Abstract
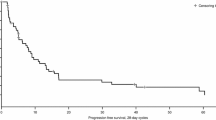
Phase I study was conducted to evaluate the safety, pharmacokinetics (PK) and efficacy of the oral mammalian target of rapamycin inhibitor, everolimus (RAD001), in patients with relapsed or refractory non-Hodgkin lymphoma (NHL). Patients received everolimus 5 or 10 mg orally once daily. Dose escalation was based on the safety assessment and the probability of dose-limiting toxicities (DLTs) using a Bayesian logistic model. DLTs were evaluated in six patients at each dose level during the initial 28 days of study treatment. A total of 13 patients were enrolled; 5 mg (seven) and 10 mg (six). No DLTs were observed at either dose level. Frequently observed potentially drug-related adverse events included leukopenia (8/13), thrombocytopenia (8/13), elevated hepatic transaminase (9/13), stomatitis (7/13), anemia (6/13), and nasopharyngitis (6/13). All adverse events were reversible. Non-infectious pneumonitis (grade 1) in one patient resolved following discontinuation of everolimus. Two patients with diffuse large B cell lymphoma and two patients with follicular lymphomas achieved objective responses with an overall response rate of 31% (4/13). The pharmacokinetic profiles were not different from those in non-Japanese patients. Everolimus was well tolerated at doses up to 10 mg/day and showed potential efficacy in relapsed or refractory NHL, warranting further investigation.



Similar content being viewed by others
References
Wullschleger S, Loewith R, Hall MN. TOR signaling in growth and metabolism. Cell. 2006;124:471–84.
Patel PH, Chadalavada RS, Chaganti RS, Motzer RJ. Targeting von Hippel-Lindau pathway in renal cell carcinoma. Clin Cancer Res. 2006;12:7215–20.
Hay N, Sonenberg N. Upstream and downstream of mTOR. Genes Dev. 2006;18:1926–45.
Uddin S, Hussain AR, Siraj AK, et al. Role of phosphatidylinositol 3′-kinase/AKT pathway in diffuse large B-cell lymphoma survival. Blood. 2006;108:4178–86.
Leseux L, Hamdi SM, Saati T, et al. Syk-dependent mTOR activation in follicular lymphoma cells. Blood. 2006;108:4156–62.
Vega F, Medeiros J, Leventaki V, et al. Activation of mammalian target of rapamycin signaling pathway contributes to tumor cell survival in anaplastic lymphoma kinase–positive anaplastic large cell lymphoma. Cancer Res. 2006;66:6589–97.
Bertoni F, Zucca E, Cotter FE. Molecular basis of mantle cell lymphoma. Br J Haematol. 2004;124:130–40.
Costa LJ. Aspects of mTOR biology and the use of mTOR inhibitors in non-Hodgkin’s lymphoma. Cancer Treat Rev. 2007;33:78–84.
Ruan J, Hajjar K, Rafii S, et al. Angiogenesis and antiangiogenic therapy in non-Hodgkin’s lymphoma. Ann Oncol. 2009;20:413–24.
Bertolini F, Paolucci M, Peccatori F, et al. Angiogenic growth factors and endostatin in non-Hodgkin’s lymphoma. Br J Haematol. 1999;106:504–9.
Faivre S, Kroemer G, Raymond E, et al. Current development of mTOR inhibitors as anticancer agents. Nat Rev Drug Discov. 2006;5:671–88.
Witzig TE, Geyer SM, Ghobrial I, et al. Phase II trial of single-agent temsirolimus (CCI-779) for relapsed mantle cell lymphoma. J Clin Oncol. 2005;23:5347–56.
Hess G, Herbrecht R, Romaguera J, et al. Phase III study to evaluate temsirolimus compared with investigator’s choice therapy for the treatment of relapsed or refractory mantle cell lymphoma. J Clin Oncol. 2009;27:3822–9.
Witzig T, Habermann T, Reeder C, et al. A phase II trial of the oral mTOR inhibitor everolimus in relapsed non-Hodgkin lymphoma and Hodgkin disease (HD). Haematologica. 2009;94(Suppl. 2):436. Abstract 1081.
Johnston PB, Ansell SM, Colgan JP, et al. Phase II trial of the oral mTOR inhibitor everolimus (RAD001) for patients with relapsed or refractory lymphoma. J Clin Oncol. 2007; 25 18S June 20 Suppl:8055 (abstract).
Okamoto I, Doi T, Ohtsu A, et al. Phase I clinical and pharmacokinetic study of RAD001 (everolimus) administered daily to Japanese patients with advanced solid tumors. Jpn J Clin Oncol. 2010;40:17–23.
Neuenschwander B, Branson M, Gsponer T. Critical aspects of the Bayesian approach to phase I cancer trials. Stat Med. 2008;27:2420–39.
O’Donnell A, Faivre S, Burris HA III, et al. Phase I pharmacokinetic and pharmacodynamic study of the oral mammalian target of rapamycin inhibitor everolimus in patients with advanced solid tumors. J Clin Oncol. 2008;26:1588–95.
Cheson BD, Horning SJ, Coiffier B, et al. Report of an international workshop to standardize response criteria for non-Hodgkin’s lymphomas. J Clin Oncol. 1999;17:1244–53.
Tabernero J, Rojo F, Calvo E, et al. Dose- and schedule-dependent inhibition of the mammalian target of rapamycin pathway with everolimus: a phase I tumor pharmacodynamic study in patients with advanced solid tumors. J Clin Oncol. 2008;26:1603–10.
Yee KWL, Zeng Z, Konopleva M, et al. Phase I/II study of the mammalian target of rapamycin inhibitor everolimus (RAD001) in patients with relapsed or refractory hematologic malignancies. Clin Cancer Res. 2006;12:5165–73.
Rizzieri DA, Feldman E, DiPersio JF, et al. A phase 2 clinical trial of deforolimus (AP23573, MK-8669), a novel mammalian target of rapamycin inhibitor, in patients with relapsed or refractory hematologic malignancies. Clin Cancer Res. 2008;14:2756–62.
Duran I, Siu LL, Oza AM, et al. Characterisation of the lung toxicity of the cell cycle inhibitor temsirolimus. Eur J Cancer. 2006;42:1875–80.
Motzer RJ, Escudier B, Oudard S, et al. Phase 3 trial of everolimus for metastatic renal cell carcinoma : final results and analysis of prognostic factors. Cancer. 2010 Jun 14 [Epub ahead of print].
Johnston PB, Inwards DJ, Colgan JP, et al. A phase II trial of the oral mTOR inhibitor everolimus in relapsed Hodgkin lymphoma. Am J Hematol. 2010;85:320–4.
Ghobrial IM, Gertz M, Laplant B, et al. Phase II trial of the oral mammalian target of rapamycin inhibitor everolimus in relapsed or refractory Waldenström macroglobulinemia. J Clin Oncol. 2010;28:1408–14.
Zent CS, LaPlant BR, Johnston PB, et al. The treatment of recurrent/refractory chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL) with everolimus results in clinical responses and mobilization of CLL cells into the circulation. Cancer. 2010;116:2201–7.
Acknowledgments
The authors thank the patients, doctors, nurses, and staff members who participated in this clinical trial for their excellent cooperation. They also thank K. Hatake, MD (Cancer Institute Hospital of Japanese Foundation for Cancer Research), H. Minami, MD (Kobe University), and N. Usui, MD (Jikei University) as members of the Independent Data Safety and Monitoring Committee, and M. Itoh and K. Kurei (Novartis Pharma K.K.). This study was supported by Novartis Pharmaceuticals Corporation. We thank Scientific Connexions for providing assistance with medical writing and editing. These services were funded by Novartis Pharmaceutical Corporation.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Tobinai, K., Ogura, M., Maruyama, D. et al. Phase I study of the oral mammalian target of rapamycin inhibitor everolimus (RAD001) in Japanese patients with relapsed or refractory non-Hodgkin lymphoma. Int J Hematol 92, 563–570 (2010). https://doi.org/10.1007/s12185-010-0707-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-010-0707-5