebolu, S. and H. Daniell (2009) Chloroplast-derived vaccine antigens and biopharmaceuticals: expression, folding, assembly and functionality. Curr. Top Microbiol. Immunol. 332: 33–54.
Marillonnet, S., C. Thoeringer, R. Kandzia, V. Klimyuk, and Y. Gleba (2005) Systemic Agrobacterium tumefaciens-mediated transfection of viral replicons for efficient transient expression in plants. Nat. Biotechnol. 23: 718–723.
Huy, N. X., N. Q. D. Tien, M. Y. Kim, T. G. Kim, Y. S. Jang, and M. S. Yang (2016) Immunogenicity of an S1D epitope from porcine epidemic diarrhea virus and cholera toxin B subunit fusion protein transiently expressed in infiltrated Nicotiana benthamiana leaves. Plant Cell Tiss. Organ Cult. 127: 369–380.
Kim, T. G., N. Q. D. Tien, M. S. Yang, J. H. Moon, and J. Y. Lee (2016) Production of monoclonal antibodies against 53-kDa protein of Porphyromonas gingivalis in transgenic rice cell suspension. Plant Cell Tiss. Organ. Cult. 126: 387–397.
Kim, T. G., M. Y. Kim, and M. S. Yang (2010) Cholera toxin B subunit-domain III of dengue virus envelope glycoprotein E fusion protein production in transgenic plants. Protein Expr. Purif. 74: 236–241.
Van Haute, E., H. Joos, M. Maes, G. Warren, M. Van Montagu, and J. Schell (1981) Intergeneric transfer and exchange recombination of restriction fragments cloned in pBR322: a novel strategy for the reversed genetics of the Ti plasmids of Agrobacterium tumefaciens. EMBO J. 2: 411–417.
Kim, T. G., N. X. Huy, M. Y. Kim, D. K. Jeong, Y. S. Jang, M. S. Yang, W. H. R. Langridge, and J. Y. Lee (2009) Immunogenicity of a cholera toxin B subunit Porphyromonas gingivalis fimbrial antigen fusion protein expressed in E. coli. Mol. Biotechnol. 41: 157–164.
Kim, T. G. and W. H. R. Langridge (2003) Assembly of cholera toxin B subunit full-length rotavirus NSP4 fusion protein oligomers in transgenic potato. Plant Cell Rep. 21: 884–890.
Kong, Q., L. Richter, Y. F. Yang, C. J. Arntzen, H. S. Mason, and Y. Thanvala (2001) Oral immunization with hepatitis surface antigen expressed in transgenic plants. Proc. Natl. Acad. Sci. USA. 98: 11539–11544.
Yoshida, T., E. Kimura, S. Koike, J. Nojima, E. Futai, N. Sasagawa, Y. Watanabe, and S. Ishiura (2011) Transgenic rice expressing amyloid b-peptide for oral immunization. Int. J. Biol. Sci. 7: 301–307.
Jespersgaard, C., G. Hajishengallis, T. E. Greenway, D. J. Smith, M. W. Russell, and S. M. Michalek (1999) Functional and immunogenic characterization of two cloned regions of Streptococcus mutans glucosyltransferase I. Infect. Immun. 67: 810–816.
Kim, T. G., B. G. Kim, M. Y. Kim, J. K. Choi, E. S. Jung, and M. S. Yang (2010) Expression and immunogenicity of enterotoxigenic Escherichia coli heat-labile toxin B subunit in transgenic rice callus. Mol. Biotechnol. 44: 14–21.
Voinnet, O., Y. M. Pinto, and D. C. Baulcombe (1999) Suppression of gene silencing: a general strategy used by diverse DNA and RNA viruses of plants. Proc. Natl. Acad. Sci. USA. 96: 14147–14152.
Sun, J. B., J. Holmgren, and C. Czerkinsky (1994) Cholera toxin B subunit: an efficient transmucosal carrier-delivery system for induction of peripheral immunological tolerance. Proc. Natl. Acad. Sci. USA. 91: 10795–10799.
Shin, E. A., J. Y. Lee, T. G. Kim, Y. K. Park, and W. H. R. Langridge (2006) Synthesis and assembly of an adjuvanted Porphyromonas gingivalis fimbrial antigen fusion protein in plants. Protein Expr. Purif. 47: 99–109.
Kim, T. G., M. Y. Kim, N. X. Huy, S. H. Kim, and Y. S. Yang (2013) M cell-targeting ligand and consensus dengue virus envelope protein domain III fusion protein in transgenic rice calli. Mol. Biotechnol. 54: 880–887.
Yuki, Y., M. Mejima, S. Kurokawa, T. Hiroiwa, Y. Takahashi, D. Tokuhara, T. Nochi, Y. Katakai, M. Kuroda, N. Takeyama, K. Kashima, M. Abe, Y. Chen, U. Nakanishi, T. Masumura, Y. Takeuchi, H. Kozuka-Hata, H. Shibata, M. Oyama, K. Tanaka, and H. Kiyono (2013) Induction of toxin-specific neutralizing immunity by molecularly uniform rice-based oral cholera toxin B subunit vaccine without plant-associated sugar modification. Plant Biotechnol. J. 11: 799–808.
Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) grant (NRF-2016R1D1A1B03932450) funded by the Korea government (MSIP; Ministry of Science, ICT & Future Planning) and Research Base Construction Fund Support Program funded by Chonbuk National University in 2018.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kim, TG., Lan, T.T. & Lee, JY. Immunogenicity of Fusion Protein of Cholera Toxin B Subunit-Porphyromonas gingivalis 53-kDa Minor Fimbrial Protein Produced in Nicotiana benthamiana. Biotechnol Bioproc E 24, 954–963 (2019). https://doi.org/10.1007/s12257-019-0175-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-019-0175-6