Abstract
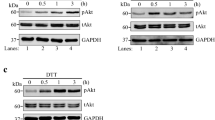
The endoplasmic reticulum (ER) has the ability to signal organelle dysfunction via a complex signaling network known as the unfolded protein response (UPR). In this work, hamster fibroblast cells exhibiting moderate levels of ER stress were compared to those exhibiting severe ER stress. Inhibition of N-linked glycosylation was accomplished via a temperature-sensitive mutation in the Dad1 subunit of the oligosaccharyltransferase (OST) complex or by direct inhibition with tunicamycin (Tm). Temperature shift (TS) treatment generated weak activation of ER stress signaling when compared to doses of Tm that are typically used in ER stress studies (500–1000 nM). A dose-response analysis of key ER stress signaling mediators, inositol-requiring enzyme 1 (IRE1) and protein kinase R (PKR)-like endoplasmic reticulum kinase (PERK), revealed 20–40 nM of Tm to generate activation intensity similar to TS treatment. In parental BHK21 cells, moderate (20–40 nM) and high doses (200–1000 nM) of Tm were compared to identify physiological and signaling-based differences in stress response. Inhibition of ER Ca2+ release via ITPR activity with 2-aminoethoxydiphenyl borate (2-APB) or Xestospongin C (XeC) was sufficient to protect against apoptosis induced by moderate but not higher doses of Tm. Analysis of kinase activation over a range of Tm exposures revealed the p38 stress-activated protein kinase (SAPK) to display increasing activation with Tm dosage. Interestingly, Tm induced the extracellular regulated kinases (Erk1/2) only at moderate doses of Tm. Inhibition of ER transmembrane stress sensors (IRE1, PERK) or cytosolic signaling mediators (p38, Jnk1, Erk1/2) was used to evaluate pathways involved in apoptosis activation during ER stress. Inhibition of either PERK or p38 was sufficient to reduce cell death and apoptosis induced by moderate, but not high, doses of Tm. During ER stress, cells exhibited a rapid decline in anti-apoptotic Mcl-1 and survivin proteins. Inhibition of PERK was sufficient to block this affect. This work reveals moderate doses of ER stress to generate patterns of stress signaling that are distinct from higher doses and that apoptosis activation at moderate levels of stress are dependent upon PERK and p38 signaling. Studies exploring ER stress signaling should recognize that this signaling acts as a rheostat rather than a simple switch, behaving distinctively in a dose-dependent manner.







Similar content being viewed by others
Abbreviations
- ER:
-
Endoplasmic reticulum
- Tm:
-
Tunicamycin
- TS:
-
Temperature shift
- 2-APB:
-
2-Aminoethoxyphenylborate
- OST:
-
Oligosaccharyltransferase
- XeC:
-
Xestospongin C
- IRE1:
-
Inositol-requiring enzyme 1
- PERK:
-
Protein kinase R (PKR)-like endoplasmic reticulum kinase
- Xbp1:
-
X-box binding protein 1
- ITPR:
-
Inositol 1,4,5-trisphosphate receptor
- RT/PCR:
-
Reverse transcription, polymerase chain reaction
- UPR:
-
Unfolded protein response
References
Arai K et al (2004) Involvement of ERK MAP kinase in endoplasmic reticulum stress in SH-SY5Y human neuroblastoma cells. J Neurochem 89(1):232–239
Ascher-Landsberg J et al (1999) The effects of 2-aminoethoxydiphenyl borate, a novel inositol 1,4, 5-trisphosphate receptor modulator on myometrial contractions. Biochem Biophys Res Commun 264(3):979–982
Ashwell JD (2006) The many paths to p38 mitogen-activated protein kinase activation in the immune system. Nat Rev Immunol 6(7):532–540
Blackshaw S et al (2000) Type 3 inositol 1,4,5-trisphosphate receptor modulates cell death. FASEB J 14(10):1375–1379
Boehning D et al (2003) Cytochrome c binds to inositol (1,4,5) trisphosphate receptors, amplifying calcium-dependent apoptosis. Nat Cell Biol 5(12):1051–1061
Boyce M et al (2005) A selective inhibitor of eIF2alpha dephosphorylation protects cells from ER stress. Science 307(5711):935–939
Brewster JL et al (2000) Deletion of Dad1 in mice induces an apoptosis-associated embryonic lethality. Genesis 26:271–278
Cnop M et al (2007) Selective inhibition of eukaryotic translation initiation factor 2 alpha dephosphorylation potentiates fatty acid-induced endoplasmic reticulum stress and causes pancreatic beta-cell dysfunction and apoptosis. J Biol Chem 282(6):3989–3997
Czabotar PE et al (2014) Control of apoptosis by the BCL-2 protein family: implications for physiology and therapy. Nat Rev Mol Cell Biol 15(1):49–63
Gardner BM, Walter P (2011) Unfolded proteins are Ire1-activating ligands that directly induce the unfolded protein response. Science 333(6051):1891–1894
Gardner BM et al (2013) Endoplasmic reticulum stress sensing in the unfolded protein response. Cold Spring Harb Perspect Biol 5(3):a013169
Ghosh S et al (2015) Cross-talk between endoplasmic reticulum (ER) stress and the MEK/ERK pathway potentiates apoptosis in human triple negative breast carcinoma cells: role of a dihydropyrimidone, nifetepimine. J Biol Chem 290(7):3936–3949
Hamanaka RB et al (2009) PERK-dependent regulation of IAP translation during ER stress. Oncogene 28(6):910–920
Hassler J, Cao SS, Kaufman RJ (2012) IRE1, a double-edged sword in pre-miRNA slicing and cell death. Dev Cell 23(5):921–923
Hetz C (2012) The unfolded protein response: controlling cell fate decisions under ER stress and beyond. Nat Rev Mol Cell Biol 13(2):89–102
Higo T et al (2010) Mechanism of ER stress-induced brain damage by IP(3) receptor. Neuron 68(5):865–878
Hiramatsu N et al (2014) Translational and posttranslational regulation of XIAP by eIF2alpha and ATF4 promotes ER stress-induced cell death during the unfolded protein response. Mol Biol Cell 25(9):1411–1420
Hiramatsu N et al (2015) Multiple mechanisms of unfolded protein response-induced cell death. Am J Pathol 185:1800–1808
Hong NA et al (2000) Mice lacking Dad1, the defender against apoptotic death-1, express abnormal N-linked glycoproteins and undergo increased embryonic apoptosis. Dev Biol 220(1):76–84
Hoppins S, Nunnari J (2012) Cell biology. Mitochondrial dynamics and apoptosis—the ER connection. Science 337(6098):1052–1054
Jiang CC et al (2007) Inhibition of MEK sensitizes human melanoma cells to endoplasmic reticulum stress-induced apoptosis. Cancer Res 67(20):9750–9761
Jiang Q et al (2014) Involvement of p38 in signal switching from autophagy to apoptosis via the PERK/eIF2alpha/ATF4 axis in selenite-treated NB4 cells. Cell Death Dis 5:e1270
Joseph SK, Hajnoczky G (2007) IP3 receptors in cell survival and apoptosis: Ca2+ release and beyond. Apoptosis 12(5):951–968
Kessel D (2006) Protection of Bcl-2 by salubrinal. Biochem Biophys Res Commun 346(4):1320–1323
Kim L et al (2005) p38 MAPK autophosphorylation drives macrophage IL-12 production during intracellular infection. J Immunol 174(7):4178–4184
Lee AS (2005) The ER chaperone and signaling regulator GRP78/BiP as a monitor of endoplasmic reticulum stress. Methods 35(4):373–381
Lim EJ, Heo J, Kim YH (2015) Tunicamycin promotes apoptosis in leukemia cells through ROS generation and downregulation of survivin expression. Apoptosis 20(8):1087–1098
Liu Z et al (2015) Protein kinase R-like ER kinase and its role in endoplasmic reticulum stress-decided cell fate. Cell Death Dis 6:e1822
Manni S et al (2012) Protein kinase CK2 protects multiple myeloma cells from ER stress-induced apoptosis and from the cytotoxic effect of HSP90 inhibition through regulation of the unfolded protein response. Clin Cancer Res 18(7):1888–1900
Maurel M et al (2014) Getting RIDD of RNA: IRE1 in cell fate regulation. Trends Biochem Sci 39(5):245–254
Miyamoto S et al (2000) Xestospongin C, a selective and membrane-permeable inhibitor of IP(3) receptor, attenuates the positive inotropic effect of alpha-adrenergic stimulation in Guinea-pig papillary muscle. Br J Pharmacol 130(3):650–654
Nakashima T et al (1993) Molecular cloning of a human cDNA encoding a novel protein, DAD1, whose defect causes apoptotic cell death in hamster BHK21 cells. Mol Cell Biol 13(10):6367–6374
Niederer KE et al (2005) Cypermethrin blocks a mitochondria-dependent apoptotic signal initiated by deficient N-linked glycosylation within the endoplasmic reticulum. Cell Signal 17:177–186
Nikonov AV et al (2002) Active translocon complexes labeled with GFP-Dad1 diffuse slowly as large polysome arrays in the endoplasmic reticulum. J Cell Biol 158(3):497–506
Opferman JT (2015) Attacking cancer’s Achilles heel: antagonism of anti-apoptotic BCL-2 family members. FEBS J. doi:10.1111/febs.13472
Ota A et al (2010) Specific regulation of noncanonical p38alpha activation by Hsp90-Cdc37 chaperone complex in cardiomyocyte. Circ Res 106(8):1404–1412
Ruggiano A, Foresti O, Carvalho P (2014) Quality control: ER-associated degradation: protein quality control and beyond. J Cell Biol 204(6):869–879
Rutkowski DT et al (2006) Adaptation to ER stress is mediated by differential stabilities of pro-survival and pro-apoptotic mRNAs and proteins. PLoS Biol 4(11):e374
Saleem H et al (2014) Interactions of antagonists with subtypes of inositol 1,4,5-trisphosphate (IP3) receptor. Br J Pharmacol 171(13):3298–3312
Sanjay A, Fu J, Kreibich G (1998) DAD1 is required for the function and the structural integrity of the oligosaccharyltransferase complex. J Biol Chem 273(40):26094–26099
Schroder M (2008) Endoplasmic reticulum stress responses. Cell Mol Life Sci 65(6):862–894
Sohn J et al (2003) The effect of ursodeoxycholic acid on the survivin in thapsigargin-induced apoptosis. Cancer Lett 191(1):83–92
Upton JP et al (2012) IRE1alpha cleaves select microRNAs during ER stress to derepress translation of proapoptotic caspase-2. Science 338(6108):818–822
Walter P, Ron D (2011) The unfolded protein response: from stress pathway to homeostatic regulation. Science 334(6059):1081–1086
Wang X et al (2011) Bcl-2 proteins regulate ER membrane permeability to luminal proteins during ER stress-induced apoptosis. Cell Death Differ 18(1):38–47
Wek RC, Jiang HY, Anthony TG (2006) Co** with stress: eIF2 kinases and translational control. Biochem Soc Trans 34(Pt 1):7–11
Wu L et al (2014) Salubrinal protects against rotenone-induced SH-SY5Y cell death via ATF4-parkin pathway. Brain Res 1549:52–62
Zhu X et al (2014) Ubiquitination of inositol-requiring enzyme 1 (IRE1) by the E3 ligase CHIP mediates the IRE1/TRAF2/JNK pathway. J Biol Chem 289(44):30567–30577
Zong WX et al (2003) Bax and Bak can localize to the endoplasmic reticulum to initiate apoptosis. J Cell Biol 162(1):59–69
Acknowledgments
The authors wish to recognize the helpful contributions of research students Celina Jones, Emily Blair, and Tina Johnson. This work was supported by grants from the National Institutes of Health (R15GM065139), the National Science Foundation (DBI-1062721), and research funding from Pepperdine University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lumley, E.C., Osborn, A.R., Scott, J.E. et al. Moderate endoplasmic reticulum stress activates a PERK and p38-dependent apoptosis. Cell Stress and Chaperones 22, 43–54 (2017). https://doi.org/10.1007/s12192-016-0740-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-016-0740-2