Abstract
Objective
Radium-223 (Ra-223) is an important treatment modality for bone-dominant metastatic castration-resistant prostate cancer (mCRPC). However, there is currently a lack of effective markers to monitor treatment response during treatment. We aim to investigate the response in prostate-specific antigen doubling time (PSADT) as a potential marker for assessing Ra-223 treatment in mCRPC patients.
Methods
We retrospectively collected data from mCRPC patients who underwent radium treatment at our institution between August 2020 and June 2023. Prostate-specific antigen (PSA) measurements prior to treatment and during treatment were collected. Baseline PSADT was calculated from PSA measurements prior to Ra-223 treatment; interim PSADT was calculated from PSA measurements before Ra-223 treatment and prior to the fourth course injection. Overall survival was calculated from the start of treatment to the date of death. Univariable and multivariable analysis using the Cox proportional hazards model were performed to examine the association of factors with overall survival.
Results
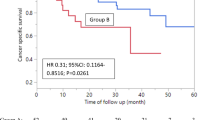
We included 35 patients from our institution, with a median overall survival of 13.3 months. Eighteen (51.4%) completed all six courses of treatment. PSA dynamic response (interim PSADT > baseline PSADT or decreased PSA) was observed in 20 patients. Overall survival was associated with a PSA dynamic response (HR = 0.318, 95% CI 0.133–0.762, p = 0.010) when compared to patients without response.
Conclusions
Dynamic changes in PSADT were associated with survival in mCRPC patients receiving radium therapy. Comparing interim and baseline PSADT could serve as a valuable marker for determining treatment benefits.



Similar content being viewed by others
Data availability
HL had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Data are available for bona fide researchers who request it from the authors.
References
SEER*Explorer: An interactive website for SEER cancer statistics. Surveillance Research Program, National Cancer Institute. 2023.
Gandaglia G, Abdollah F, Schiffmann J, Trudeau V, Shariat SF, Kim SP, et al. Distribution of metastatic sites in patients with prostate cancer: a population-based analysis. Prostate. 2014;74:210–6.
Parker C, Nilsson S, Heinrich D, Helle SI, O’Sullivan JM, Fosså SD, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369:213–23.
Dizdarevic S, Jessop M, Begley P, Main S, Robinson A. (223)Ra-Dichloride in castration-resistant metastatic prostate cancer: improving outcomes and identifying predictors of survival in clinical practice. Eur J Nucl Med Mol Imaging. 2018;45:2264–73.
Lopes F, Pedras MV, Ferreira ÁRS, Ledesma PM, Dias P, Pedras FV. Behavior of total alkaline phosphatase after radium-233 therapy in metastatic castration-resistant prostate cancer: a single-center, real-world retrospective study. Radiol Bras. 2023;56:125–30.
Viani G. Insights into the prognostic potential of total alkaline phosphatase in metastatic castration-resistant prostate cancer treated with radium-223. Radiol Bras. 2023. https://doi.org/10.1590/0100-3984.2023.56.3e2.
Pound CR, Partin AW, Eisenberger MA, Chan DW, Pearson JD, Walsh PC. Natural history of progression after PSA elevation following radical prostatectomy. JAMA. 1999;281:1591–7.
Badrising SK, Louhanepessy RD, van der Noort V, Coenen J, Hamberg P, Beeker A, et al. A prospective observational registry evaluating clinical outcomes of Radium-223 treatment in a nonstudy population. Int J Cancer. 2020;147:1143–51.
Castello A, Macapinlac HA, Lopci E, Santos EB. Prostate-specific antigen flare induced by (223)RaCl(2) in patients with metastatic castration-resistant prostate cancer. Eur J Nucl Med Mol Imaging. 2018;45:2256–63.
Freedland SJ, Ramaswamy K, Huang A, Sandin R, Mardekian J, Schultz NM, et al. survival and economic impact of rapid prostate-specific antigen doubling time in patients with nonmetastatic castration-resistant prostate cancer. Clin Genitourin Cancer. 2023;21:419–29.
Hakozaki Y, Yamada Y, Takeshima Y, Taguchi S, Kawai T, Nakamura M, et al. Low hemoglobin and PSA kinetics are prognostic factors of overall survival in metastatic castration-resistant prostate cancer patients. Sci Rep. 2023;13:2672.
Xu XS, Ryan CJ, Stuyckens K, Smith MR, Saad F, Griffin TW, et al. Correlation between prostate-specific antigen kinetics and overall survival in abiraterone acetate-treated castration-resistant prostate cancer patients. Clin Cancer Res. 2015;21:3170–7.
Miyazawa Y, Sekine Y, Shimizu N, Takezawa Y, Nakamura T, Miyao T, et al. An exploratory retrospective multicenter study of prognostic factors in mCRPC patients undergoing enzalutamide treatment: focus on early PSA decline and kinetics at time of progression. Prostate. 2019;79:1462–70.
Hashimoto K, Miyoshi Y, Shindo T, Hori M, Tsuboi Y, Kobayashi K, et al. Dynamic changes of bone metastasis predict bone-predominant status to benefit from radium-223 dichloride for patients with castration-resistant prostate cancer. Cancer Med. 2020;9:8579–88.
Kawaguchi G, Akazawa K, Ikeda T, Ikeda Y, Hara N, Nishiyama T. Prostate-specific antigen doubling time following radium-223 treatment as a predictor of the clinical course in patients with metastatic castration-resistant prostate cancer. SAGE Open Med. 2023;11:20503121231168492.
Sartor O, Coleman RE, Nilsson S, Heinrich D, Helle SI, O’Sullivan JM, et al. An exploratory analysis of alkaline phosphatase, lactate dehydrogenase, and prostate-specific antigen dynamics in the phase 3 ALSYMPCA trial with radium-223. Ann Oncol. 2017;28:1090–7.
van der Doelen MJ, Stockhaus A, Ma Y, Mehra N, Yachnin J, Gerritsen WR, et al. Early alkaline phosphatase dynamics as biomarker of survival in metastatic castration-resistant prostate cancer patients treated with radium-223. Eur J Nucl Med Mol Imaging. 2021;48:3325–34.
James ND, Heinrich D, Castro E, George S, Song DY, Dizdarevic S, et al. Alkaline phosphatase (ALP) decline and overall survival (OS) in patients (pts) with metastatic castration-resistant prostate cancer (mCRPC) treated with radium-223 (Ra-223) in the REASSURE study. J Clin Oncol. 2022. https://doi.org/10.1200/JCO.2022.40.16_suppl.5041.
EMA. EMA Restricts Use of Prostate Cancer Medicine XOFIGO. Available online: https://www.ema.europa.eu/en/news/ema-restricts-use-prostate-cancer-medicine-xofigo. Accessed 16 Oct 2023.
Kitajima K, Igeta M, Kuyama J, Kawahara T, Suga T, Otani T, et al. Novel nomogram developed for determining suitability of metastatic castration-resistant prostate cancer patients to receive maximum benefit from radium-223 dichloride treatment-Japanese Ra-223 Therapy in prostate cancer using bone scan Index (J-RAP-BSI) Trial. Eur J Nucl Med Mol Imaging. 2023;50:1487–98.
Kuppen MC, Westgeest HM, van der Doelen MJ, van den Eertwegh AJ, Coenen JL, Aben KK, et al. Real-world outcomes of radium-223 dichloride for metastatic castration resistant prostate cancer. Future Oncol. 2020;16:1371–84.
Jarvis P, Ho A, Sundram F. Radium-223 therapy for metastatic castration-resistant prostate cancer: survival benefit when used earlier in the treatment pathway. Nucl Med Commun. 2021;42:332–6.
George DJ, Agarwal N, Sartor O, Sternberg CN, Tombal B, Saad F, et al. Real-world patient characteristics associated with survival of 2 years or more after radium-223 treatment for metastatic castration-resistant prostate cancer (EPIX study). Prostate Cancer Prostatic Dis. 2022;25:306–13.
Higano CS, George DJ, Shore ND, Sartor O, Miller K, Conti PS, et al. Clinical outcomes and treatment patterns in REASSURE: planned interim analysis of a real-world observational study of radium-223 in metastatic castration-resistant prostate cancer. EClinicalMedicine. 2023;60: 101993.
Acknowledgements
None.
Funding
No funding was received.
Author information
Authors and Affiliations
Contributions
HL designed the study and performed data extraction; HL and MT performed statistical analysis and wrote the draft of the manuscript; NC provided administrative support. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The study was approved by the Institutional Review Board of National Cheng Kung University Hospital [B-ER-112-204], and the requirement of informed consent was waived due to the retrospective nature of the study.
Consent for publication
During the preparation of this work, the author(s) used ChatGPT to correct spelling and grammatical errors. After using this tool/service, the author(s) reviewed and edited the content as needed and take(s) full responsibility for the content of the publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lu, HH., Chiu, NT. & Tsai, MH. Increase of prostate-specific antigen doubling time predicts survival in metastatic castration-resistant prostate cancer patients undergoing radium therapy. Ann Nucl Med 38, 508–515 (2024). https://doi.org/10.1007/s12149-024-01924-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-024-01924-6