Abstract
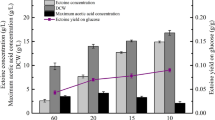
Tryptone has multiple and complex effects on cell physiology and process performance in pulse fed-batch cultivation of recombinant Escherichia coli. By applying feedback control of dissolved oxygen signal responding to pulse in the feed rate, the production of acetate was avoided and the optimization of production of recombinant human epidermal growth factor (hEGF) was successfully achieved. With the addition of an optimum amount of tryptone along with glucose in the pulse fedbatch cultivation of E. coli, the ability of the cell to divide and the stability of the plasmid within the bacteria were improved. Consequently, segregation of the cells into a viable but non-culturable physiological state was alleviated. Addition of tryptone also enhanced cell respiration before and after hEGF expression and thus further benefited the production of recombinant hEGF. Excessive addition of tryptone resulted in low sensitivity of the oscillation of dissolved oxygen signal and poor operability of pulse fed-batch cultivation as this led to an accumulation of acetate, which weakened the dissolved oxygen signal responses. Consequently, the production of recombinant protein was considerably reduced. By combining the process performance and the positive effect of complex media pulse addition on bacterial metabolism, the optimal production conditions of hEGF were successfully determined. A high cell density of 91 g/L dry cell weight was obtained under these optimal production conditions. Furthermore, a high level of 0.24 g/L hEGF was attained leading to a 32.6% increase in product yield as compared to the controls.
Similar content being viewed by others
References
Yee, L. and H. W. Blanch (1993) Recombinant trypsin production in high cell density fed-batch cultures in Escherichia coli. Biotechnol. Bioeng. 41: 781–790.
Aristidou, A. A., K. Y. San, and G. N. Bennett (1994) Modification of central metabolic pathway in Escherichia coil to reduce acetate accumulation by heterologous expression of the Bacillus subtilis acetolactate synthase gene. Biotechnol. Bioeng. 44: 944–951.
Seeger, A., B. Schneppe, J. E. G. McCarthy, W. D. Deckwer, and U. Rinas (1995) Comparison of temperature- and isopropyl-β-D-thiogalacto-pyranoside-induced synthesis of basic fibroblast growth factor in high-cell- density cultures of recombinant Escherichia coli. Enzyme Microb. Technol. 17: 947–953.
Borth, N., R. Mitterbauer, D. Mattanovich, W. Kramer, K. Bayer, and H. Katinger (1998) Flow cytometric analysis of bacterial physiology during induction of foreign protein synthesis in recombinant Escherichia coli cells. Cytometry 31: 125–129.
Lin, H., F. Hoffmann, A. Rozkov, S. O. Enfors, U. Rinas, and P. Neubauer (2004) Change of extracellular cAMP concentration is a sensitive reporter for bacterial fitness in high-cell-density cultures of Escherichia coli. Biotechnol. Bioeng. 87: 602–613.
Hewitt, C. J., H. Onyeaka, G. Lewis, I. W. Taylor, and A. W. Nienow (2007) A comparison of high cell density fed-batch fermentations involving both induced and non-induced recombinant Escherichia coli under wellmixed small-scale and simulated poorly mixed largescale conditions. Biotechnol. Bioeng. 96: 495–505.
Gschaedler, A. and J. Boudrant (1994) Amino acids utilization during batch and continuous cultures of Escherichia coli on a semi-synthetic medium. J. Biotechnol. 37: 235–251.
Prüss, B. M., J. M. Nelms, C. Park, and A. J. Wolfe (1994) Mutations in NADH: ubiquinone oxidoreductase of Escherichia coli affect growth on mixed amino acids. J. Bacteriol. 176: 2143–2150.
Suárez, D. C., C. W. Liria, and B. V. Kilikian (1998) Effect of yeast extract on Escherichia coli growth and acetic acid production. World J. Microbiol. Biotechnol. 14: 331–335.
Zheng, Z. Y., S. J. Yao, and D. Q. Lin (2005) Using a kinetic model that considers cell segregation to optimize hEGF expression in fed-batch cultures of recombinant Escherichia coli. Bioprocess Biosyst. Eng. 27: 143–152.
Johnston, W., R. Cord-Ruwisch, and M. J. Cooney (2002) Industrial control of recombinant E. coli fedbatch culture: new perspectives on traditional controlled variables. Bioprocess Biosyst. Eng. 25: 111–120.
Sumner, J. B. and G. F. Somers (1949) Dinitrosalicylic method for glucose. pp. 38–39. In: J. B. Sumner and G. F. Somers (eds.). Laboratory Experiments in Biological Chemistry. Academic Press, New York, USA.
Thom, S. M., R. W. Horobin, E. Seidler, and M. R. Barer (1993) Factors effecting the selection and use of tetrazolium salts as cytochemical indicators of microbial viability and activity. J. Appl. Bacteriol. 74: 433–443.
Velut, S., A. Castan, K. A. Short, J. P. Axelsson, P. Hagander, B. A. Zditosky, C. W. Rysenga, L. de Maré, and J. Haglund (2006) Influence of bioreactor scale and complex medium on probing control of glucose feeding in cultivations of recombinant strains of Escherichia coli. Biotechnol. Bioeng. 97: 816–824.
Lee, S. Y. (1996) High cell-density culture of Escherichia coli. Trends Biotechnol. 14: 98–105.
Zhang, J., J. Reddy, B. Buckland, and R. Greasham (2003) Toward consistent and productive complex media for industrial fermentations: studies on yeast extract for a recombinant yeast fermentation process. Biotechnol. Bioeng. 82: 640–652.
Åkesson, M., E. N. Karlsson, P. Hagander, J. P. Axelsson, and A. Tocaj (1999) On-line detection of acetate formation in Escherichia coli cultures using dissolved oxygen responses to feed transients. Biotechnol. Bioeng. 64: 590–598.
Swartz, J. R. (1996) Escherichia coli recombinant DNA technology. pp. 1693–1711. In: F. C. Neidhardt, R. Curtiss III, and J. L. Ingraham (eds.). Escherichia coli and Salmonella: Cellular and Molecular Biology. ASM Press, Washington, DC, USA.
Wang, Z. and N. A. Da Silva (1993) Improved protein synthesis and secretion through medium enrichment in a stable recombinant yeast strain. Biotechnol. Bioeng. 42: 95–102.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zheng, ZY., Yao, SJ., Zhan, X. et al. Improvement of hEGF production with enhanced cell division ability using dissolved oxygen responses to pulse addition of tryptone. Biotechnol Bioproc E 14, 52–59 (2009). https://doi.org/10.1007/s12257-008-0052-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-008-0052-1