Abstract
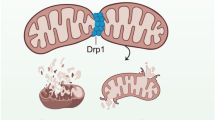
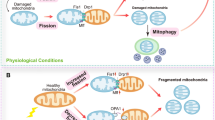
Myocardial microvessels are composed of a monolayer of endothelial cells, which play a crucial role in maintaining vascular barrier function, luminal latency, vascular tone, and myocardial perfusion. Endothelial dysfunction is a key factor in the development of cardiac microvascular injury and diabetic cardiomyopathy. In addition to their role in glucose oxidation and energy metabolism, mitochondria also participate in non-metabolic processes such as apoptosis, intracellular ion handling, and redox balancing. Mitochondrial dynamics and mitophagy are responsible for regulating the quality and quantity of mitochondria in response to hyperglycemia. However, these endogenous homeostatic mechanisms can both preserve and/or disrupt non-metabolic mitochondrial functions during diabetic endothelial damage and cardiac microvascular injury. This review provides an overview of the molecular features and regulatory mechanisms of mitochondrial dynamics and mitophagy. Furthermore, we summarize findings from various investigations that suggest abnormal mitochondrial dynamics and defective mitophagy contribute to the development of diabetic endothelial dysfunction and myocardial microvascular injury. Finally, we discuss different therapeutic strategies aimed at improving endothelial homeostasis and cardiac microvascular function through the enhancement of mitochondrial dynamics and mitophagy.




Similar content being viewed by others
References
Abbatecola AM, Evans W, Paolisso G (2009) PUFA supplements and type 2 diabetes in the elderly. Curr Pharm Des 15:4126–4134. https://doi.org/10.2174/138161209789909782
Apaijai N, Pintana H, Chattipakorn SC, Chattipakorn N (2013) Effects of vildagliptin versus sitagliptin, on cardiac function, heart rate variability and mitochondrial function in obese insulin-resistant rats. Br J Pharmacol 169:1048–1057. https://doi.org/10.1111/bph.12176
Ashrafi G, Schwarz TL (2013) The pathways of mitophagy for quality control and clearance of mitochondria. Cell Death Differ 20:31–42. https://doi.org/10.1038/cdd.2012.81
Avogaro A, Fadini GP (2019) Microvascular complications in diabetes: a growing concern for cardiologists. Int J Cardiol 291:29–35. https://doi.org/10.1016/j.ijcard.2019.02.030
Bakuy V, Unal O, Gursoy M et al (2014) Electron microscopic evaluation of internal thoracic artery endothelial morphology in diabetic coronary bypass patients. Ann Thorac Surg 97:851–857. https://doi.org/10.1016/j.athoracsur.2013.09.102
Barclay AW, Flood VM, Rochtchina E, Mitchell P, Brand-Miller JC (2007) Glycemic index, dietary fiber, and risk of type 2 diabetes in a cohort of older Australians. Diabetes Care 30:2811–2813. https://doi.org/10.2337/dc07-0784
Barrett EJ, Liu Z, Khamaisi M et al (2017) Diabetic microvascular disease: an Endocrine Society Scientific Statement. J Clin Endocrinol Metab 102:4343–4410. https://doi.org/10.1210/jc.2017-01922
Bassuk SS, Manson JE (2005) Epidemiological evidence for the role of physical activity in reducing risk of type 2 diabetes and cardiovascular disease. J Appl Physiol 1985(99):1193–1204. https://doi.org/10.1152/japplphysiol.00160.2005
Batchuluun B, Inoguchi T, Sonoda N et al (2014) Metformin and liraglutide ameliorate high glucose-induced oxidative stress via inhibition of PKC-NAD(P)H oxidase pathway in human aortic endothelial cells. Atherosclerosis 232:156–164. https://doi.org/10.1016/j.atherosclerosis.2013.10.025
Bernardini JP, Lazarou M, Dewson G (2017) Parkin and mitophagy in cancer. Oncogene 36:1315–1327. https://doi.org/10.1038/onc.2016.302
Bhatt MP, Lim YC, Kim YM, Ha KS (2013) C-peptide activates AMPKα and prevents ROS-mediated mitochondrial fission and endothelial apoptosis in diabetes. Diabetes 62:3851–3862. https://doi.org/10.2337/db13-0039
Blomhoff R (2005) Dietary antioxidants and cardiovascular disease. Curr Opin Lipidol 16:47–54. https://doi.org/10.1097/00041433-200502000-00009
Bravo-San Pedro JM, Kroemer G, Galluzzi L (2017) Autophagy and mitophagy in cardiovascular disease. Circ Res 120:1812–1824. https://doi.org/10.1161/circresaha.117.311082
Buse JB, Ginsberg HN, Bakris GL et al (2007) Primary prevention of cardiovascular diseases in people with diabetes mellitus: a scientific statement from the American Heart Association and the American Diabetes Association. Circulation 115:114–126. https://doi.org/10.1161/circulationaha.106.179294
Caballero AE (2003) Endothelial dysfunction in obesity and insulin resistance: a road to diabetes and heart disease. Obes Res 11:1278–1289. https://doi.org/10.1038/oby.2003.174
Cai C, Guo Z, Chang X et al (2022) Empagliflozin attenuates cardiac microvascular ischemia/reperfusion through activating the AMPKα1/ULK1/FUNDC1/mitophagy pathway. Redox Biol 52:102288. https://doi.org/10.1016/j.redox.2022.102288
Ceriello A, De Nigris V, Pujadas G et al (2016) The simultaneous control of hyperglycemia and GLP-1 infusion normalize endothelial function in type 1 diabetes. Diabetes Res Clin Pract 114:64–68. https://doi.org/10.1016/j.diabres.2016.01.019
Ceriello A, Esposito K, La Sala L et al (2014) The protective effect of the Mediterranean diet on endothelial resistance to GLP-1 in type 2 diabetes: a preliminary report. Cardiovasc Diabetol 13:140. https://doi.org/10.1186/s12933-014-0140-9
Chan DC (2020) Mitochondrial dynamics and its involvement in disease. Annu Rev Pathol 15:235–259. https://doi.org/10.1146/annurev-pathmechdis-012419-032711
Chan NN (2001) Improved endothelial function with metformin in type 2 diabetes mellitus. J Am Coll Cardiol 38:2131–2132. https://doi.org/10.1016/s0735-1097(01)01630-8
Chang X, Li Y, Cai C et al (2022a) Mitochondrial quality control mechanisms as molecular targets in diabetic heart. Metabolism 137:155313. https://doi.org/10.1016/j.metabol.2022.155313
Chang X, Lochner A, Wang HH et al (2021) Coronary microvascular injury in myocardial infarction: perception and knowledge for mitochondrial quality control. Theranostics 11:6766–6785. https://doi.org/10.7150/thno.60143
Chang X, Toan S, Li R, Zhou H (2022b) Therapeutic strategies in ischemic cardiomyopathy: focus on mitochondrial quality surveillance. EBioMedicine 84:104260. https://doi.org/10.1016/j.ebiom.2022.104260
Cheang WS, Wong WT, Wang L et al (2019) Resveratrol ameliorates endothelial dysfunction in diabetic and obese mice through sirtuin 1 and peroxisome proliferator-activated receptor δ. Pharmacol Res 139:384–394. https://doi.org/10.1016/j.phrs.2018.11.041
Chen F, He L, Li J et al (2022) Polyethylene glycol loxenatide injection (GLP-1) protects vascular endothelial cell function in middle-aged and elderly patients with type 2 diabetes by regulating gut microbiota. Front Mol Biosci 9:879294. https://doi.org/10.3389/fmolb.2022.879294
Chen W, **ang H, Chen R et al (2019) S1PR2 antagonist ameliorate high glucose-induced fission and dysfunction of mitochondria in HRGECs via regulating ROCK1. BMC Nephrol 20:135. https://doi.org/10.1186/s12882-019-1323-0
Cho SS, Qi L, Fahey GC Jr, Klurfeld DM (2013) Consumption of cereal fiber, mixtures of whole grains and bran, and whole grains and risk reduction in type 2 diabetes, obesity, and cardiovascular disease. Am J Clin Nutr 98:594–619. https://doi.org/10.3945/ajcn.113.067629
Clyne AM (2021) Endothelial response to glucose: dysfunction, metabolism, and transport. Biochem Soc Trans 49:313–325. https://doi.org/10.1042/bst20200611
Cooper CE, Vollaard NB, Choueiri T, Wilson MT (2002) Exercise, free radicals and oxidative stress. Biochem Soc Trans 30:280–285
De Jager J, Kooy A, Lehert P et al (2005) Effects of short-term treatment with metformin on markers of endothelial function and inflammatory activity in type 2 diabetes mellitus: a randomized, placebo-controlled trial. J Intern Med 257:100–109. https://doi.org/10.1111/j.1365-2796.2004.01420.x
de Lemos E T, Reis F, Baptista S, et al. (2007). Exercise training is associated with improved levels of C-reactive protein and adiponectin in ZDF (type 2) diabetic rats. Med Sci Monit 13:Br168-174.
Di Tomo P, Alessio N, Falone S et al (2021) Endothelial cells from umbilical cord of women affected by gestational diabetes: a suitable in vitro model to study mechanisms of early vascular senescence in diabetes. FASEB J 35:e21662. https://doi.org/10.1096/fj.202002072RR
Doğanlar ZB, Doğanlar O, Kurtdere K et al (2021) Melatonin prevents blood-retinal barrier breakdown and mitochondrial dysfunction in high glucose and hypoxia-induced in vitro diabetic macular edema model. Toxicol In Vitro 75:105191. https://doi.org/10.1016/j.tiv.2021.105191
Donnelly D (2012) The structure and function of the glucagon-like peptide-1 receptor and its ligands. Br J Pharmacol 166:27–41. https://doi.org/10.1111/j.1476-5381.2011.01687.x
Drew BG, Ribas V, Le JA et al (2014) HSP72 is a mitochondrial stress sensor critical for Parkin action, oxidative metabolism, and insulin sensitivity in skeletal muscle. Diabetes 63:1488–1505. https://doi.org/10.2337/db13-0665
Duraisamy AJ, Mohammad G, Kowluru RA (2019) Mitochondrial fusion and maintenance of mitochondrial homeostasis in diabetic retinopathy. Biochim Biophys Acta Mol Basis Dis 1865:1617–1626. https://doi.org/10.1016/j.bbadis.2019.03.013
Durand MJ, Ait-Aissa K, Levchenko V et al (2019) Visualization and quantification of mitochondrial structure in the endothelium of intact arteries. Cardiovasc Res 115:1546–1556. https://doi.org/10.1093/cvr/cvy294
Ergul A (2011) Endothelin-1 and diabetic complications: focus on the vasculature. Pharmacol Res 63:477–482. https://doi.org/10.1016/j.phrs.2011.01.012
Ersoy C, Kiyici S, Budak F et al (2008) The effect of metformin treatment on VEGF and PAI-1 levels in obese type 2 diabetic patients. Diabetes Res Clin Pract 81:56–60. https://doi.org/10.1016/j.diabres.2008.02.006
Fatima S, Ambreen S, Mathew A et al (2022) ER-stress and senescence coordinately promote endothelial barrier dysfunction in diabetes-induced atherosclerosis. Nutrients 14. https://doi.org/10.3390/nu14142786
Few JD, Unwin RJ, Carmichael DJ, James VH (1987) Diurnal fluctuation in saliva aldosterone concentration. J Steroid Biochem 26:265–271. https://doi.org/10.1016/0022-4731(87)90081-1
Gao S, Hu J (2021) Mitochondrial fusion: the machineries in and out. Trends Cell Biol 31:62–74. https://doi.org/10.1016/j.tcb.2020.09.008
Gey KF (1998) Vitamins E plus C and interacting conutrients required for optimal health. A critical and constructive review of epidemiology and supplementation data regarding cardiovascular disease and cancer. Biofactors 7:113–174. https://doi.org/10.1002/biof.5520070115
Giacco F, Brownlee M (2010) Oxidative stress and diabetic complications. Circ Res 107:1058–1070. https://doi.org/10.1161/circresaha.110.223545
Gillespie KM (2006) Type 1 diabetes: pathogenesis and prevention. Cmaj 175:165–170. https://doi.org/10.1503/cmaj.060244
Groschner L N, Waldeck-Weiermair M, Malli R, and Graier W F (2012). Endothelial mitochondria--less respiration, more integration. Pflugers Arch 464:63-76. https://doi.org/10.1007/s00424-012-1085-z.
Halvorsen BL, Carlsen MH, Phillips KM et al (2006) Content of redox-active compounds (ie, antioxidants) in foods consumed in the United States. Am J Clin Nutr 84:95–135. https://doi.org/10.1093/ajcn/84.1.95
Henning RJ (2018) Type-2 diabetes mellitus and cardiovascular disease. Future Cardiol 14:491–509. https://doi.org/10.2217/fca-2018-0045
Holst JJ (2007) The physiology of glucagon-like peptide 1. Physiol Rev 87:1409–1439. https://doi.org/10.1152/physrev.00034.2006
Hooper PL, Balogh G, Rivas E, Kavanagh K, Vigh L (2014) The importance of the cellular stress response in the pathogenesis and treatment of type 2 diabetes. Cell Stress Chaperones 19:447–464. https://doi.org/10.1007/s12192-014-0493-8
Hu J, Zhang H, Li J et al (2020) ROCK1 activation-mediated mitochondrial translocation of Drp1 and cofilin are required for arnidiol-induced mitochondrial fission and apoptosis. J Exp Clin Cancer Res 39:37. https://doi.org/10.1186/s13046-020-01545-7
Iannantuoni F, de Marañon AM, Abad-Jiménez Z et al (2020) Mitochondrial alterations and enhanced human leukocyte/endothelial cell interactions in type 1 diabetes. J Clin Med 9. https://doi.org/10.3390/jcm9072155
Ichikawa K, Miyoshi T, Osawa K et al (2022) High pericoronary adipose tissue attenuation on computed tomography angiography predicts cardiovascular events in patients with type 2 diabetes mellitus: post-hoc analysis from a prospective cohort study. Cardiovasc Diabetol 21:44. https://doi.org/10.1186/s12933-022-01478-9
Ji Z, Liu GH, Qu J (2022) Mitochondrial sirtuins, metabolism, and aging. J Genet Genomics 49:287–298. https://doi.org/10.1016/j.jgg.2021.11.005
Jia G, Whaley-Connell A, Sowers JR (2018) Diabetic cardiomyopathy: a hyperglycaemia- and insulin-resistance-induced heart disease. Diabetologia 61:21–28. https://doi.org/10.1007/s00125-017-4390-4
** H, Zhu Y, Li Y et al (2019) BDNF-mediated mitophagy alleviates high-glucose-induced brain microvascular endothelial cell injury. Apoptosis 24:511–528. https://doi.org/10.1007/s10495-019-01535-x
Kaur R, Kaur M, Singh J (2018) Endothelial dysfunction and platelet hyperactivity in type 2 diabetes mellitus: molecular insights and therapeutic strategies. Cardiovasc Diabetol 17:121. https://doi.org/10.1186/s12933-018-0763-3
Kelly AS, Bergenstal RM, Gonzalez-Campoy JM, Katz H, Bank A J (2012) Effects of exenatide vs. metformin on endothelial function in obese patients with pre-diabetes: a randomized trial. Cardiovasc Diabetol 11:64. https://doi.org/10.1186/1475-2840-11-64
Kim D, Roy S (2020) Effects of diabetes on mitochondrial morphology and its implications in diabetic retinopathy. Invest Ophthalmol Vis Sci 61:10. https://doi.org/10.1167/iovs.61.10.10
Kim D, Sesaki H, Roy S (2021) Reduced levels of Drp1 protect against development of retinal vascular lesions in diabetic retinopathy. Cells 10. https://doi.org/10.3390/cells10061379
Kim YM, Youn SW, Sudhahar V et al (2018) Redox regulation of mitochondrial fission protein Drp1 by protein disulfide isomerase limits endothelial senescence. Cell Rep 23:3565–3578. https://doi.org/10.1016/j.celrep.2018.05.054
Kluge MA, Fetterman JL, Vita JA (2013) Mitochondria and endothelial function. Circ Res 112:1171–1188. https://doi.org/10.1161/circresaha.111.300233
Knapp M, Tu X, Wu R (2019) Vascular endothelial dysfunction, a major mediator in diabetic cardiomyopathy. Acta Pharmacol Sin 40:1–8. https://doi.org/10.1038/s41401-018-0042-6
Kowluru RA, Mohammad G (2020) Epigenetics and mitochondrial stability in the metabolic memory phenomenon associated with continued progression of diabetic retinopathy. Sci Rep 10:6655. https://doi.org/10.1038/s41598-020-63527-1
Krysiak R, Okopien B (2012) Lymphocyte-suppressing and systemic anti-inflammatory effects of high-dose metformin in simvastatin-treated patients with impaired fasting glucose. Atherosclerosis 225:403–407. https://doi.org/10.1016/j.atherosclerosis.2012.09.034
Krysiak R, Okopien B (2013) The effect of metformin on monocyte secretory function in simvastatin-treated patients with impaired fasting glucose. Metabolism 62:39–43. https://doi.org/10.1016/j.metabol.2012.06.009
Lahera V, de Las HN, López-Farré A, Manucha W, Ferder L (2017) Role of mitochondrial dysfunction in hypertension and obesity. Curr Hypertens Rep 19:11. https://doi.org/10.1007/s11906-017-0710-9
Laukkanen T, Kunutsor SK, Khan H et al (2018) Sauna bathing is associated with reduced cardiovascular mortality and improves risk prediction in men and women: a prospective cohort study. BMC Med 16:219. https://doi.org/10.1186/s12916-018-1198-0
Lee HZ, Yeh FT, Wu CH (2004) The effect of elevated extracellular glucose on adherens junction proteins in cultured rat heart endothelial cells. Life Sci 74:2085–2096. https://doi.org/10.1016/j.lfs.2003.06.046
Losón OC, Song Z, Chen H, Chan DC (2013) Fis1, Mff, MiD49, and MiD51 mediate Drp1 recruitment in mitochondrial fission. Mol Biol Cell 24:659–667. https://doi.org/10.1091/mbc.E12-10-0721
Ma L, Zou R, Shi W et al (2022) SGLT2 inhibitor dapagliflozin reduces endothelial dysfunction and microvascular damage during cardiac ischemia/reperfusion injury through normalizing the XO-SERCA2-CaMKII-coffilin pathways. Theranostics 12:5034–5050. https://doi.org/10.7150/thno.75121
Ma X, Zhang J, Wu Z, Wang X (2021) Chicoric acid attenuates hyperglycemia-induced endothelial dysfunction through AMPK-dependent inhibition of oxidative/nitrative stresses. J Recept Signal Transduct Res 41:378–392. https://doi.org/10.1080/10799893.2020.1817076
Maeda D, Dotare T, Matsue Y et al (2023) Blood pressure in heart failure management and prevention. Hypertens Res. https://doi.org/10.1038/s41440-022-01158-x
Makino A, Scott BT, Dillmann WH (2010) Mitochondrial fragmentation and superoxide anion production in coronary endothelial cells from a mouse model of type 1 diabetes. Diabetologia 53:1783–1794. https://doi.org/10.1007/s00125-010-1770-4
Matsushima S, Sadoshima J (2015) The role of sirtuins in cardiac disease. Am J Physiol Heart Circ Physiol 309:H1375–H1389. https://doi.org/10.1152/ajpheart.00053.2015
Merdler I, Arbel Y (2021) Type II diabetes mellitus and endothelial dysfunction: what can we do? Isr Med Assoc J 23:121–122
Minamiyama Y, Takemura S, Bito Y et al (2008) Supplementation of alpha-tocopherol improves cardiovascular risk factors via the insulin signalling pathway and reduction of mitochondrial reactive oxygen species in type II diabetic rats. Free Radic Res 42:261–271. https://doi.org/10.1080/10715760801898820
Mone P, Varzideh F, Jankauskas SS et al (2022) SGLT2 inhibition via empagliflozin improves endothelial function and reduces mitochondrial oxidative stress: insights from frail hypertensive and diabetic patients. Hypertension 79:1633–1643. https://doi.org/10.1161/hypertensionaha.122.19586
Morgan PE, Sheahan PJ, Davies MJ (2014) Perturbation of human coronary artery endothelial cell redox state and NADPH generation by methylglyoxal. PloS One 9:e86564. https://doi.org/10.1371/journal.pone.0086564
Nazıroğlu M, Akkuş S, Soyupek F et al (2010) Vitamins C and E treatment combined with exercise modulates oxidative stress markers in blood of patients with fibromyalgia: a controlled clinical pilot study. Stress 13:498–505. https://doi.org/10.3109/10253890.2010.486064
Ng ACT, Delgado V, Borlaug BA, Bax JJ (2021) Diabesity: the combined burden of obesity and diabetes on heart disease and the role of imaging. Nat Rev Cardiol 18:291–304. https://doi.org/10.1038/s41569-020-00465-5
Nguyen TN, Padman BS, Lazarou M (2016) Deciphering the molecular signals of PINK1/Parkin mitophagy. Trends Cell Biol 26:733–744. https://doi.org/10.1016/j.tcb.2016.05.008
Ning P, Jiang X, Yang J et al (2022) Mitophagy: a potential therapeutic target for insulin resistance. Front Physiol 13:957968. https://doi.org/10.3389/fphys.2022.957968
Petite SE, Hill MC (2021) Evaluation of dipeptidyl peptidase-IV inhibitor use in hospitalized patients with diabetes. Ann Pharmacother 55:1326–1332. https://doi.org/10.1177/1060028021996337
Pintana H, Apaijai N, Chattipakorn N, Chattipakorn SC (2013) DPP-4 inhibitors improve cognition and brain mitochondrial function of insulin-resistant rats. J Endocrinol 218:1–11. https://doi.org/10.1530/joe-12-0521
Piwkowska A, Rogacka D, Jankowski M et al (2010) Metformin induces suppression of NAD(P)H oxidase activity in podocytes. Biochem Biophys Res Commun 393:268–273. https://doi.org/10.1016/j.bbrc.2010.01.119
Radak Z, Zhao Z, Koltai E, Ohno H, Atalay M (2013) Oxygen consumption and usage during physical exercise: the balance between oxidative stress and ROS-dependent adaptive signaling. Antioxid Redox Signal 18:1208–1246. https://doi.org/10.1089/ars.2011.4498
Ranganath LR (2008) Incretins: pathophysiological and therapeutic implications of glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1. J Clin Pathol 61:401–409. https://doi.org/10.1136/jcp.2006.043232
Roberts AC, Porter KE (2013) Cellular and molecular mechanisms of endothelial dysfunction in diabetes. Diab Vasc Dis Res 10:472–482. https://doi.org/10.1177/1479164113500680
Rohlenova K, Veys K, Miranda-Santos I, De Bock K, Carmeliet P (2018) Endothelial cell metabolism in health and disease. Trends Cell Biol 28:224–236. https://doi.org/10.1016/j.tcb.2017.10.010
Salmanoglu DS, Gurpinar T, Vural K et al (2016) Melatonin and L-carnitin improves endothelial disfunction and oxidative stress in type 2 diabetic rats. Redox Biol 8:199–204. https://doi.org/10.1016/j.redox.2015.11.007
Sena CM, Matafome P, Louro T et al (2011) Metformin restores endothelial function in aorta of diabetic rats. Br J Pharmacol 163:424–437. https://doi.org/10.1111/j.1476-5381.2011.01230.x
Sena CM, Nunes E, Louro T et al (2008) Effects of alpha-lipoic acid on endothelial function in aged diabetic and high-fat fed rats. Br J Pharmacol 153:894–906. https://doi.org/10.1038/sj.bjp.0707474
Sena CM, Pereira AM, Seiça R (2013) Endothelial dysfunction - a major mediator of diabetic vascular disease. Biochim Biophys Acta 1832:2216–2231. https://doi.org/10.1016/j.bbadis.2013.08.006
Shao Q, Meng L, Lee S et al (2019) Empagliflozin, a sodium glucose co-transporter-2 inhibitor, alleviates atrial remodeling and improves mitochondrial function in high-fat diet/streptozotocin-induced diabetic rats. Cardiovasc Diabetol 18:165. https://doi.org/10.1186/s12933-019-0964-4
Shenouda SM, Widlansky ME, Chen K et al (2011) Altered mitochondrial dynamics contributes to endothelial dysfunction in diabetes mellitus. Circulation 124:444–453. https://doi.org/10.1161/circulationaha.110.014506
Shi Y, Fan S, Wang D et al (2018) FOXO1 inhibition potentiates endothelial angiogenic functions in diabetes via suppression of ROCK1/Drp1-mediated mitochondrial fission. Biochim Biophys Acta Mol Basis Dis 1864:2481–2494. https://doi.org/10.1016/j.bbadis.2018.04.005
Sun D, Wang J, Toan S et al (2022) Molecular mechanisms of coronary microvascular endothelial dysfunction in diabetes mellitus: focus on mitochondrial quality surveillance. Angiogenesis 25:307–329. https://doi.org/10.1007/s10456-022-09835-8
Tang B L (2016). Sirt1 and the mitochondria. Mol Cells 39:87-95. 10.14348/molcells.2016.2318.
Teixeira-Lemos E, Nunes S, Teixeira F, Reis F (2011) Regular physical exercise training assists in preventing type 2 diabetes development: focus on its antioxidant and anti-inflammatory properties. Cardiovasc Diabetol 10:12. https://doi.org/10.1186/1475-2840-10-12
Teng X, Ji C, Zhong H et al (2019) Selective deletion of endothelial cell calpain in mice reduces diabetic cardiomyopathy by improving angiogenesis. Diabetologia 62:860–872. https://doi.org/10.1007/s00125-019-4828-y
Triggle CR, Ding H (2017) Metformin is not just an antihyperglycaemic drug but also has protective effects on the vascular endothelium. Acta Physiol (Oxf) 219:138–151. https://doi.org/10.1111/apha.12644
Tromp J, Ouwerkerk W, van Veldhuisen DJ et al (2022) A systematic review and network meta-analysis of pharmacological treatment of heart failure with reduced ejection fraction. JACC Heart Fail 10:73–84. https://doi.org/10.1016/j.jchf.2021.09.004
Vallon V (2011) The proximal tubule in the pathophysiology of the diabetic kidney. Am J Physiol Regul Integr Comp Physiol 300:R1009–R1022. https://doi.org/10.1152/ajpregu.00809.2010
van Ruiten CC, Smits MM, Kok MD et al (2022) Mechanisms underlying the blood pressure lowering effects of dapagliflozin, exenatide, and their combination in people with type 2 diabetes: a secondary analysis of a randomized trial. Cardiovasc Diabetol 21:63. https://doi.org/10.1186/s12933-022-01492-x
Venuraju SM, Lahiri A, Jeevarethinam A et al (2019) Duration of type 2 diabetes mellitus and systolic blood pressure as determinants of severity of coronary stenosis and adverse events in an asymptomatic diabetic population: PROCEED study. Cardiovasc Diabetol 18:51. https://doi.org/10.1186/s12933-019-0855-8
Vierck HB, Darvin ME, Lademann J et al (2012) The influence of endurance exercise on the antioxidative status of human skin. Eur J Appl Physiol 112:3361–3367. https://doi.org/10.1007/s00421-011-2296-2
Virgana R, Atik N, Gunadi JW et al (2022) MitoTEMPOL inhibits ROS-induced retinal vascularization pattern by modulating autophagy and apoptosis in rat-injected streptozotocin model. Life (Basel):12. https://doi.org/10.3390/life12071061
Von Schulze AT, Deng F, Morris JK, Geiger PC (1985) (2020). Heat therapy: possible benefits for cognitive function and the aging brain. J Appl Physiol 129:1468–1476. https://doi.org/10.1152/japplphysiol.00168.2020
Wada J, and Nakatsuka A (2016). Mitochondrial dynamics and mitochondrial dysfunction in diabetes. Acta Med Okayama 70:151-158. 10.18926/amo/54413.
Wang H, Wang AX, Aylor K, Barrett EJ (2013a) Nitric oxide directly promotes vascular endothelial insulin transport. Diabetes 62:4030–4042. https://doi.org/10.2337/db13-0627
Wang J, Toan S, Zhou H (2020a) Mitochondrial quality control in cardiac microvascular ischemia-reperfusion injury: new insights into the mechanisms and therapeutic potentials. Pharmacol Res 156:104771. https://doi.org/10.1016/j.phrs.2020.104771
Wang J, Toan S, Zhou H (2020b) New insights into the role of mitochondria in cardiac microvascular ischemia/reperfusion injury. Angiogenesis 23:299–314. https://doi.org/10.1007/s10456-020-09720-2
Wang J, Zhou H (2020) Mitochondrial quality control mechanisms as molecular targets in cardiac ischemia-reperfusion injury. Acta Pharm Sin B 10:1866–1879. https://doi.org/10.1016/j.apsb.2020.03.004
Wang M, Li Y, Li S, Lv J (2022) Endothelial dysfunction and diabetic cardiomyopathy. Front Endocrinol (Lausanne) 13:851941. https://doi.org/10.3389/fendo.2022.851941
Wang W, Wang Y, Long J et al (2012) Mitochondrial fission triggered by hyperglycemia is mediated by ROCK1 activation in podocytes and endothelial cells. Cell Metab 15:186–200. https://doi.org/10.1016/j.cmet.2012.01.009
Wang X, Zhang JQ, **u CK et al (2020c) Ginseng-Sanqi-Chuanxiong (GSC) extracts ameliorate diabetes-induced endothelial cell senescence through regulating mitophagy via the AMPK pathway. Oxid Med Cell Longev 2020:7151946. https://doi.org/10.1155/2020/7151946
Wang Y, Chun OK, Song WO (2013b) Plasma and dietary antioxidant status as cardiovascular disease risk factors: a review of human studies. Nutrients 5:2969–3004. https://doi.org/10.3390/nu5082969
Wang Y, Zhang X, Wang P et al (2019) Sirt3 overexpression alleviates hyperglycemia-induced vascular inflammation through regulating redox balance, cell survival, and AMPK-mediated mitochondrial homeostasis. J Recept Signal Transduct Res 39:341–349. https://doi.org/10.1080/10799893.2019.1684521
Weickert MO, Pfeiffer AFH (2018) Impact of dietary fiber consumption on insulin resistance and the prevention of type 2 diabetes. J Nutr 148:7–12. https://doi.org/10.1093/jn/nxx008
Wu H, Wang Y, Li W et al (2019) Deficiency of mitophagy receptor FUNDC1 impairs mitochondrial quality and aggravates dietary-induced obesity and metabolic syndrome. Autophagy 15:1882–1898. https://doi.org/10.1080/15548627.2019.1596482
Wu SL (2015) Staging of type 2 diabetes mellitus. Genet Mol Res 14:2118–2121. https://doi.org/10.4238/2015.March.20.22
Wu W, Xu H, Wang Z et al (2015) PINK1-Parkin-mediated mitophagy protects mitochondrial integrity and prevents metabolic stress-induced endothelial injury. PloS One 10:e0132499. https://doi.org/10.1371/journal.pone.0132499
** J, Rong Y, Zhao Z et al (2021) Scutellarin ameliorates high glucose-induced vascular endothelial cells injury by activating PINK1/Parkin-mediated mitophagy. J Ethnopharmacol 271:113855. https://doi.org/10.1016/j.jep.2021.113855
**ang J, Zhang C, Di T et al (2022) Salvianolic acid B alleviates diabetic endothelial and mitochondrial dysfunction by down-regulating apoptosis and mitophagy of endothelial cells. Bioengineered 13:3486–3502. https://doi.org/10.1080/21655979.2022.2026552
**ng H, Zhang Z, Shi G et al (2021) Chronic inhibition of mROS protects against coronary endothelial dysfunction in mice with diabetes. Front Cell Dev Biol 9:643810. https://doi.org/10.3389/fcell.2021.643810
Xu HZ, Le YZ (2011) Significance of outer blood-retina barrier breakdown in diabetes and ischemia. Invest Ophthalmol Vis Sci 52:2160–2164. https://doi.org/10.1167/iovs.10-6518
Yang MY, Fan Z, Zhang Z, Fan J (2021) MitoQ protects against high glucose-induced brain microvascular endothelial cells injury via the Nrf2/HO-1 pathway. J Pharmacol Sci 145:105–114. https://doi.org/10.1016/j.jphs.2020.10.007
Yoshibayashi M, Kume S, Yasuda-Yamahara M et al (2020) Protective role of podocyte autophagy against glomerular endothelial dysfunction in diabetes. Biochem Biophys Res Commun 525:319–325. https://doi.org/10.1016/j.bbrc.2020.02.088
Yoshida T, Okuno A, Tanaka J et al (2009) Metformin primarily decreases plasma glucose not by gluconeogenesis suppression but by activating glucose utilization in a non-obese type 2 diabetes Goto-Kakizaki rats. Eur J Pharmacol 623:141–147. https://doi.org/10.1016/j.ejphar.2009.09.003
Zhang H, Shen Y, Kim IM, Weintraub NL, Tang Y (2021a) The impaired bioenergetics of diabetic cardiac microvascular endothelial cells. Front Endocrinol (Lausanne) 12:642857. https://doi.org/10.3389/fendo.2021.642857
Zhang J (2022). Biomarkers of endothelial activation and dysfunction in cardiovascular diseases. Rev Cardiovasc Med 23:73. 10.31083/j.rcm2302073.
Zhang MY, Zhu L, Bao X et al (2022) Inhibition of Drp1 ameliorates diabetic retinopathy by regulating mitochondrial homeostasis. Exp Eye Res 220:109095. https://doi.org/10.1016/j.exer.2022.109095
Zhang MY, Zhu L, Zheng X et al (2021b) TGR5 activation ameliorates mitochondrial homeostasis via regulating the PKCδ/Drp1-HK2 signaling in diabetic retinopathy. Front Cell Dev Biol 9:759421. https://doi.org/10.3389/fcell.2021.759421
Zhang Y, Zhou H, Wu W et al (2016) Liraglutide protects cardiac microvascular endothelial cells against hypoxia/reoxygenation injury through the suppression of the SR-Ca(2+)-XO-ROS axis via activation of the GLP-1R/PI3K/Akt/survivin pathways. Free Radic Biol Med 95:278–292. https://doi.org/10.1016/j.freeradbiomed.2016.03.035
Zheng H, Zhu H, Liu X et al (2021) Mitophagy in diabetic cardiomyopathy: roles and mechanisms. Front Cell Dev Biol 9:750382. https://doi.org/10.3389/fcell.2021.750382
Zhou H, Ren J, Toan S, Mui D (2021) Role of mitochondrial quality surveillance in myocardial infarction: from bench to bedside. Ageing Res Rev 66:101250. https://doi.org/10.1016/j.arr.2020.101250
Zhou H, Toan S (2020) Pathological roles of mitochondrial oxidative stress and mitochondrial dynamics in cardiac microvascular ischemia/reperfusion injury. Biomolecules 10. https://doi.org/10.3390/biom10010085
Zhou H, Wang S, Zhu P et al (2018) Empagliflozin rescues diabetic myocardial microvascular injury via AMPK-mediated inhibition of mitochondrial fission. Redox Biol 15:335–346. https://doi.org/10.1016/j.redox.2017.12.019
Zhu H, Toan S, Mui D, Zhou H (2021) Mitochondrial quality surveillance as a therapeutic target in myocardial infarction. Acta Physiol (Oxf) 231:e13590. https://doi.org/10.1111/apha.13590
Zhu W, Yuan Y, Liao G et al (2018) Mesenchymal stem cells ameliorate hyperglycemia-induced endothelial injury through modulation of mitophagy. Cell Death Dis 9:837. https://doi.org/10.1038/s41419-018-0861-x
Zou R, Shi W, Qiu J et al (2022) Empagliflozin attenuates cardiac microvascular ischemia/reperfusion injury through improving mitochondrial homeostasis. Cardiovasc Diabetol 21:106. https://doi.org/10.1186/s12933-022-01532-6
Funding
This study was supported by the Chinese Academy of Chinese Medical Sciences Doctoral Talents Training Fund (grant number: 2021) and Special Program for Training Outstanding Young Talents of Chinese Academy of Traditional Chinese Medicine (ZZ16-YQ-021). Innovative cultivation project of Guang'anmen Hospital, Chinese Academy of Chinese Medical Sciences (2022s481). The funders had no role in paper design, data collection, data analysis, interpretation, and writing of the paper.
Author information
Authors and Affiliations
Contributions
Tong Wang, **nwei Wang, Tong Fu, Yanchun Ma, Qi Wang, and Shuxiang Zhang collected the data and related references. **ao Zhang, Hao Zhou, **ng Chang, and Ying Tong contributed to study concept and design, supervision of the study, literature search, and writing of the manuscript, obtained funding, and revised the final version of manuscript. All the authors read the article and approved the submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, T., Wang, X., Fu, T. et al. Roles of mitochondrial dynamics and mitophagy in diabetic myocardial microvascular injury. Cell Stress and Chaperones 28, 675–688 (2023). https://doi.org/10.1007/s12192-023-01384-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-023-01384-3