Abstract
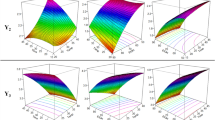
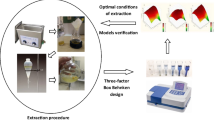
The use of statistical tools in the optimization of bioactive extraction processes increases efficiency and reduces process costs, indicating the optimal conditions for obtaining the best results. The objective of this work was to optimize an ultrasound-assisted extraction procedure and to quantify phenolic compounds in Capsicum frutescens samples. In the optimization of the extraction solution, simplex-centroid mixture design for the proportions optimization of the extractor components (water, methanol, and ethanol) was used. A two-level factorial design (23) was used for the preliminary evaluation of the factors that influenced the extraction process; before this step, a Box-Behnken design was carried out to optimize the factors of solvent volume, sonication time, and temperature with the desirability function as a response. The best mixture composition was 95% methanol and 5% water and the best experimental conditions for the process variables were 16 mL, 15 min, and 55 °C. Applying the method to pepper samples, twelve compounds were determined by HPLC–DAD. The relative standard deviations (RSD) found for all analytes were lower than 3.0%. The accuracy was evaluated using addition/recovery test with recoveries between 81 and 95%. The Box-Behnken and mixture designs are efficient tools for optimizing the analytical method using ultrasound to extract phenolic compounds from C. frutescens. The procedure was applied for extraction phenolics compounds in three papers samples and the concentrations found were trans-cinnamic acid (< LOQ-23 µg g−1), gallic acid (0.94–1.56 mg g−1), quercetin (< LOQ-0.11 mg g−1), syringic acid (< LOQ 0.24 mg g−1), ellagic acid (2.2–3.1 mg g−1), catechin (1.7–3.24 mg g−1), ferulic acid (< LOQ 1.1 mg g−1), p-coumaric acid (57–67 µg g−1), chlorogenic acid (0.12–0.20 mg g−1), caffeic acid (0.090–0.12 mg g−1), vanillic acid (1.1–2.7 mg g−1), and kaempferol (< LOQ 1.2 mg g−1).




Similar content being viewed by others
References
Al-Dhabi NA, Ponmurugan K, Jeganathan PM (2017) Development and validation of ultrasound-assisted solid-liquid extraction of phenolic compounds from waste spent coffee grounds. Ultrason Sonochem 34:206–213. https://doi.org/10.1016/j.ultsonch.2016.05.005
Ashraf MA, Iqbal MA, Rasheed R, Hussain I, Riaz M, Arif MS (2018) Environmental stress and secondary metabolites in plants: an overview. Ahmad P, Ahanger MA, Singh VP, Tripathi DK, Alam P, Alyemeni MN (eds.) Plant Metabolites and Regulation Under Environmental Stress. Academic Press, London, pp. 153–167. https://doi.org/10.1016/B978-0-12-812689-9.00008-X
Baenas N, Belović M, Ilic N, Moreno DA, García-Viguera C (2019) Industrial use of pepper (Capsicum annum L.) derived products: technological benefits and biological advantages. Food Chem. 274:872–885. https://doi.org/10.1016/j.foodchem.2018.09.047
Dahmoune F, Spigno G, Moussi K, Remini H, Cherbal A, Madani K (2014) Pistacia lentiscus leaves as a source of phenolic compounds: microwave-assisted extraction optimized and compared with ultrasound-assisted and conventional solvent extraction. Ind Crops Prod 61:31–40. https://doi.org/10.1016/j.indcrop.2014.06.035
de Sousa CBdoC, dos Anjos GL, Nóbrega RSA, Magaton AdaS, de Miranda FM, Dias FdeS (2020) Greener ultrasound-assisted extraction of bioactive phenolic compounds in Croton heliotropiifolius Kunth leaves. Microchem J: 105525. https://doi.org/10.1016/j.microc.2020.105525
Dias F de S, dos Santos WNL, Costa ACS, Welz B, Vale MGR, Ferreira SLC (2007) Application of multivariate techniques for optimization of direct method for determination of lead in naphtha and petroleum condensate by electrothermal atomic absorption spectrometry. Microchimica Acta. 158:321–326. https://doi.org/10.1007/s00604-006-0705-8
Dias FS, Klassen A, Tavares MFM, David JM (2013) Fast determination of phenolic compounds in Brazilian wines from Vale do São Francisco region by CE. Chromatographia 76:559–563. https://doi.org/10.1007/s10337-013-2399-5
Dias FS, David JM, David JP (2016) Determination of phenolic acids and quercetin in Brazilian red wines from Vale do São Francisco region using liquid-liquid ultrasound-assisted extraction and HPLC-DAD-MS. J Braz Chem Soc 26:1055–1059. https://doi.org/10.5935/0103-5053.20150363
Dias ALB, Sergio CSA, Santos P, Barbero GF, Rezende CA, Martínez J (2017) Ultrasound-assisted extraction of bioactive compounds from dedo de moça pepper (Capsicum baccatum L.): effects on the vegetable matrix and mathematical modeling. J. Food Eng. 198:36–44. https://doi.org/10.1016/j.jfoodeng.2016.11.020
Ferreira SLC, Lemos VA, Carvalho VS, Silva EGP, Queiroz AFS, Felix CSA, Silva DLF, Dourado GB, Oliveira RV (2018) Multivariate optimization techniques in analytical chemistry-an overview. Microchem J 140:176–182. https://doi.org/10.1016/j.microc.2018.04.002
Goldsmith CD, Vuong QV, Stathopoulos CE, Roach PD, Scarlett CJ (2018) Ultrasound increases the aqueous extraction of phenolic compounds with high antioxidant activity from olive pomace. LWT-Food Sci Technol 89:284–290. https://doi.org/10.1016/j.lwt.2017.10.065
Gomes SVF, Portugal LA, Anjos JP, Jesus ON, Oliveira EJ, David JP, David JM (2017) Accelerated solvent extraction of phenolic compounds exploiting a Box-Behnken design and quantification of five flavonoids by HPLC-DAD in Passiflora species. Microchem J 132:28–35. https://doi.org/10.1016/j.microc.2016.12.021
Huang HW, Hsu CP, Yang BB, Wang CY (2013) Advances in the extraction of natural ingredients by high pressure extraction technology. Trends Food Sci Technol 33:54–62. https://doi.org/10.1016/j.tifs.2013.07.001
Ibiza VP, Blanca J, Cañizares J, Nuez F (2012) Taxonomy and genetic diversity of domesticated Capsicum species in the Andean region. Genet Resour Crop Evol 59:1077–1088. https://doi.org/10.1007/s10722-011-9744-z
Ince AE, Sahin S, Sumnu G (2014) Comparison of microwave and ultrasound-assisted extraction techniques for leaching of phenolic compounds from nettle. J Food Sci Technol: 2776–2782. https://doi.org/10.1007/s13197-012-0828-3
Irakli M, Chatzopoulou P, Ekateriniadou L (2018) Optimization of ultrasound-assisted extraction of phenolic compounds: oleuropein, phenolic acids, phenolic alcohols and flavonoids from olive leaves and evaluation of its antioxidant activities. Ind Crops Prod 124:382–388. https://doi.org/10.1016/j.indcrop.2018.07.070
Li J, Ma C, Ma Y, Li Y, Zhou W, Xu P (2007) Medium optimization by combination of response surface methodology and desirability function: an application in glutamine production. Appl Microbiol Biotechnol 74:563–571. https://doi.org/10.1007/s00253-006-0699-5
Marques GS, Monteiro RPM, Leão WF, Lyra MAM, Peixoto MS, Rolim-Neto PJ, ..., Soares LA (2012) Avaliação de procedimentos para quantificação espectrofotométrica de flavonoides totais em folhas de Bauhinia forficata Link, Quim. Nova 35(3): 517–522. https://doi.org/10.1590/S0100-40422012000300014
Moreira GC, Dias FS (2018) Mixture design and Doehlert matrix for optimization of the ultrasonic assisted extraction of caffeic acid, rutin, catechin and trans-cinnamic acid in Physalis angulata L. and determination by HPLC DAD. Microchem J 141:247–252. https://doi.org/10.1016/j.microc.2018.04.035
Muñiz-Márquez DB, Martínez-Ávila GC, Wong-Paz JE, Belmares-Cerda R, Rodríguez-Herrera R, Aguilar CN (2013) Ultrasound-assisted extraction of phenolic compounds from Laurus nobilis L. and their antioxidant activity, Ultrason. Sonochem: 1149–1154. https://doi.org/10.1016/j.ultsonch.2013.02.008
Pico Y (2013) Ultrasound-assisted extraction for food and environmental samples. Trends Analyt Chem 43:84–99. https://doi.org/10.1016/j.trac.2012.12.005
Pimentel-Moral S, Borrás-Linares I, Lozano-Sánchez J, Arráez-Román D, Martínez-Férez A, Segura-Carretero A (2018) Microwave-assisted extraction for Hibiscus sabdariffa bioactive compounds. J Pharm Biomed Anal 156:313–322. https://doi.org/10.1016/j.jpba.2018.04.050
Purohit AJ, Gogate PR (2015) Ultrasound-assisted extraction of β-carotene from waste carrot residue: effect of operating parameters and type of ultrasonic irradiation. Sep Sci Technol 50(10):1507–1517. https://doi.org/10.1080/01496395.2014.978472
Safdar MN, Kausar T, Jabbar S, Mumtaz A, Ahad K, Saddozai AA (2017) Extraction and quantification of polyphenols from kinnow (Citrus reticulate L.) peel using ultrasound and maceration techniques. J Food Drug Anal 25:488–500. https://doi.org/10.1016/j.jfda.2016.07.010
Sahin S, Samli R (2013) Optimization of olive leaf extract obtained by ultrasound assisted extraction with response surface methodology. Ultrason Sonochem 20:595–602. https://doi.org/10.1016/j.ultsonch.2012.07.029
Šamec D, Valek-Žulj L, Martinez S, Grúz J, Piljac A, Piljac-Žegarac J (2016) Phenolic acids significantly contribute to antioxidant potency of Gynostemma pentaphyllum aqueous and methanol extracts. Ind Crops Prod 84:104–107. https://doi.org/10.1016/j.indcrop.2016.01.035
Santos WNL, Sauthier MCS, Santos AMP, Santana DA, Azevedo RSA, Caldas JC (2017) Microchem J 133:583–592. https://doi.org/10.1016/j.microc.2017.04.029
Slinkard K, Singleton VL (1977) Total phenol analyses: automation and comparison with manual methods. Am J Enol Viticult 28:49–55
Tian SL, Lu BY, Gong ZH, Muhammad-Shah SN (2014) Effects of drought stress on capsanthin during fruit development and ripening in pepper (Capsicum annuum L.). Agric Water Manage. 137:46–51. https://doi.org/10.1016/j.agwat.2014.02.007
Veljković VB, Veličković AV, Avramović JM, Stamenković OS (2018) Modeling of biodiesel production: performance comparison of box–Behnken, face central composite or full factorial design. Chin J Chem Eng 27:1690–1698. https://doi.org/10.1016/j.cjche.2018.08.002
Wang X, Wu Y, Chen G, Yue W, Liang Q, Wu Q (2013) Optimisation of ultrasound assisted extraction of phenolic compounds from Sparganii rhizoma with response surface methodology. Ultrason Sonochem 20:846–854. https://doi.org/10.1016/j.ultsonch.2012.11.007
Wen C, Zhang J, Zhang H, Dzah CS, Zandile M, Duan Y, Ma H, Luo X (2018) Advances in ultrasound assisted extraction of bioactive compounds from cash crops - a review. Ultrason Sonochem 48:538–549. https://doi.org/10.1016/j.ultsonch.2018.07.018
Yahya NA, Attan N, Wahab RA (2018) An overview of cosmeceutically relevant plant extracts and strategies for extraction of plant-based bioactive compounds. Food Bioprod Process 112:69–85. https://doi.org/10.1016/j.fbp.2018.09.002
Zhang QW, Lin LG, Ye WC (2018) Techniques for extraction and isolation of natural products: a comprehensive review. Chin Med 13(20):1–26. https://doi.org/10.1186/s13020-018-0177-x
Zhao Y, Hou Y, Tang G, Cai E, Liu S, Yang H, Zhang L, Wang S (2014) Optimization of ultrasonic extraction of phenolic compounds from Epimedium brevicornum Maxim using response surface methodology and evaluation of its antioxidant activities in vitro. J Anal Methods Chem: 1–7. https://doi.org/10.1155/2014/864654
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Conflict of Interest
The author Gilvanda Leão dos Anjos declares that she has no conflict of interest. The author Candice N. Carneiro declares that she has no conflict of interest. The author Fabio de Souza Dias declares that he has no conflict of interest. Gisele C. Moreira declares that she has no conflict of interest. Jamile da C. Caldas declares that she has no conflict of interest. Isaac M. de J. Silva declares that he has no conflict of interest. Walter N. L. dos Santos declares that he has no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
dos Anjos, G.L., Moreira, G.C., Carneiro, C.N. et al. Multivariate optimization of an ultrasound-assisted extraction method of bioactive phenolic compounds in malagueta peppers (Capsicum frutescens) . Food Anal. Methods 14, 2607–2616 (2021). https://doi.org/10.1007/s12161-021-02088-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-021-02088-z