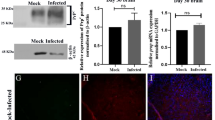
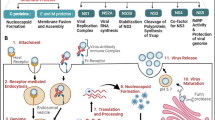
Abstract
Japanese encephalitis virus (JEV) is a leading cause of viral encephalitis in endemic regions of Asia. The neurotropism of JEV and its high-efficiency replication in neurons are the key events for pathogenesis. Revealing the interplay between virus and host cells in metabolic facet is of great importance both for unraveling the pathogenesis mechanisms and providing novel antiviral targets. This study took advantage of the integration analysis of metabolomics and transcriptomics to depict the metabolic profiles of neurons during the early stage of JEV infection. Increased glycolysis and its branched pentose phosphate pathway (PPP) flux and impaired oxidative phosphorylation (OXPHOS) in glucose utilization, and the catabolic patterns of lipid metabolism were created to facilitate the biosynthesis of precursors needed for JEV replication in neurons. Pharmacological inhibitions of both glycolysis pathway and PPP in neurons suggested its indispensable role in maintaining the optimal propagation of JEV. In addition, analysis of metabolomic-transcriptomic regulatory network showed the pivotal biological function of lipid metabolism during JEV infection. Several pro-inflammatory lipid metabolites were significantly up-regulated and might partially be responsible for the progression of encephalitis. These unique metabolic reprogramming features might give deeper insight into JEV infected neurons and provide promising antiviral approaches targeting metabolism.





Similar content being viewed by others
Change history
24 November 2021
A Correction to this paper has been published: https://doi.org/10.1007/s12250-021-00468-7
References
Allonso D, Andrade IS, Conde JN, Coelho DR, Rocha DC, Da SM, Ventura GT, Silva EM, Mohana-Borges R (2015) Dengue virus NS1 protein modulates cellular energy metabolism by increasing glyceraldehyde-3-phosphate dehydrogenase activity. J Virol 89:11871–11883
Barrows NJ, Campos RK, Liao KC, Prasanth KR, Soto-Acosta R, Yeh SC, Schott-Lerner G, Pompon J, Sessions OM, Bradrick SS, Garcia-Blanco MA (2018) Biochemistry and molecular biology of flaviviruses. Chem Rev 118:4448–4482
Bian P, Zheng X, Wei L, Ye C, Fan H, Cai Y, Zhang Y, Zhang F, Jia Z, Lei Y (2017) MLKL Mediated necroptosis accelerates JEV-induced neuroinflammation in mice. Front Microbiol 8:303
Brooks GA (2020) Lactate as a fulcrum of metabolism. Redox Biol 35:101454
Cai TT, Ye SB, Liu YN, He J, Chen QY, Mai HQ, Zhang CX, Cui J, Zhang XS, Busson P, Zeng YX, Li J (2017) LMP1-mediated glycolysis induces myeloid-derived suppressor cell expansion in nasopharyngeal carcinoma. PLoS Pathog 13:e1006503
Chen YH, Du W, Hagemeijer MC, Takvorian PM, Pau C, Cali A, Brantner CA, Stempinski ES, Connelly PS, Ma HC, Jiang P, Wimmer E, Altan-Bonnet G, Altan-Bonnet N (2015) Phosphatidylserine vesicles enable efficient en bloc transmission of enteroviruses. Cell 160:619–630
Erlanger TE, Weiss S, Keiser J, Utzinger J, Wiedenmayer K (2009) Past, present, and future of Japanese encephalitis. Emerg Infect Dis 15:1–7
Fahy E, Cotter D, Sud M, Subramaniam S (2011) Lipid classification, structures and tools. Biochim Biophys Acta 1811:637–647
Fontaine KA, Sanchez EL, Camarda R, Lagunoff M (2015) Dengue virus induces and requires glycolysis for optimal replication. J Virol 89:2358–2366
Hanna VS, Hafez E (2018) Synopsis of arachidonic acid metabolism: a review. J Adv Res 11:23–32
Leier HC, Messer WB, Tafesse FG (2018) Lipids and pathogenic flaviviruses: an intimate union. PLoS Pathog 14:e1006952
Ma T, Patel H, Babapoor-Farrokhran S, Franklin R, Semenza GL, Sodhi A, Montaner S (2015) KSHV induces aerobic glycolysis and angiogenesis through HIF-1-dependent upregulation of pyruvate kinase 2 in Kaposi’s sarcoma. Angiogenesis 18:477–488
Martin-Acebes MA, Jimenez DON, Saiz JC (2019) Lipid metabolism as a source of druggable targets for antiviral discovery against zika and other flaviviruses. Pharmaceuticals (basel) 12:97
Martin-Acebes MA, Vazquez-Calvo A, Saiz JC (2016) Lipids and flaviviruses, present and future perspectives for the control of dengue, Zika, and West Nile viruses. Prog Lipid Res 64:123–137
Mayer KA, Stockl J, Zlabinger GJ, Gualdoni GA (2019) Hijacking the supplies: metabolism as a novel facet of virus-host interaction. Front Immunol 10:1533
Mayer N, Schweiger M, Romauch M, Grabner GF, Eichmann TO, Fuchs E, Ivkovic J, Heier C, Mrak I, Lass A, Hofler G, Fledelius C, Zechner R, Zimmermann R, Breinbauer R (2013) Development of small-molecule inhibitors targeting adipose triglyceride lipase. Nat Chem Biol 9:785–787
Mazzon M, Mercer J (2014) Lipid interactions during virus entry and infection. Cell Microbiol 16:1493–1502
Miller S, Krijnse-Locker J (2008) Modification of intracellular membrane structures for virus replication. Nat Rev Microbiol 6:363–374
Muller-McNicoll M, Rossbach O, Hui J, Medenbach J (2019) Auto-regulatory feedback by RNA-binding proteins. J Mol Cell Biol 11:930–939
Passalacqua KD, Lu J, Goodfellow I, Kolawole AO, Arche JR, Maddox RJ, Carnahan KE, O'Riordan M, Wobus CE (2019) Glycolysis Is an Intrinsic Factor for Optimal Replication of a Norovirus. mBio 10:e02175–18
Rodriguez-Sanchez I, Munger J (2019) Meal for Two: Human Cytomegalovirus-Induced Activation of Cellular Metabolism. Viruses 11:273
Strating JR, van Kuppeveld FJ (2017) Viral rewiring of cellular lipid metabolism to create membranous replication compartments. Curr Opin Cell Biol 47:24–33
Su MA, Huang YT, Chen IT, Lee DY, Hsieh YC, Li CY, Ng TH, Liang SY, Lin SY, Huang SW, Chiang YA, Yu HT, Khoo KH, Chang GD, Lo CF, Wang HC (2014) An invertebrate warburg effect: a shrimp virus achieves successful replication by altering the host metabolome via the PI3K-Akt-mTOR pathway. PLoS Pathog 10:e1004196
Thaker SK, Chapa T, Garcia GJ, Gong D, Schmid EW, Arumugaswami V, Sun R, Christofk HR (2019a) Differential metabolic reprogramming by zika virus promotes cell death in human versus mosquito cells. Cell Metab 29:1206–1216
Thaker SK, Ch’Ng J, Christofk HR (2019b) Viral hijacking of cellular metabolism. BMC Biol 17:59
Thyrsted J, Holm CK (2020) Virus-induced metabolic reprogramming and innate sensing hereof by the infected host. Curr Opin Biotechnol 68:44–50
Vilela MC, Lima GK, Rodrigues DH, Lacerda-Queiroz N, Pedroso VS, de Miranda AS, Rachid MA, Kroon EG, Campos MA, Teixeira MM, Teixeira AL (2016) Platelet activating factor (PAF) receptor deletion or antagonism attenuates severe HSV-1 meningoencephalitis. J Neuroimmune Pharmacol 11:613–621
Warburg O (1956) On the origin of cancer cells. Science 123:309–314
Zhang W, Wang G, Xu ZG, Tu H, Hu F, Dai J, Chang Y, Chen Y, Lu Y, Zeng H, Cai Z, Han F, Xu C, ** G, Sun L, Pan BS, Lai SW, Hsu CC, Xu J, Chen ZZ, Li HY, Seth P, Hu J, Zhang X, Li H, Lin HK (2019a) Lactate is a natural suppressor of RLR signaling by targeting MAVS. Cell 178:176–189
Zhang Z, He G, Filipowicz NA, Randall G, Belov GA, Kopek BG, Wang X (2019b) Host lipids in positive-strand RNA Virus genome replication. Front Microbiol 10:286
Acknowledgements
This research was funded by the National Major Science and Technology Projects of China, grant number 2017ZX10202203-007-003.
Author information
Authors and Affiliations
Contributions
JL and YL designed the experiments. ML and JY carried out the experiments and wrote the original draft. CY, XY, HZ, CL and ZX analyzed the data. JL and YL reviewed the manuscript and provided the financial support. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Animal and Human Rights Statement
This article does not contain any studies with human or animal subjects performed by any of the authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, M., Yang, J., Ye, C. et al. Integrated Metabolomics and Transcriptomics Analyses Reveal Metabolic Landscape in Neuronal Cells during JEV Infection. Virol. Sin. 36, 1554–1565 (2021). https://doi.org/10.1007/s12250-021-00445-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12250-021-00445-0