Abstract
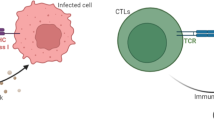
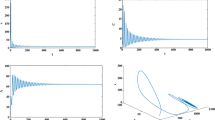
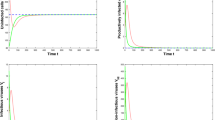
HIV (human immunodeficiency virus) is a dangerous virus that constantly diminishes an individual’s immune system by explicitly targeting CD4 cells, which are the body’s key protectors against disease by obstructing them to make duplicates of themselves. If left untreated, it can lead to AIDS (acquired immunodeficiency syndrome). The Treatment involves antiretroviral therapy which maintains the immunity to a specific level and thus helps in suppressing the virus replication. This paper emphasizes on the development of the model involving healthy and infected population, virus population, antibodies and CTL cells. The investigation encapsulates the local stability based on thresholds followed by the local bifurcation analysis based on \(\beta _1\) and \(R_0\). The global stability analysis is done the using Graph- theoretic approach. Further the optimal control problem is discussed using Linear feedback control method which aims to reduce the viral load by kee** antibodies to a certain level. Numerical discussion includes surface plots of the thresholds based on various parameters, graphs showing the comparison between without control and with control especially for the virus population, infected population and antibodies which are our target. Finally, we have also shown the curve-fitting for our data using optimized Nedlar-Mean algorithm.



















Similar content being viewed by others
References
Aditya, N.M., Puneet, S.K., Kshitija, H.K.: Simple linear regression with least square estimation: an overview. Int. J. Comput. Sci. Inf. Technol. 7, 2394–2396 (2016)
Allali, K., Meskaf, A., Tridane, A.: Mathematical modeling of the adaptive immune responses in the early stage of the HBV infection. Int. J. Differ. Equ. 2018, 28 (2018)
Allali, K., Tabit, Y., Harroudi, S.: On HIV model with sdaptive immune response, two saturated rates and therapy. Math. Model. Nat. Phenom. 12, 1–14 (2017)
Amalesh, M.K., Dasi, A.: Bifurcation analysis of an SIR model. Commun. Appl. Anal. 20, 317–323 (2016)
Arnaout, R.A., Wodarz, D.: HIV-1 dynamics revisited: biphasic decay by cytotoxic T lymphocyte killing. Proc. R. Soc. Lond. B Biol. Sci. 267, 1347–1354 (2000)
Bakare, E.A., Nwagwo, A., Danso-Addo, E.: Optimal control analysis of an SIR epidemic model with constant recruitment, International Journal of Applied. Math. Res. 3, 273–285 (2014)
Barati, R.: Parameter estimation of nonlinear Muskingum models using Nelder-Mead simplex algorithm. J. Hydrol. Eng. 16, 946–954 (2011)
Barik, M., Chauhan, S., Bhatia, S.K., Misra, O.P.: Understanding role of CTL cells and antibodies on a delayed HIV mathematical model: a dynamical analysis. Math. Eng. Sci. Aerosp. 12, 109–123 (2021)
Barik, M., Swarup, C., Singh, T., HAbbi, S., Chauhan, S.: Dynamical analysis, optimal control and spatial pattern in an influenza model with adaptive immunity in two stratified population. AIMS Math. 7, 4898–4935 (2022)
Burg, D., Rong, L., Neumann, A.U., Dahari, H.: Mathematical modeling of viral kinetics under immune control during primary HIV-1 infection. J. Theor. Biol. 259, 751–759 (2009)
Ciupe, S.M., Riberio, R.M., Nelson, P.W., Perelson, A.S.: Modelling the mechanism of acute hepatitis B virus infection. J. Theor. Biol. 247, 23–35 (2009)
Ciupe, S.M., Riberio, R.M., Nelson, P.W., Dusheiko, G., Perelson, A.S.: The role of cells refractory to productive infection in acute hepatitis B viral dynamics. Proc United States of America 104, 5050–5057 (2007)
Ciupe, S.M., Riberio, R.M., Perelson, A.S.: Antibody responses during hepatitis B viral infection. PLoS Comput. Biol. 10, 1–16 (2014)
Cohen, Y.Z., Caskey, M.: Broadly neutralizing antibodies for treatment and prevention of HIV-1 infection. Curr. Opin. Biotechnol. 13, 366–373 (2018)
Davenport, M.P., Ribeiro, R.M., Zhang, L., Wilson, D.P., Perelson, A.S.: Understanding the mechanisms and limitations of immune control of HIV. Immunol. Rev. 216, 164–175 (2007)
David, M., Margolis, D.M., Koup, R.A., Ferrari, G.: HIV antibodies for treatment of HIV infection. Immunol. Rev. 275, 313–323 (2017)
De Boer, R.J., Perelson, A.S.: Target cell limited and immune control models of HIV infection: a comparison. J. Theor. Biol. 190, 201–214 (1998)
Gaff, H., Schaefer, E.: Optimal control applied to vaccination and treatment strategies for various epidemiological models. Math. Biosci. Eng. 6, 469–492 (2009)
Global HIV Statistics, Communications and Global Advocacy, (2016) Fact sheet—UNAIDS https://www.unaids.org
Gourley, S.A., Kaung, Y., Nagy, J.D.: Dynamics of a delay differential equation model of hepatitis B virus infection. J. Biol. Dyn. 2, 140–153 (2008)
Ghosh, B., Kar, T.K.: Sustainable use of prey species in a prey-predator system: jointly determined ecological thresholds and economic trade-offs. Ecol. Model. 272, 49–58 (2014)
Gulati, P., Chauhan, S., Mubayi, A., Singh, T., Rana, P.: Dynamical analysis, optimum control and pattern formation in the biological pest(EFSB) control model. Chaos Solitons Fractals 147, 110920 (2021)
Guo, H., Li, M.Y., Shuai, Z.: A graph-theoretic approach to the method of global Lyapunov functions. Proc. Am. Math. Soci. 136, 2793–2802 (2008)
Hamilton, F.: Parameter estimation in differential equations: a numerical study of shooting methods. SIAM Undergrad. Res. Online 4, 1 (2011)
Hews, S., Eikenberry, S., Nagy, J.D., Kaung, Y.: Rich dynamics of a hepatitis B viral infection model with logistic hepatocyte growth. J. Math. Biol. 60, 573–590 (2010)
Kar, T.K., Ghosh, B.: Sustainability and optimal control of an exploited prey predator system through provision of alternative food to predator. Biosystems 109, 220–232 (2012)
Li, M.Y., Shuai, Z.: Global-stability problem for coupled systems of differential equations on networks. J. Differ. Eq. 248, 1–20 (2010)
Mahardika, R., Sumanto, Y.D.: Routh-hurwitz criterion and bifurcation method for stability analysis of tuberculosis transmission model. J. Phys. Conf. Series 1217, 012056 (2019)
Mbogo, W.R., Luboobi, L.S., Odhiambo, J.: Stochastic model for in-host HIV dynamics with therapeutic intervention. ISRN Biomath. 2013, 1–11 (2013)
Michalopoulos, G.K., DeFrances, D.C.: Liver regeneration. Science 276, 60–65 (1997)
Morisa, A., Pereiraa, M., Chakrabarti, L.: A role for antibodies in natural HIV control. Curr. Opin. Biotechnol. 14, 265–272 (2019)
Nagumo, M.: Uber die Lage der Integralkurven gew onlicher differentialgleichungen. Proc. Phys. Math. Soc. Jpn. 24, 551–559 (1942)
Nampala, H., Luboobi, L.S., Mugisha, J.Y., Obua, C.: Mathematical modeling of liver enzyme elevation in HIV mono-infection. Mathematical Biosciences 242, 77–85 (2013)
Nowak, M.A., May, R.M.: Virus Dynamics: Mathematical Principles of Immunology and Virology. Oxford University Press, Oxford (2000)
Overview:Data & Trends:Global:HIV Statistics, HIV.gov(2020)
Ogunlaran, O.M., OukouomiNoutchie, S.C.: Mathematical model for an effective management of HIV infection, BioMed Research International, 2016 (2016)
Perko, L.: Differential Equations and Dynamical Systems. International Journal for Theory, Real World Modelling and Simulations, 20, 3rd edn. New York, NY, USA (2001)
Rafiko, M.: Mathematical modelling of the biological pest control of the sugarcane borer. Int. J. Comput. Math. 89, 390–401 (2012)
Ramsay, J.O., Hooker, G., Campbell, D., Cao, J.: Parameter estimation for differential equations: a generalized smoothing approach, series B statistical methodology. J. Royal Stat. Soc. 69, 741–796 (2007)
Shuai, Z., Driessche, V.D.: Global stability of infectious disease models using Lyapunov functions. SIAM J. Appl. Math. 73, 1513–1532 (2013)
Wang, Y., Zhou, Y., Brauer, F., Heffernan, J.M.: Viral dynamics model with CTL immune response incorporating antiretroviral therapy. J. Math. Biol. 67, 901–934 (2013)
Whalley, S.A., Murray, J.M., Brown, D.: Kinetics of acute hepatitis b virus infection in humans. J. Exp. Med. 193, 847–853 (2001)
Wilson, D.P., Law, M.G., Grulich, A.E., Cooper, D.A., Kaldor, J.M.: Relation between HIV viral load and infectiousness: a model-based analysis. Lancet 372, 314–320 (2008)
Yusuf, T.T., Benyah, F.: Optimal strategy for controlling the spread of HIV/AIDS disease: a case study of South Africa. J. Biol. Dyn. 6, 475–494 (2012)
Zhou, X., Shi, X., Zhang, Z., Song, X.: Dynamical behavior of a virus dynamics model with CTL immune response. Appl. Math. Comput. 213, 329–347 (2009)
Author information
Authors and Affiliations
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Barik, M., Chauhan, S., Misra, O.P. et al. Optimal control using linear feedback control and neutralizing antibodies for an HIV model with dynamical analysis. J. Appl. Math. Comput. 68, 4361–4389 (2022). https://doi.org/10.1007/s12190-022-01710-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12190-022-01710-5
Keywords
- Immune response
- Local stability
- Graph theoretic approach
- Local bifurcation analysis
- Linear feedback control
- Curve-fit