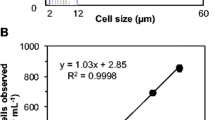
Abstract
The nutrient uptake capacity of microalgae can be applied in advanced wastewater treatment processes. This study was set to develop a rapid and effective method for assessing the viability (i.e., live or dead) of microalgae applied for advanced wastewater treatment using a flow cytometry. Especially, a methodology to selectively evaluate the quantitative viability of only microalgae present in wastewaters having the co-culture of microalgae and bacteria was proposed. The live and dead cells of microalgae could be discriminated using chlorophyll autofluorescence and the SYTOX Green method developed in this study. Cell viability in live and dead conditions of microalgae showed about 90% accuracy, and about 40% viability ratio was derived in half-live and half-dead conditions. This result shows the potential to quantitatively evaluate the activity of microalgae in the wastewater environment.
Similar content being viewed by others
References
Choi K-J, Han TH, Yoo G, Cho MH, Hwang S-J (2018) Co-culture consortium of Scenedesmus dimorphus and nitrifiers enhances the removal of nitrogen and phosphorus from artificial wastewater. KSCE Journal of Civil Engineering 22(9):3215–3221, DOI: https://doi.org/10.1007/s12205-017-0730-7
Daliry S, Hallajisani A, Mohammadi Roshandeh J, Nouri H, Golzary A (2017) Investigation of optimal condition for Chlorella vulgaris microalgae growth. Global Journal of Environmental Science and Management 3(2):217–230, DOI: https://doi.org/10.22034/gjesm.2017.03.02.010
de la Noue J, de Pauw N (1988) The potential of microalgal biotechnology: A review of production and uses of microalgae. Biotechnology Advances 6(4):725–770, DOI: https://doi.org/10.1016/0734-9750(88)91921-0
Di Caprio F (2020) Methods to quantify biological contaminants in microalgae cultures. Algal Research 49:101943, DOI: https://doi.org/10.1016/j.algal.2020.101943
García D, Esther P, Carlos G, Saúl B, Sonia M-P, Gabriel A, Pedro G-E, Silvia B, Raúl M (2017) Comparative evaluation of piggery wastewater treatment in algal-bacterial photobioreactors under indoor and outdoor conditions. Bioresource Technology 245:483–490, DOI: https://doi.org/10.1016/j.biortech.2017.08.135
Gasol JM, Giorgio PAD (2000) Using flow cytometry for counting natural planktonic bacteria and understanding the structure of planktonic bacterial communities. Scientia Marina 64(2):197–224, DOI: https://doi.org/10.3989/scimar.2000.64n2197
Hyka P, Lickova S, Pfibyl P, Melzoch K, Kovar K (2013) Flow cytometry for the development of biotechnological processes with microalgae. Biotechnology Advances 31(1):2–16, DOI: https://doi.org/10.1016/j.biotechadv.2012.04.007
Kuba TG, Smolders MCM, Van Loosdrecht, Heijnen JJ (1993) Biological phosphorus removal from wastewater by anaerobic-anoxic sequencing batch reactor. Water Science and Technology 27(5–6):241–252, DOI: https://doi.org/10.2166/wst.1993.0504
Lee M-J, Song J-H, Hwang S-J (2009) Effects of acid pre-treatment on bio-hydrogen production and microbial communities during dark fermentation. Bioresource Technology 100(3):1491–1493, DOI: https://doi.org/10.1016/j.biortech.2008.08.019
Li WKW, Dickie PM (2001) Monitoring phytoplankton bacterioplankton, and virioplankton in a coastal inlet (Bedford Basin) by flow cytometry. Cytometry: The Journal of the International Society for Analytical Cytology 44(3):236–246, DOI: https://doi.org/10.1002/1097-0320(20010701)44:3<236::aid-cyto1116>3.0.co;2-5
Liddeli PA, Nemeth GA, Lehman WR, Joy AM, Moore AL, Bensasson RV, Moore TA, Gust D (1982) Mimicry of carotenoid function in photosynthesis: Synthesis and photophysical properties of a carotenopyropheophorbide. Photochemistry and Photobiology 36(6): 641–645, DOI: https://doi.org/10.1111/j.1751-1097.1982.tb09484.x
Marie D, Partensky F, Vaulot D, Brussaard C (1999) Enumeration of phytoplankton, bacteria, and viruses in marine samples. Current Protocols in Cytometry 10(1), DOI: https://doi.org/10.1002/0471142956.cy1111s10
Mohsenpour SF, Hennige S, Willoughby N, Adeloye A, Gutierrez T (2021) Integrating micro-algae into wastewater treatment: A review. Science of The Total Environment 752:142168, DOI: https://doi.org/10.1016/j.scitotenv.2020.142168
Peperzak L, Brussaard CPD (2011) Flow cytometric applicability of fluorescent vitality probes on phytoplankton 1. Journal of Phycology 47(3):692–702, DOI: https://doi.org/10.1111/j.1529-8817.2011.00991.x
Rachlin JW, Grosso A (1991) The effects of pH on the growth of chlorella vulgaris and its interactions with cadmium toxicity. Archives of Environmental Contamination and Toxicology 20(4):505–508, DOI: https://doi.org/10.1007/BF01065839
Rychtecký P, Znachor P, Nedoma J (2014) Spatio-temporal study of phytoplankton cell viability in a eutrophic reservoir using SYTOX Green nucleic acid stain. Hydrobiologia 740(1):177–189, DOI: https://doi.org/10.1007/s10750-014-1952-z
Sato M, Murata Y, Mizusawa M, Iwahashi H, Oka S (2004) A simple and rapid dual-fluorescence viability assay for microalgae. Microbiol Cult Coll 20(2):53–59, http://jsmrs.jp/journal/No20_2/No20_2_53.pdf
Scragg AH, Morrison J, Shales SW (2003) The use of a fuel containing Chlorella vulgaris in a diesel engine. Enzyme and Microbial Technology 33(7):884–889, DOI: https://doi.org/10.1016/j.enzmictec.2003.01.001
Suo Y, Gu Z, Wei X (2020) Advances of in vivo flow cytometry on cancer studies. Cytometry Part A 97(1):15–23, DOI: https://doi.org/10.1002/cyto.a.23851
Taborda T, Moniz P, Reis A, da Silva TL (2021) Evaluating low-cost substrates for Crypthecodinium cohnii lipids and DHA production, by flow cytometry. Journal of Applied Phycology 33(1):263–274, DOI: https://doi.org/10.1007/s10811-020-02304-z
Tait K, White DA, Kimmance SA, Tarran G, Rooks P, Jones M, Llewellyn CA (2019) Characterisation of bacteria from the cultures of a Chlorella strain isolated from textile wastewater and their growth enhancing effects on the axenic cultures of Chlorella vulgaris in low nutrient media. Algal Research 44:101666, DOI: https://doi.org/10.1016/j.algal.2019.101666
Tang C-C, Zuo W, Tian Y, Sun N, Wang Z-W, Zhang J (2016) Effect of aeration rate on performance and stability of algal-bacterial symbiosis system to treat domestic wastewater in sequencing batch reactors. Bioresource Technology 222:156–164, DOI: https://doi.org/10.1016/j.biortech.2016.09.123
Tiron O, Bumbac C, Manea E, Stefanescu M, Lazar MN (2017) Overcoming microalgae harvesting barrier by activated algae granules. Scientific Reports 7(1):1–11, DOI: https://doi.org/10.1038/s41598-017-05027-3
Verma R, Suthar S, Chand N, Mutiyar PK (2022) Phycoremediation of milk processing wastewater and lipid-rich biomass production using Chlorella vulgaris under continuous batch system. Science of The Total Environment 833:155110, DOI: https://doi.org/10.1016/j.scitotenv.2022.155110
Wang H, Li F, Keller AA, Xu R (2009) Chemically enhanced primary treatment (CEPT) for removal of carbon and nutrients from municipal wastewater treatment plants: A case study of Shanghai. Water Science and Technology 60(7):1803–1809, DOI: https://doi.org/10.2166/wst.2009.547
Zetsche E-M, Meysman FJR (2012) Dead or alive? Viability assessment of micro-and mesoplankton. Journal of Plankton Research 34(6):493–509, DOI: https://doi.org/10.1093/plankt/fbs018
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) (Grant number 2019R1A2C1084155).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, KH., Jho, E.H. & Hwang, SJ. Quantitative Viability Assessment of Microalgae for Advanced Wastewater Treatment by Flow Cytometry. KSCE J Civ Eng 27, 3714–3719 (2023). https://doi.org/10.1007/s12205-023-0125-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12205-023-0125-x