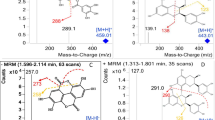
Abstract
This work establishes an effective detection method for simultaneous determination of eight phenolic acid compounds in rapeseed by combing accelerated solvent extraction (ASE) and solid-phase extraction (SPE) with ultra-high-performance liquid chromatography-tandem mass spectrometry (UHPLC-MS/MS). The effects of different extraction solvents, extraction temperature, static extraction time, and the strength and volume of the eluent on the extraction of phenolic acid compounds are discussed. Experimental results showed that the best extraction efficiency was achieved with a methanol aqueous solution (V/V = 80:20) as the extractant, extraction temperature of 100 °C, static extraction time of 6 min, methanol:ethyl acetate eluent (V/V = 80:20), and an elution volume of 8 mL. Under the optimized experimental conditions, the recoveries of the eight target compounds ranged from 89.4 to 105.2%, the relative standard deviations ranged from 1.6 to 4.9%, and the limits of detection and quantification ranged from 0.006 to 1.58 mg/kg and 0.024 to 4.28 mg/kg. The results show that the method has the advantages of good reproducibility, accurate and reliable analysis results, and is suitable for determining phenolic acids in rapeseed.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aladedunye F, Matthäus B (2014) Phenolic extracts from Sorbus aucuparia (L.) and Malus baccata (L.) berries: antioxidant activity and performance in rapeseed oil during frying and storage. Food Chem 159:273–281. https://doi.org/10.1016/j.foodchem.2014.02.139
Cui J, **ao Yb, Dai L, Zhao XH, Wang Y (2013) Speciation of organoarsenic species in food of animal origin using accelerated solvent extraction (ASE) with determination by HPLC-hydride generation-atomic fluorescence spectrometry (HG-AFS). Food Anal Methods 6:370–379. https://doi.org/10.1007/s12161-012-9533-0
Heredia-Sandoval NG, Granados-Nevárez MC, Calderón de la Barca AM, Vásquez-Lara F, Malunga LN, Apea-Bah FB, Beta T, Islas-Rubio AR (2020) Phenolic acids, antioxidant capacity, and estimated glycemic index of cookies added with brewer’s spent grain. Plant Foods Hum Nutr 75:41–47. https://doi.org/10.1007/s11130-019-00783-1
Jiao Z, Zhu D, Yao W (2015) Combination of accelerated solvent extraction and micro-solid-phase extraction for determination of trace antibiotics in food samples. Food Anal Methods 8:2163–2168. https://doi.org/10.1007/s12161-015-0105-y
Kampa M, Alexaki VI, Notas G et al (2004) Antiproliferative and apoptotic effects of selective phenolic acids on T47D human breast cancer cells: potential mechanisms of action. Breast Cancer Res 6:R63–R74. https://doi.org/10.1186/bcr752
Li JB, Guo Z (2016) Identification and quantification of phenolic compounds in rapeseed originated lecithin and antioxidant activity evaluation. LWT 73:397–405. https://doi.org/10.1016/j.lwt.2016.06.039
Li Q, Li Q, Tan WQ, Zhang JJ, Guo ZY (2020) Phenolic-containing chitosan quaternary ammonium derivatives and their significantly enhanced antioxidant and antitumor properties. Carbohyd Res 498:108169. https://doi.org/10.1016/j.carres.2020.108169
Liu Z, Zhang JG, Lu SM, Tang WM, Zhou YB, Quek SY (2022) Effects of different drying methods on phenolic components and in vitro hypoglycemic activities of pulp extracts from two Chinese bayberry (Myrica rubra Sieb. et Zucc.) cultivars. Food Sci Hum Well 11(2):366–373. https://doi.org/10.1016/j.fshw.2021.11.014
López-Linares JC, Ballesteros I, Tourán J, Cara C, Castro E, Ballesteros M, Romero I (2015) Optimization of uncatalyzed steam explosion pretreatment of rapeseed straw for biofuel production. Bioresour Technol 190:97–105. https://doi.org/10.1016/j.biortech.2015.04.066
Luo D, Mu TH, Sun HN (2021) Profiling of phenolic acids and flavonoids in sweet potato (Ipomoea batatas L.) leaves and evaluation of their anti-oxidant and hypoglycemic activities. Food Biosci 39:100801. https://doi.org/10.1016/j.fbio.2020.100801
Magalhães M, Manadas B, Efferth T, Cabral C (2021) Chemoprevention and therapeutic role of essential oils and phenolic compounds: modeling tumor microenvironment in glioblastoma. Pharmacol Res 169:105638. https://doi.org/10.1016/j.phrs.2021.105638
Mandal SM, Mandal M, Das AK, Pati BR, Ghosh AK (2009) Stimulation of indoleacetic acid production in a Rhizobium isolate of Vigna mungo by root nodule phenolic acids. Arch Microbiol 191:395. https://doi.org/10.1007/s00203-009-0461-3
Méndez-Lagunas LL, Cruz-Gracida M, Barriada-Bernal LG, Rodríguez-Méndez LI (2020) Profile of phenolic acids, antioxidant activity and total phenolic compounds during blue corn tortilla processing and its bioaccessibility. J Food Sci Tech 57:4688–4696. https://doi.org/10.1007/s13197-020-04505-3
Mikołajczak N, Tańska M, Ogrodowska D (2021) Phenolic compounds in plant oils: a review of composition, analytical methods, and effect on oxidative stability. Trends Food Sci Tech 113:110–138. https://doi.org/10.1016/j.tifs.2021.04.046
Naczk M, Pink J, Zadernowski R, Pink D (2002) Multivariate model for the prediction of total phenolic acids in crude extracts of polyphenols from canola and rapeseed meals: a preliminary study. J Am Oil Chem Soc 79(8):759–762. https://doi.org/10.1007/s11746-002-0555-x
Pang L, Yang P, Ge L, Du JJ, Zhang HZ (2017) Accelerated solvent extraction combined with solid phase extraction for the determination of organophosphate esters from sewage sludge compost by UHPLC-MS/MS. Anal Bioanal Chem 409:1435–1440. https://doi.org/10.1007/s00216-016-0078-8
Śliwiński L, Pytlik M, Zych M, Cegieła U, Nowińska B, Kaczmarczyk-Sedlak I, Folwarczna J (2013) Effects of major active components of coffee, caffeine and phenolic acids, on osteoclastogenesis and osteoblast activity in vitro. Pharmacol Rep 65:91. https://doi.org/10.1016/S1734-1140(13)71394-X
Sun Y, Hui QR, Chen R, Li HY, Peng H, Chen F, Deng ZY (2018) Apoptosis in human hepatoma HepG2 cells induced by the phenolics of Tetrastigma hemsleyanum leaves and their antitumor effects in H22 tumor-bearing mice. J Funct Foods 40:349–364. https://doi.org/10.1016/j.jff.2017.11.017
Szydłowska-Czerniak A, Rabiej D, Kyselka J, Dragoun M, Filip V (2018) Antioxidative effect of phenolic acids octyl esters on rapeseed oil stability. LWT 96:193–198. https://doi.org/10.1016/j.lwt.2018.05.033
Wang B, Zhao X, **e X, **e KZ, Zhang GX, Zhang T, Liu XZ (2019) Development of an accelerated solvent extraction approach for quantitative analysis of chloramphenicol, thiamphenicol, florfenicol, and florfenicol amine in poultry eggs. Food Anal Methods 12:1705–1714. https://doi.org/10.1007/s12161-019-01517-4
Wu XQ, Ge XS, Liang SX, Lv YK, Sum HW (2015) A novel selective accelerated solvent extraction for effective separation and rapid simultaneous determination of six anthraquinones in Tartary Buckwheat and its products by UPLC-DAD. Food Anal Methods 8:1124–1132. https://doi.org/10.1007/s12161-014-9976-6
**e LS, Wang GH, Sang W, Li J, Zhang Z, Li WX, Yan J, Zhao Q, Dai YL (2021) Phenolic immunogenic cell death nanoinducer for sensitizing tumor to PD-1 checkpoint blockade immunotherapy. Biomaterials 269:120638. https://doi.org/10.1016/j.biomaterials.2020.120638
Yates K, Pohl F, Busch M, Mozer A, Watters L, Shiryaev A, Kong P, Lin L (2019) Determination of sinapine in rapeseed pomace extract: its antioxidant and acetylcholinesterase inhibition properties. Food Chem 276:768–775. https://doi.org/10.1016/j.foodchem.2018.10.045
Yener I, Kocakaya SO, Ertas A, Erhan B, Kaplaner E, Oral EV, Yilmaz-Ozden T, Yilmaz MA, Ozturk M, Kolak U (2020) Selective in vitro and in silico enzymes inhibitory activities of phenolic acids and flavonoids of food plants: relations with oxidative stress. Food Chem 327:127045. https://doi.org/10.1016/j.foodchem.2020.127045
Zago E, Lecomte J, Barouh N, Aouf C, Carré P, Fine F, Villeneuve P (2015) Influence of rapeseed meal treatments on its total phenolic content and composition in sinapine, sinapic acid and canolol. Ind Crop Prod 76:1061–1070. https://doi.org/10.1016/j.indcrop.2015.08.022
Zhu JF, Zhang DX, Tang HY, Zhao GH (2018) Structure relationship of non-covalent interactions between phenolic acids and arabinan-rich pectic polysaccharides from rapeseed meal. Int J Biol Macromol 120:2597–2603. https://doi.org/10.1016/j.ijbiomac.2018.09.036
Funding
This work was supported by the School Cultivation Subject of Heilongjiang Bayi Agricultural University (No.PTJH201905, ZRCQC201910).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Conflict of Interest
Peng Sun declares that he has no conflict of interest. Chao Wang declares that he has no conflict of interest. Lei Xu declares that she has no conflict of interest. Yuling Gao declares that she has no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, P., Wang, C., Xu, L. et al. Simultaneous Determination of Eight Phenolic Acids in Rapeseed by Accelerated Solvent Extraction-Solid Phase Extraction Ultra-High-Performance Liquid Chromatography-Tandem Mass Spectrometry. Food Anal. Methods 15, 2625–2632 (2022). https://doi.org/10.1007/s12161-022-02310-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-022-02310-6